Application of Neogenin in preparation of medicine for preventing, relieving and/or treating myocardial infarction and related diseases thereof
A technology for myocardial infarction and ischemic cardiomyopathy, applied in the relief and/or treatment of drugs for myocardial infarction and its related diseases, multi-functional transmembrane receptor neogenin, in the field of preparation and prevention, can solve problems such as unclear effects , achieve the effect of improving heart function, significant protection, and reducing myocardial remodeling
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
experiment example 1
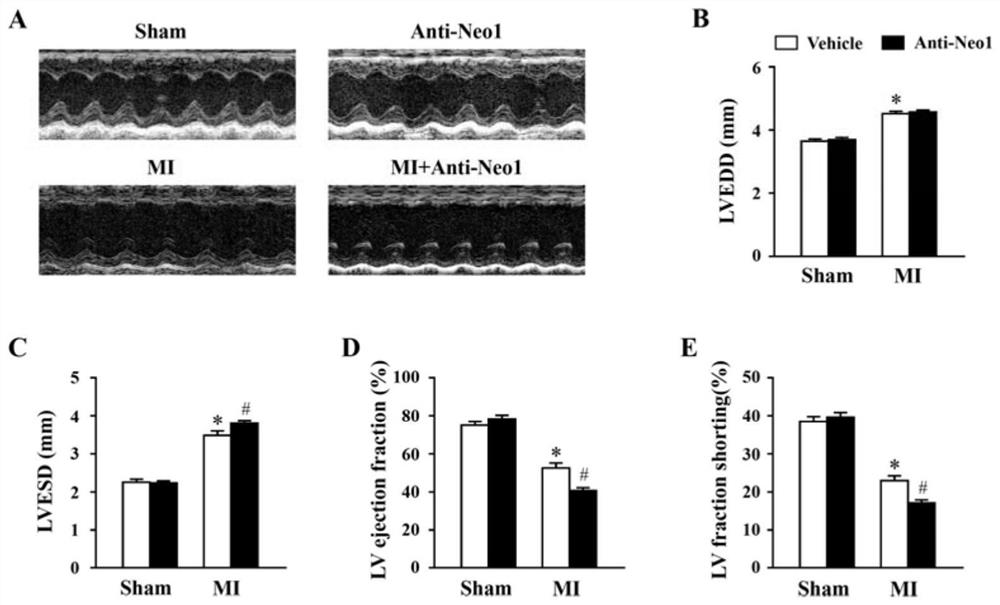
[0028] Experimental Example 1 Neo1 neutralizing antibody aggravates cardiac function after myocardial infarction
[0029] 1. Inject Neo1 neutralizing antibody
[0030]Neo1 neutralizing antibody (Cat. No. AF1079) was purchased from RD Company in the United States. It can effectively bind to the receptor Neo1, block the interaction between Neo1 and its related ligands, and complete the inhibition of Neo1 function in vivo. 2 hours before myocardial infarction, the mice were fixed, and Neo1 neutralizing antibody (the solvent was normal saline) was injected through the tail vein at a dose of 2 μg / mouse, and the control group was given a placebo (vehicle, normal saline). The experiment was divided into 4 groups according to the principle of randomization and blinding: no operation (sham)+vehicle, sham+Anti-Neo1, myocardial infarction group (MI)+vehicle, MI+Anti-Neo1.
[0031] 2. Obtaining the myocardial infarction model
[0032] (1) Anesthetize by intraperitoneal injection of 3% s...
experiment example 2
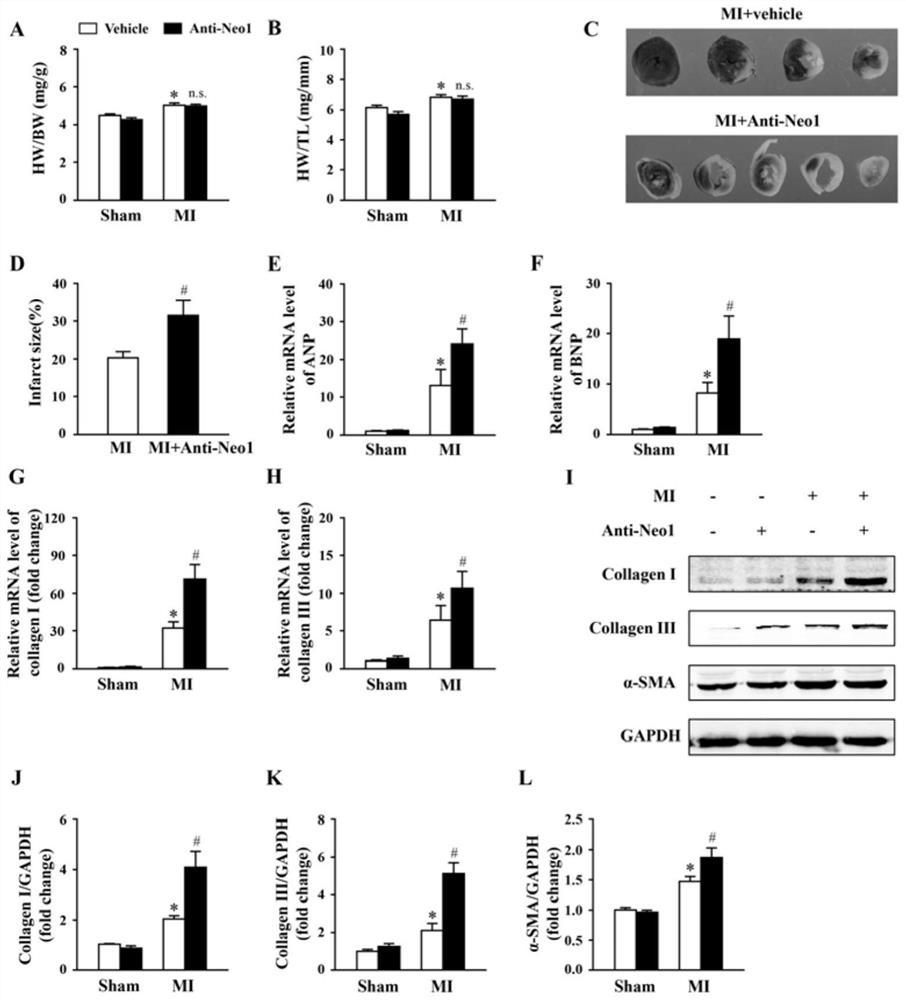
[0042] Experimental Example 2 Neo1 neutralizing antibody increases infarct size and aggravates myocardial remodeling after myocardial infarction
[0043] 1. 2,3,5-Triphenyltetrazolium chloricej (TTC) staining
[0044] (1) After the mice were sacrificed 3 days after myocardial infarction, the cardiac tissue was quickly taken out and placed in a 10% KCl solution.
[0045] (2) After washing the heart, place it in a -20°C refrigerator for 30 minutes.
[0046] (3) Take out the heart tissue, cut it into slices evenly along the long axis of the heart from the apex to the bottom of the heart, immediately place the slices in 10 mL of 2% TTC solution, and incubate at 37°C for 20 min. Normal cardiac tissue stained bright red, while the infarcted area was pale.
[0047] (4) Fix the brain tissue sections with 10% neutral formalin solution, take a general picture, and calculate the infarct size.
[0048] 2. Evaluation of myocardial remodeling after myocardial infarction
[0049] 2.1 Det...
experiment example 3
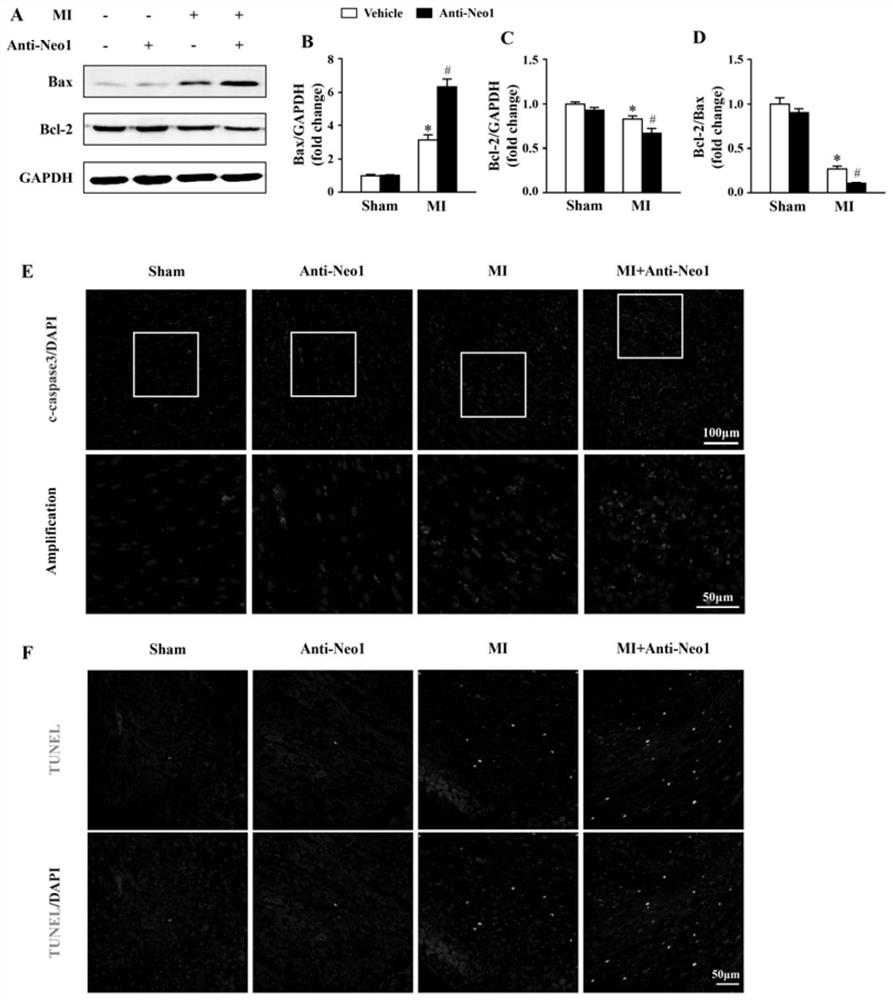
[0110] Experimental Example 3Neo1 Neutralizing Antibody Aggravated Cardiomyocyte Apoptosis After Myocardial Infarction
[0111] 3.1 Western blot monitoring of apoptosis-related proteins
[0112] Protein extraction, electrophoresis gel as above, using primary antibodies Bax, Bcl-2 and GAPDH.
[0113] 3.2 Immunofluorescence
[0114] 3.2.1 Preparation of paraffin specimens and sections
[0115] (1) The mouse heart tissue was removed from the 10% formalin solution, the heart tissue was trimmed in a fume hood, and the trimmed heart tissue and the corresponding label were placed in a dehydration box.
[0116] (2) Dehydration and wax dipping: Put the dehydration box into the dehydrator for dehydration and wax dipping. 75% ethanol (4h)→85% ethanol (2h)→90% ethanol (2h)→95% ethanol (1h)→anhydrous ethanol I (30min)→anhydrous ethanol II (30min)→alcohol benzene (5-10min) )→Xylene I (5-10min)→Xylene II (5-10min)→65°C melted paraffin I (1h)→65°C melted paraffin II (1h)→65°C melted paraf...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More