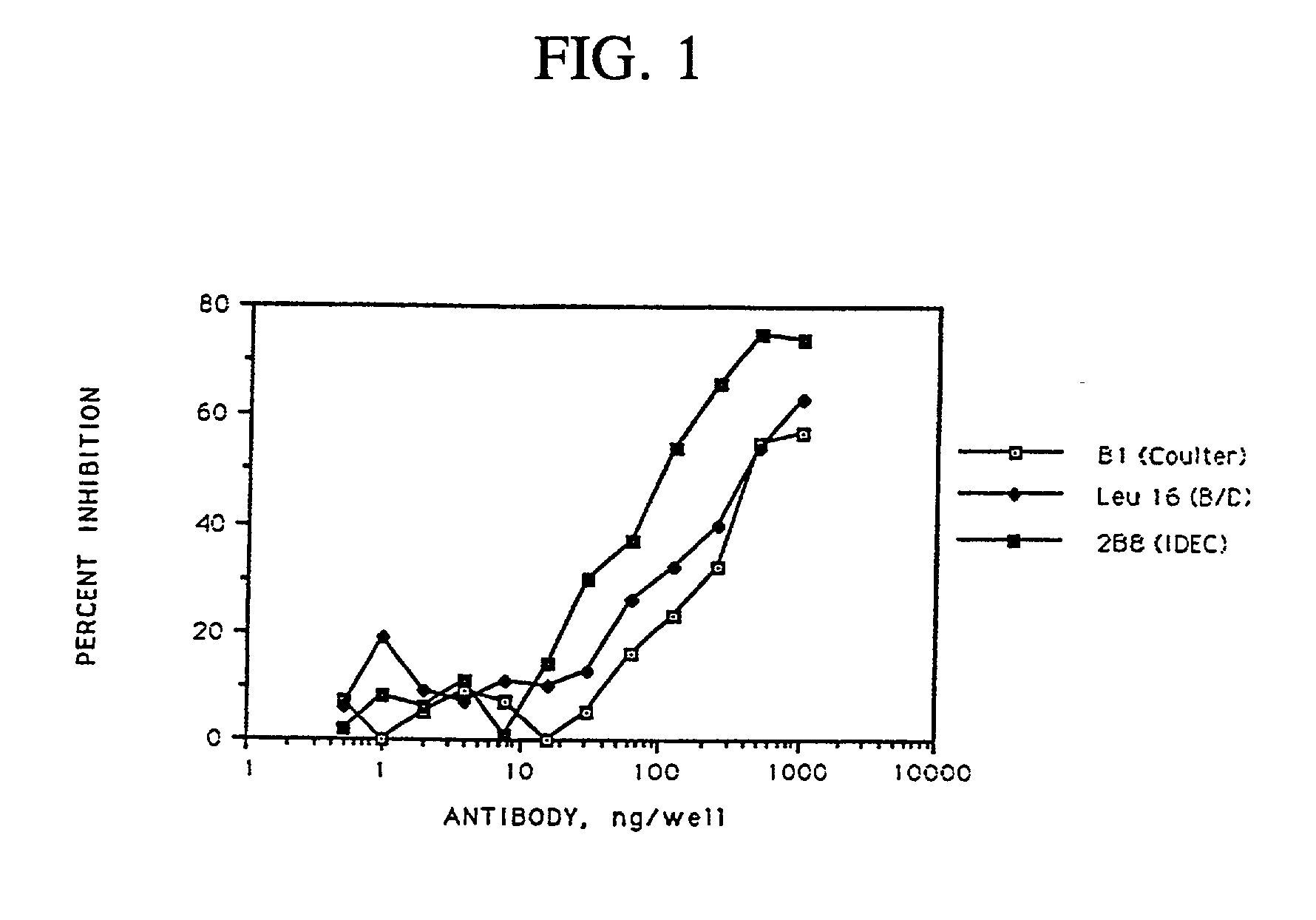
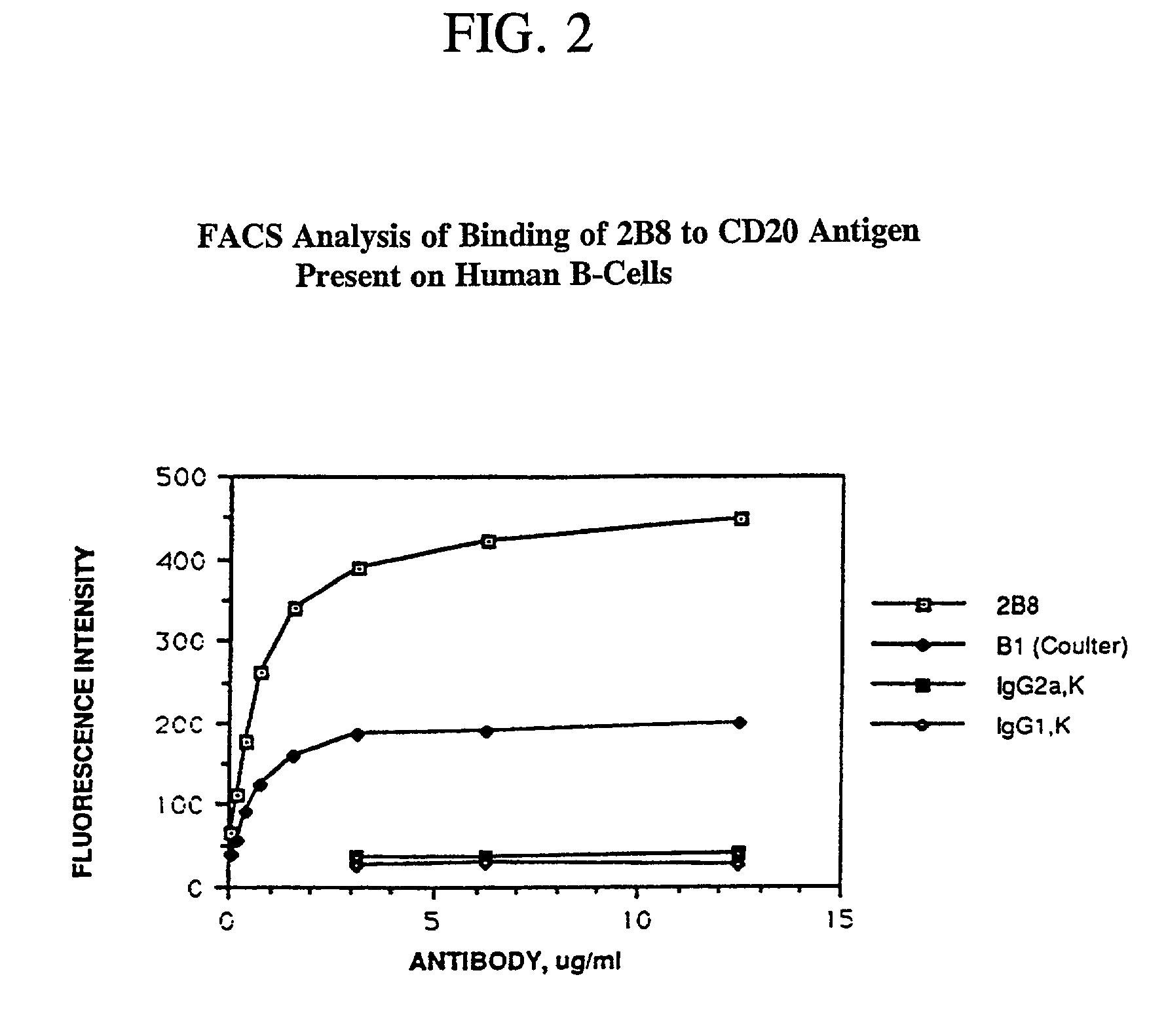
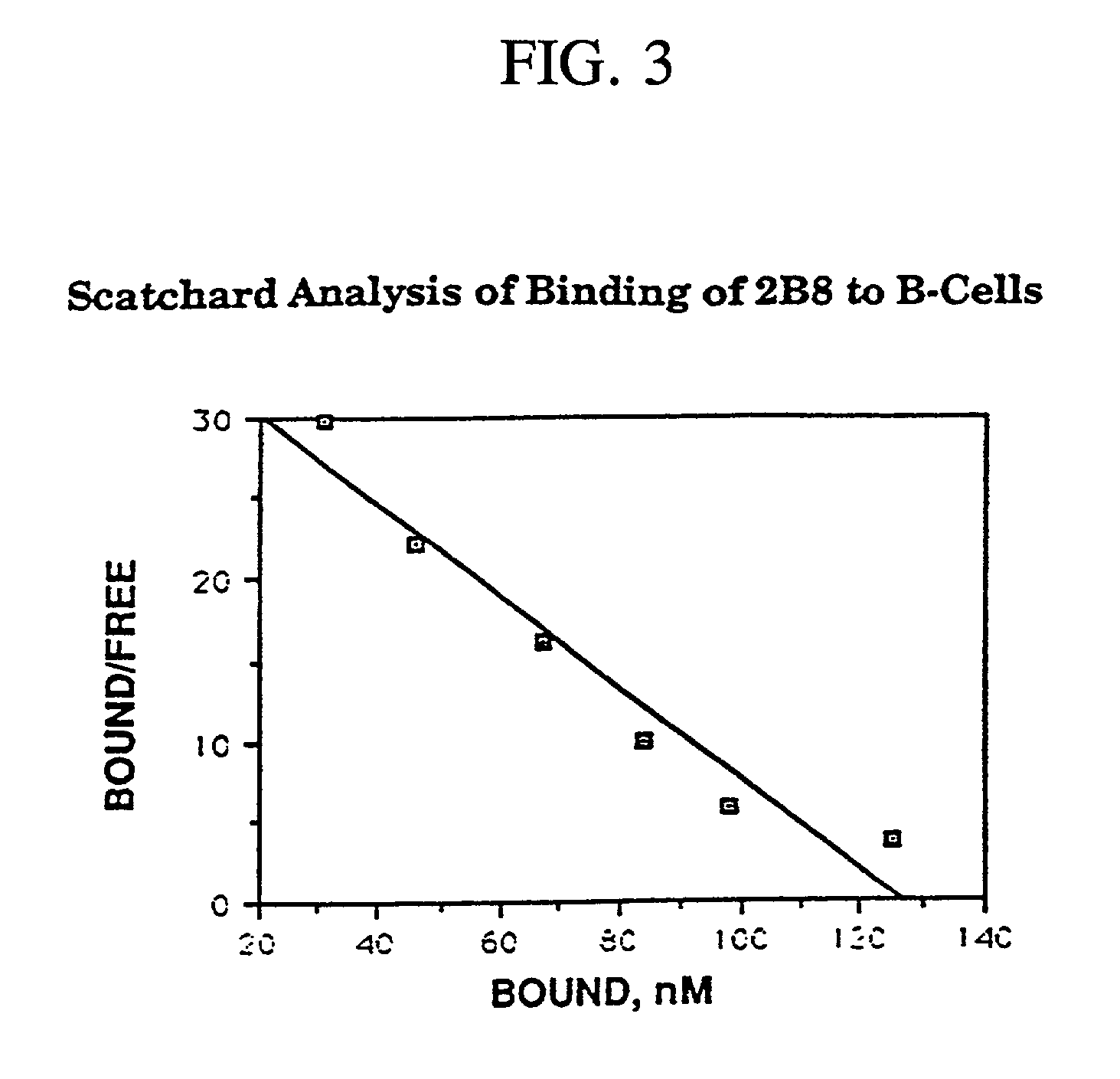
[0280] When the 2B8 antibody was conjugated to methylbenzyl
diethylenetriamine-pentaacetic acid (MX-DTPA), virtually no reduction in immunoreactivity, relative to the native antibody, was observed. Further, radiolabeling the conjugate with either .sup.111In or .sup.90Y produced labeled conjugates with immunoreactivities of 100% and 60%, respectively. Stability studies of .sup.111In or .sup.90Y-labeled conjugates incubated in human serum for 96 hours at 37.degree. C. indicated negligible loss of the radiometal during the course of the study, suggesting that the conjugates will be stable when used clinically.
[0286] A .sup.90Y-labeled murine
monoclonal anti-CD20 antibody (2B8) has been evaluated in a Phase I
clinical trial for the treatment of relapsed B-
cell lymphoma. The original protocol used for the preparation of the
yttrium-labeled antibody used a high performance liquid chromatographic (HPLC) step for removal of non-
protein bound radioisotope prior to formulation and administration to patients. Unfortunately, this process is particularly
time consuming, resulting in a longer
exposure of the antibody to radioisotope in an unprotected state. This results in increased
radiolysis of the antibody with a concomitant decrease in immunoreactivity. Additionally, the laborious aspect of the process makes it difficult to prepare more than one
dose per day at the radiopharmacy. Simplification of the process would expedite implementation at the clinical site as an alternative to using NIPI
Pharmacy Services as a radiopharmacy.
[0299] All manipulations of reagents were performed to minimize the possibility of
metal contamination.
Polypropylene or
polystyrene plastic containers such as flasks, beakers and graduated cylinders were used. These were washed with Alconox and exhaustively rinsed with Milli-Q water or Water for
Irrigation (WFIr) before use.
Metal-free
pipette tips (BioRad) were used for accurately manipulating small volumes. Larger volumes of reagents were manipulated using sterile, plastic serological pipettes. Reactions were conveniently performed in 1.8 mL screw-top microfuge tubes made from
polypropylene.
[0335] The original radiolabeling protocol for preparing .sup.90Y-labeled 2B8-MX-DTPA utilized a particularly laborious and time-consuming HPLC purification step for removing non-
protein bound .sup.90Y from the preparation. In order to simplify this process and make it more amenable to use at the clinical site, efforts were directed at eliminating the HPLC step in favor of what has been termed a "mix-and-
shoot" protocol. The goal was to identify radiolabeling conditions which would result in a very high radioincorporation of
isotope into the conjugate, thereby obviating the need for the purification step. It was discovered that >95% radioincorporation could be-obtained at pH 3.6 with a five to ten minute incubation. An additional benefit of this protocol was increased retention of immunoreactivity (>70%), presumably due to the shorter
exposure time of the antibody to the
high energy radioisotope before the addition of
human serum albumin which provides protection against
radiolysis. This retention of immunoreactivity is superior to that previously observed using the
HPLC method.
[0340] We have developed a labeling procedure, referred to as the "mix-and-
shoot" method, for the preparation of clinical doses of .sup.90Y-labeled 2B8-MX-DTPA which obviates the need for the currently used high performance liquid chromatographic (HPLC) step for removal of non-
protein bound radioisotope. The simplified protocol eliminates this laborious purification step while maintaining a high level of radioisotope incorporation (>95%) and improved retention of immunoreactivity (>70%). The clinically-formulated radiolabeled conjugate was found to be stable
in vitro when incubated at 4.degree. C. for 48 hours based on retention of radioisotope and immunoreactivity. Additionally, the radiolabeled conjugate was stable when incubated in human serum at 37.degree. C. for 72 hours.
Biodistribution studies in BALB / c mice demonstrated no unusual tissue deposition, including bone. Estimates of
radiation absorbed doses to a "standard" 70 Kg human were comparable to those obtained in an on-going
clinical trial using .sup.90Y-labeled 2B8-MX-DTPA. The results of these studies showed that .sup.90Y-labeled 2B8-MX-DTPA produced using the "mix-and-
shoot" protocol was comparable to that prepared using the conventional HPLC process. Validation of the scale-up protocol for preparing clinical-grade radiolabeled conjugate showed that the method was reproducible and that the product was comparable to that produced using the current
HPLC method. The results of these pre-clinical studies indicate that this new "mix-&-shoot" protocol can be used to prepare .sup.90Y-labeled 2B8-MX-DTPA suitable for use in clinical trials.
 Login to View More
Login to View More