[0024] The concentration of the nucleic acids of interest and / or any additional nucleic acid is optionally low in the methods of the invention, e.g., about 1 molecule per aliquot. For example, the sample can be diluted to a concentration of about 1 molecule of interest per nanoliter or less. Optionally, diluted aliquots are each diluted to the same degree; however, diluted aliquots can also be differentially diluted (e.g., to form a
dilution series). The volume of the aliquots can be quite low to keep
reagent costs low, e.g., in microfluidic applications. For example, the aliquots can be less than about 100 nl in volume, e.g., less than about 10 nl in volume, or, e.g., about 1 nl in volume or less.
[0031] Optionally, the components of the
system can be treated with one or more reagents between operational runs to reduce cross
contamination between operations. For example, the amplification channel can have acid or base flowed into the channel between amplification reactions to reduce unwanted
contamination from one or more previous amplification products.
[0044] In a preferred embodiment of differentiating lengths of nucleic acids of interest in a sample, an amplification reaction can be used to enhance the sensitivity of the
assay. The nucleic acid of interest is contacted with a first primer pair and a second primer pair having at least one primer that is outside of the sequence defined by the first primer pair, the nucleic acid of interest is amplified in a reaction mixture in a microchannel or microchamber with
polymerase extensions from the primers to produce first amplicons defined by the first primer pair or second amplicons defined by the second primer pair. First and second probes complimentary to the first and second
amplicon and having detectable markers are introduced into the reaction mixture to hybridize with available complimentary sequences, and one or more signals are detected from the probes. Detection of a
signal from only one of the probes indicates a fragmented nucleic acid of interest and detecting signals from both probes indicates a nucleic acid that is not fragmented. In preferred embodiments of this sensitive
assay, the reaction mixture detected contains only a
single copy of the nucleic acid of interest. Reaction mixtures detected in the methods can be homogenous mixtures, e.g., not requiring separation of labeled constituents before detection of signals.
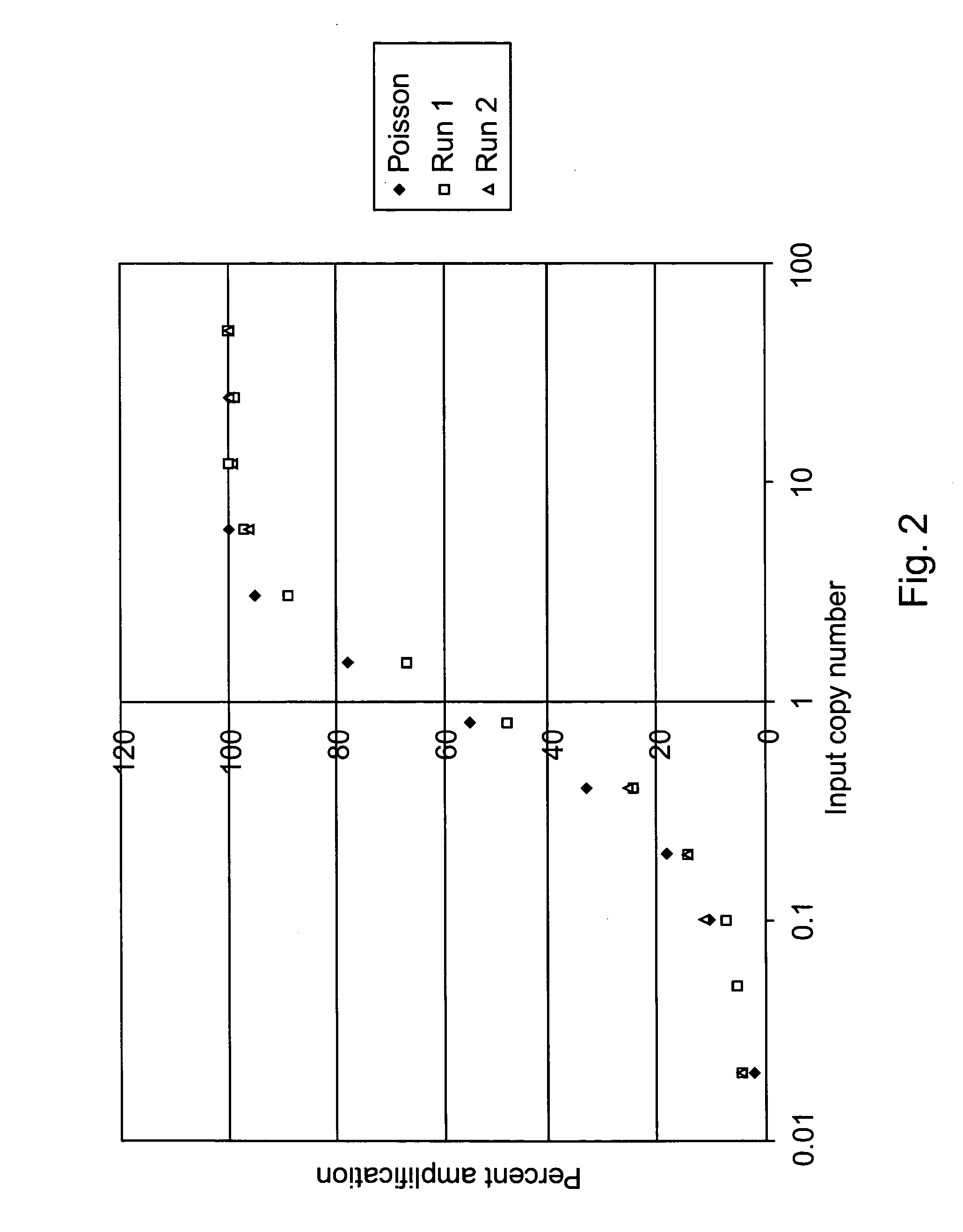
[0051] Samples containing unknown amounts of a nucleic acid of interest can be quantified by comparing to their
signal peaks to sets of standard
signal peaks. For example, a nucleic acid of interest in a sample can be quantified by amplifying a
dilution series of standard materials containing known amounts of the nucleic acid of interest through a certain number of amplification cycles, detecting signals associated with standard amplicons produced from the standard materials, amplifying the sample nucleic acid of interest the number of amplification cycles, detecting a signal associated with sample amplicons produced from the sample nucleic acid of interest, and comparing one or more standard
amplicon signals to the sample
amplicon signal to determine a concentration value for the nucleic acid of interest in the sample. Sample and standard signal parameters for comparison can include, e.g., the shape of their signal peaks, points of inflection on the signal peaks, slopes of the signal peaks, signal peak amplitudes, signal peak areas, signal peak widths at half height, and / or the like. The reliability of results can be enhanced through various schemes of repeated testing. For example, the amplifying, detecting, and comparing steps can be repeated one or more times, with different numbers of amplification cycles, to determine additional concentration values for the sample nucleic acid of interest for statistical evaluation providing more precise or more accurate concentration value results for the nucleic acid of interest in the sample.
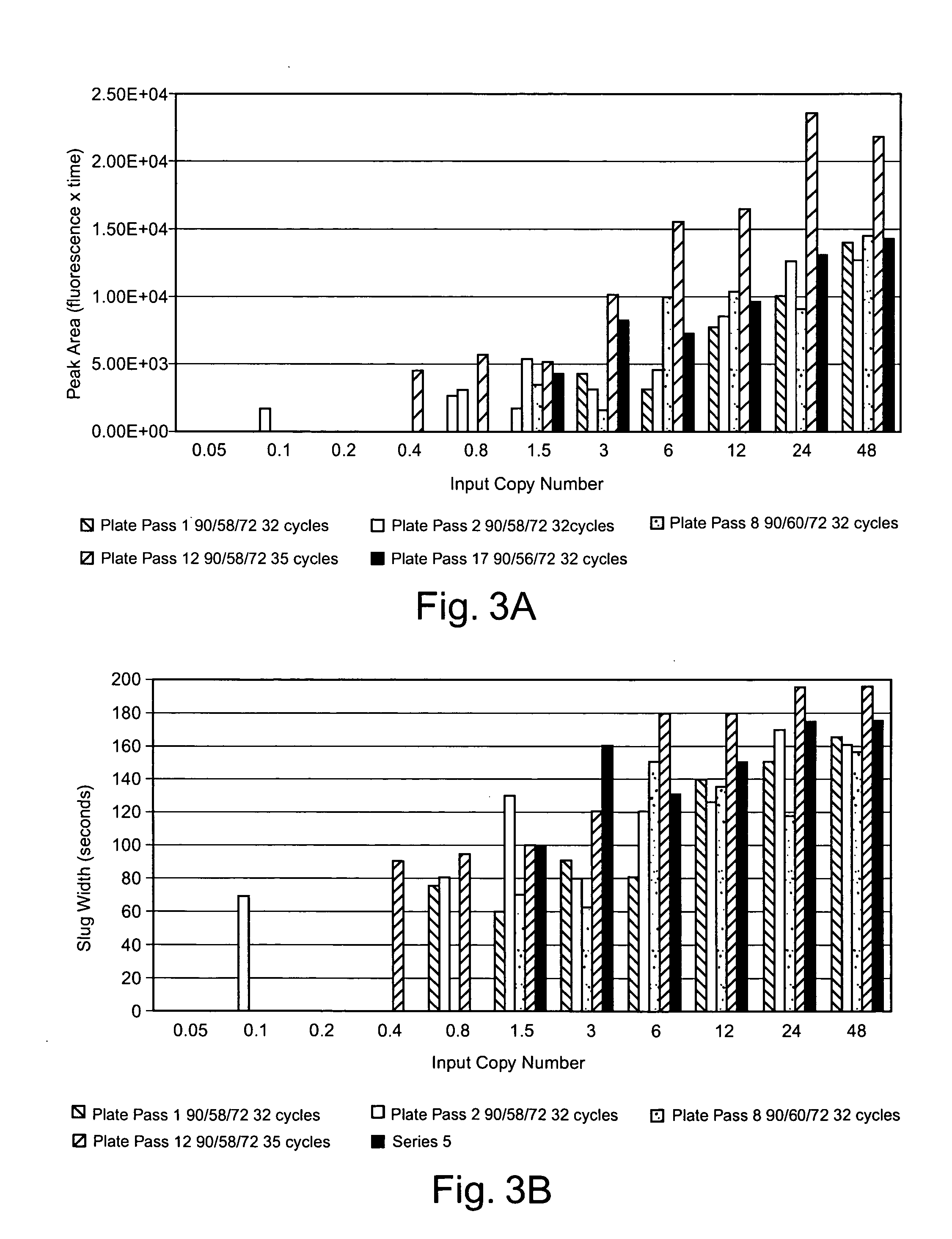
[0052] Improved
assay results can be obtained by gathering signal data after amplifications through two or more different numbers of cycles. A major benefit of running the
quantitative assay at different amplifications is to broaden the
usable range of the assay. Typically, statistical evaluation of the additional data provided by analysis at multiple amplification levels can enhance other assay parameters, such as precision, accuracy, and sensitivity. Quantifying a nucleic acid of interest in a sample based on detection of multiple amplifications can include: amplifying the nucleic acid of interest through more than one number of amplification cycles, detecting signals associated with amplicons produced from two or more of the amplification cycle numbers, preparing a sample curve of a
signal parameter versus number of amplification cycles, and comparing one or more identifiable points from the sample curve to a
standard curve of the identifiable points versus concentration to quantify the nucleic acid of interest. Exemplary identifiable points from signal curves include points of inflection, points having a certain slope, points having a certain signal amplitude, points having a certain fraction of a maximum signal amplitude, and / or the like.
 Login to View More
Login to View More