Cellular immunity inducing vaccine
a cell-mediated immunity and vaccine technology, applied in the field of vaccines, can solve the problems of low antigen presentation efficiency, oil adjuvants, and inability to efficiently produce antitumor, and achieve the effects of high immune-inducing capacity, excellent ability to induce cell-mediated immunity, and high level
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
examples
[0085]The present invention is described below more concretely based on Examples. However, the present invention is not limited to the Examples.
1. Design of Microgenes for Creating Artificial Proteins
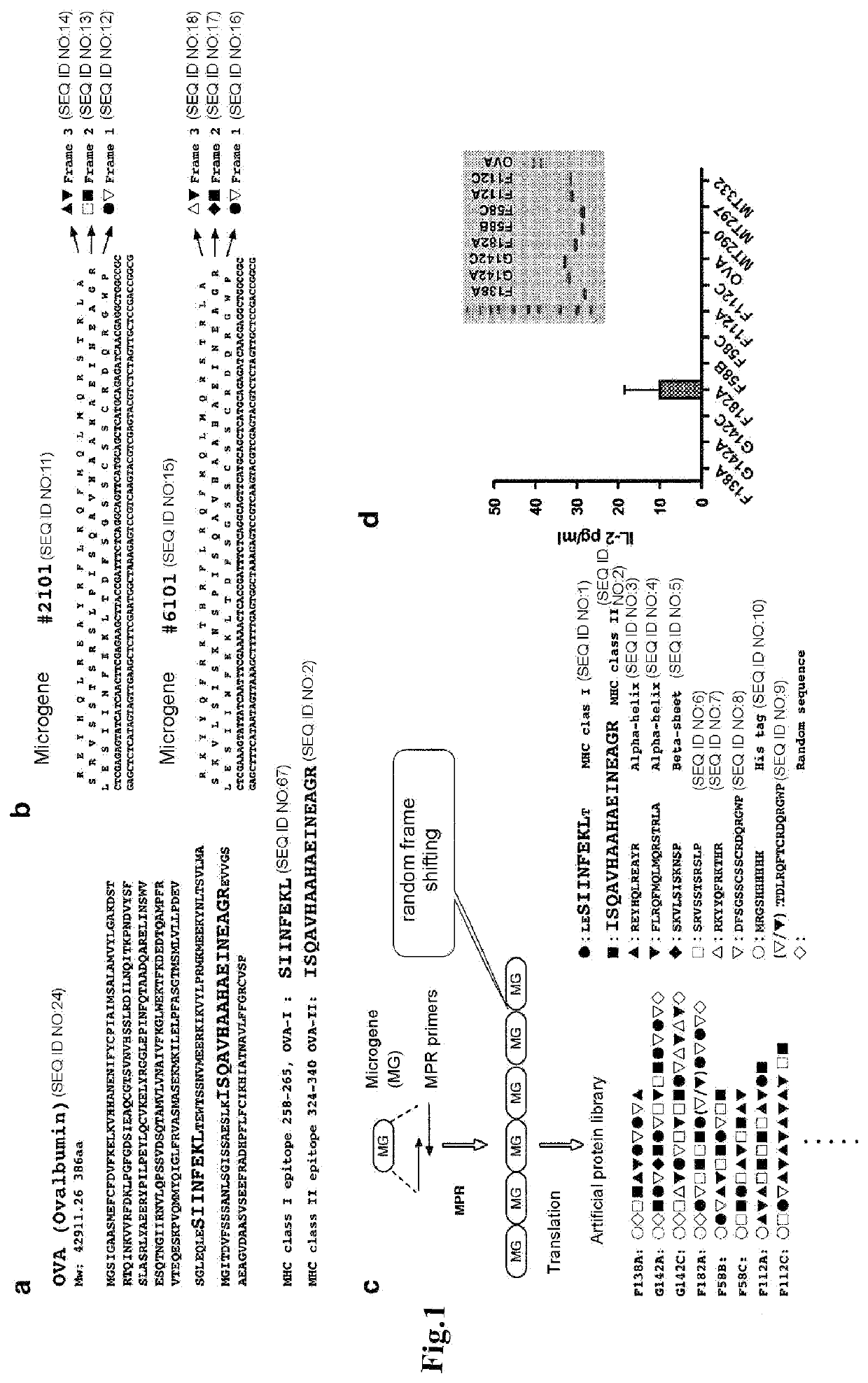
[0086]From native antigen OVA (SEQ ID NO:24), OVA-I: ● (OVA MHC class I epitope, OVA258-265, SIINFEKL; SEQ ID NO: 67) and OVA-II: ▪ (OVA MHC class II epitope, OVA324-340, ISQAVHAAHAEINEAGR; SEQ ID NO: 2) were selected (FIG. 1a).
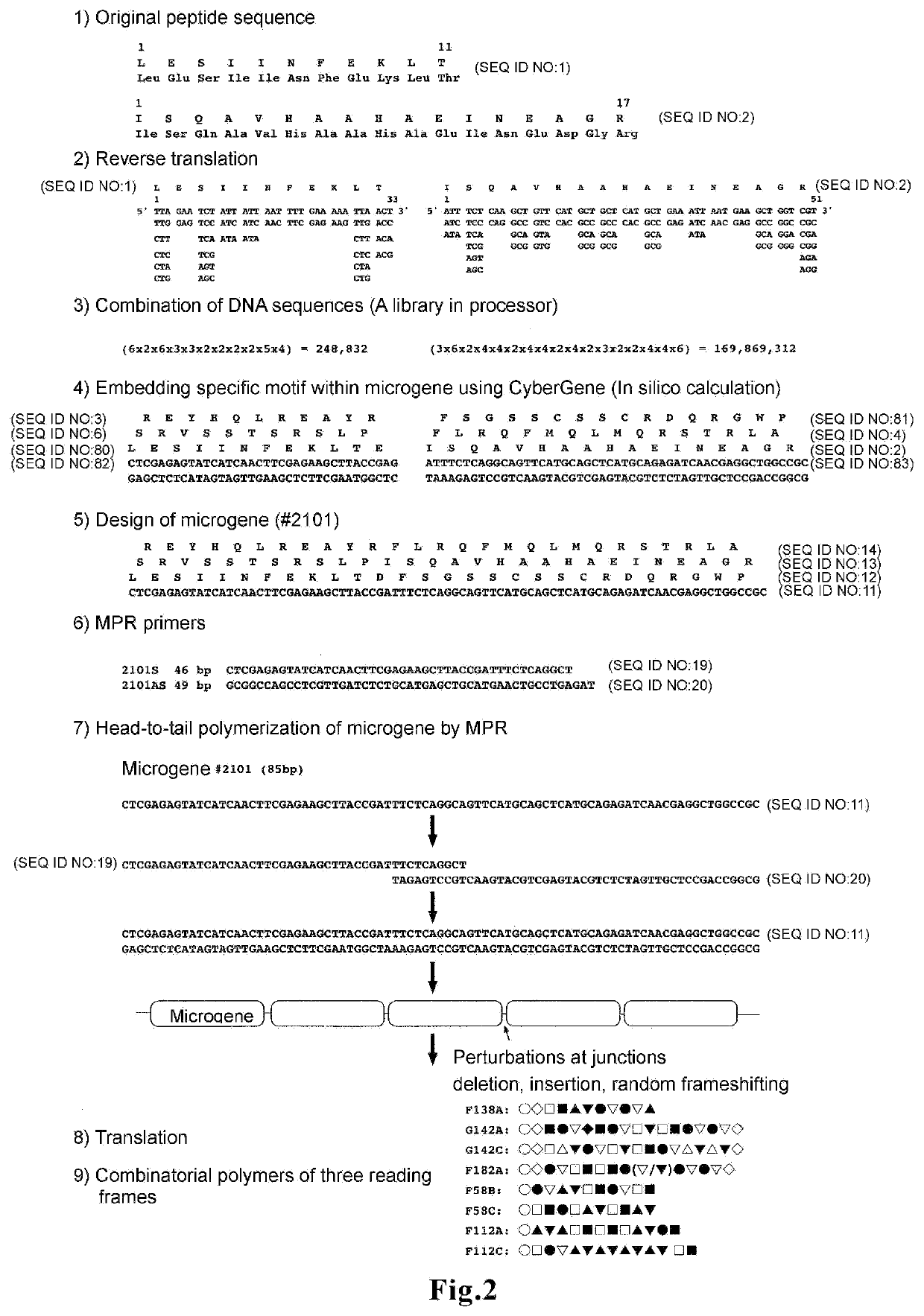
[0087]Microgenes #2101 and #6101, in which the MHC class I epitope OVA-I is encoded in the first sequence, and the MHC class II epitope OVA-II is encoded in the second sequence, were designed using CyberGene software, developed by Kiyotaka Shiba et al. (K. Shiba, Journal of Molecular Catalysis B: Enzymatic 28 (2004) 145-153) (FIG. 1b). The designing process is shown in 1) to 5) in FIG. 2. Since the two amino acids adjacent to the N-terminus of the MHC class I epitope (SIINFEKL) in native antigen OVA are known to influence degradation by aminopeptidase in the cell...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More