A macrocyclic oxidation substituted pentacyclic triterpanoids derivative and preparation method and use thereof
A technology of pentacyclic triterpenoids and compounds, which is applied in the field of organic chemistry and medicinal chemistry, and can solve problems such as research in the ascendant
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
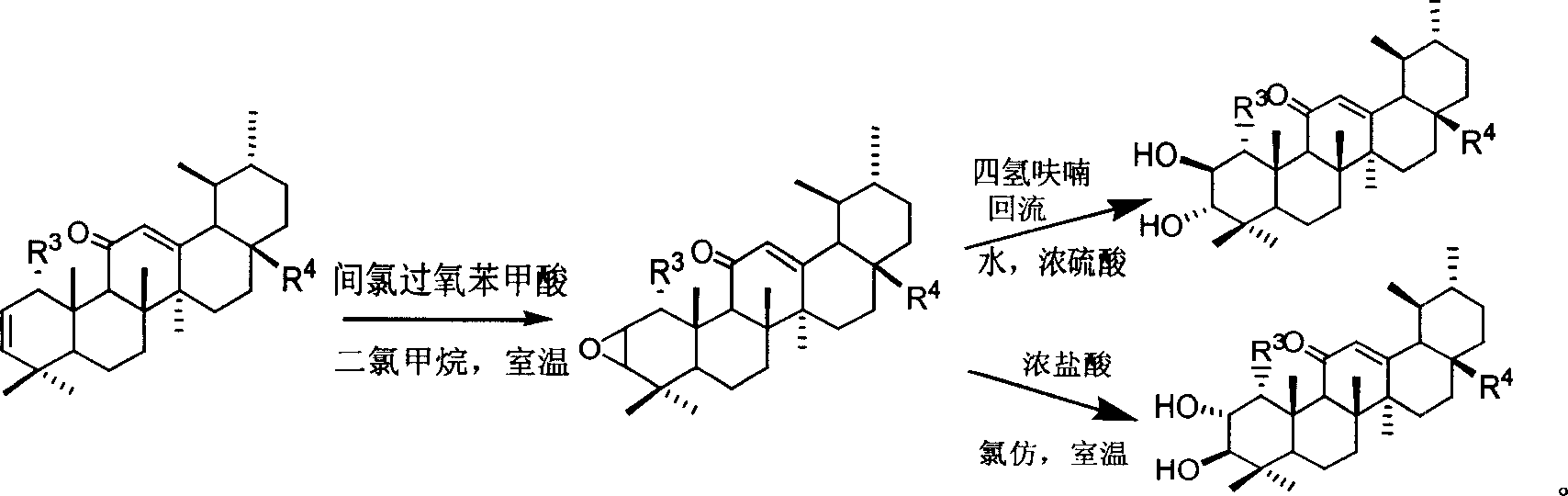
[0038] Embodiment 1: the preparation of compound Ig
[0039]Under ice-salt bath, ursolic acid (1.0 mmol) was dissolved in 10 ml of dichloromethane, 6 ml of triethylamine was added, and methanesulfonyl chloride (16.3 mmol) was added dropwise at 0°C. Stir at room temperature for 4 hours. After filtering, the filtrate was washed with 5 ml of 1 mol / L hydrochloric acid solution, washed with water until neutral, and dried over anhydrous magnesium sulfate. Precipitation under reduced pressure gave 3β-methylsulfonyloxy-arbutane-28-acid-12-ene. Dissolve in 10 ml of acetic acid (containing 0.5 ml of acetic anhydride), add chromium trioxide (4.0 mmol), and stir overnight at room temperature. 10 ml of water and 10 ml of dichloromethane were added to the reaction solution, and the layers were separated, and the aqueous layer was extracted with 10 ml of dichloromethane. The organic layers were combined, washed with 10 ml of saturated sodium bicarbonate solution, washed with water until n...
Embodiment 2~ Embodiment 9
[0042] According to the method of Example 1, the compounds of Example 2 to Example 9 shown below were prepared
Embodiment 2
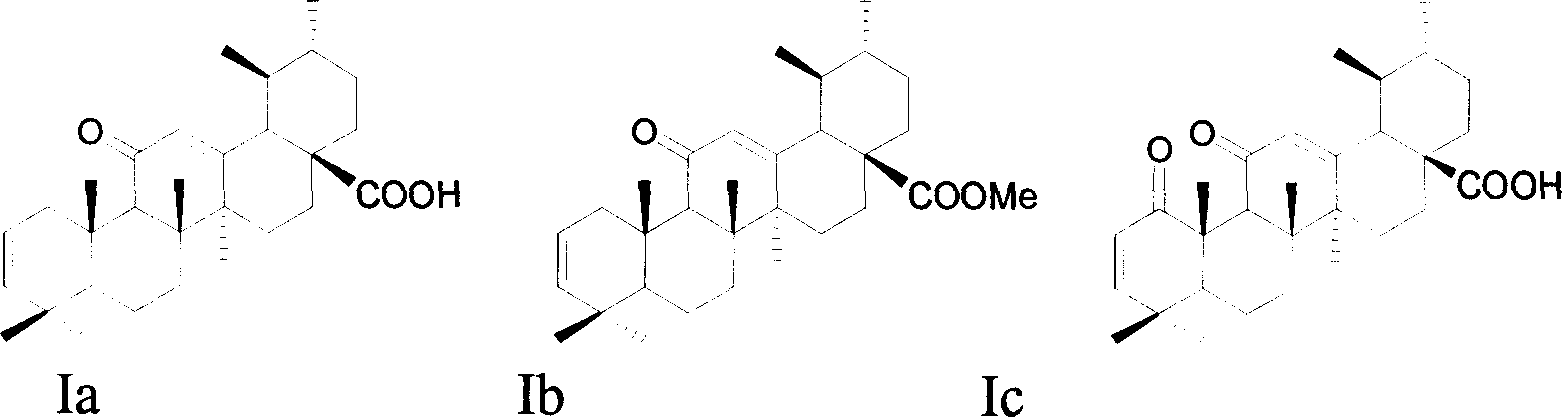
[0043] Example 2: Preparation of 11-carbonyl-arbutane-28-acid-2,12-diene (Ia), C 30 h 44 o 3 , MS: ESI m / e 452 (M + ); Rf[(petroleum ether / ethyl acetate: 3 / 1)+1%HCOOH]: 0.60; 1 HNMR (400MHz) δ: 0.87(d, 3H, J=6.0Hz), 0.97(d, 3H, J=6.4Hz), 5.36(dd, 1H, J=2.0, 10.0Hz, H-3), 5.43( dd, 1H, J=5.6, 10.0 Hz, H-2), 5.62 (s, 1H, H-12).
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More