Pharmaceutical composition with antimicrobial activity for parenteral administration and process for preparing same
A technology for parenteral administration and antibacterial drugs, applied in the field of antibacterial drug preparations and its manufacturing, to achieve the effect of no waste price
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
example 1
[0036] Example 1. Manufacture of solid compositions: β-lactam antibiotics – finely dispersed nanostructured silica.
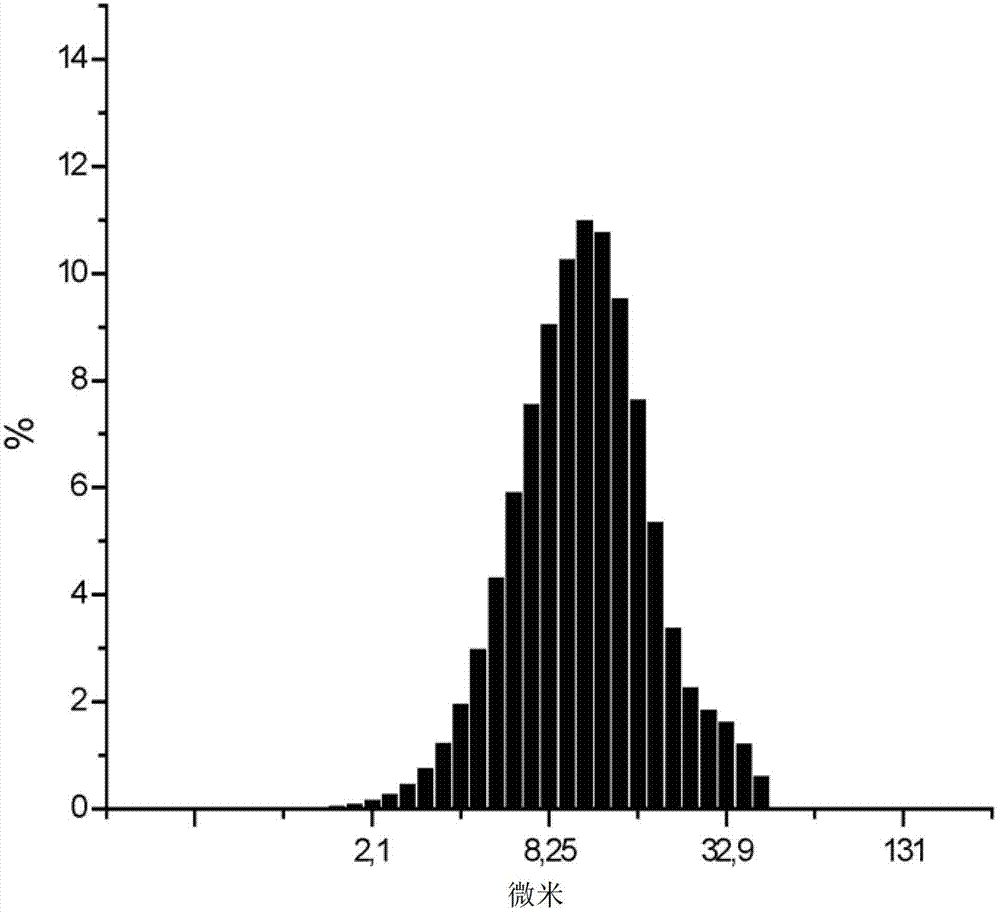
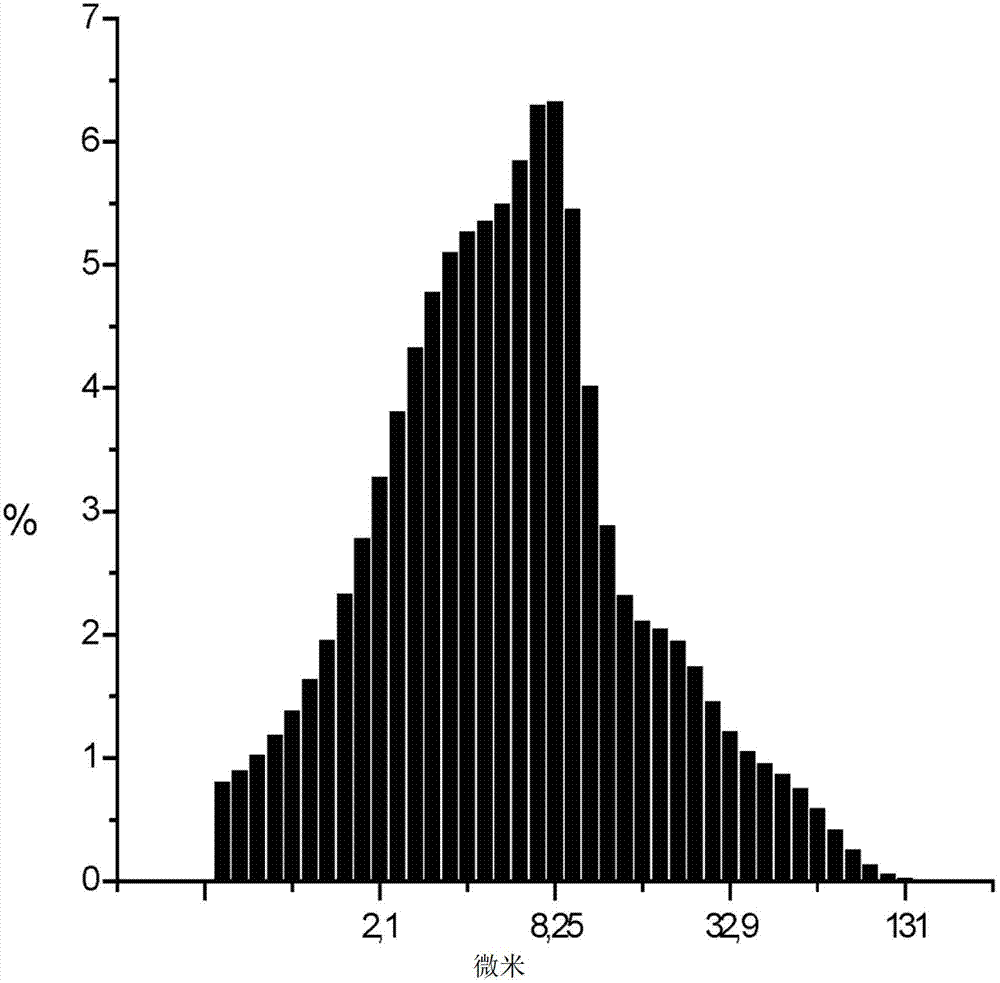
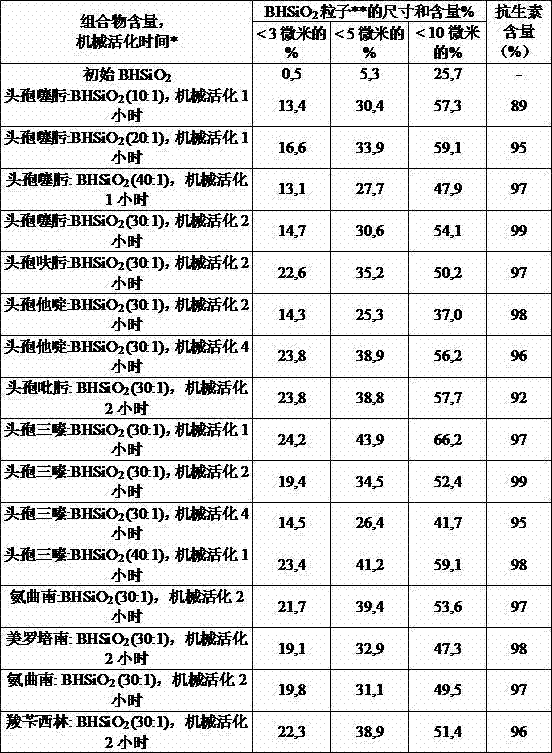
[0037] Processing of β-lactam antibiotics and ВНSiO in weight ratios of 10:1, 20:1, 30:1 and 40:1 in a circular rotary mill 2 mixture for 1, 2 and 4 hours. The data on the particle size composition of the aqueous suspension and the HPLC analysis of the antibiotic content (in % of starting material) are listed in Table 2.
[0038] Table 2
[0039] Particle size composition and antibiotic content of aqueous suspensions in different compositional variants
[0040]
[0041] * - Finely dispersed nanostructured silica** - mechanically activated
[0042] As can be seen from Table 2, the selected composition manufacturing conditions make the finely dispersed ВНSiO 2 The fraction of fractions (particle size less than 5 microns) is increased to a certain value (not less than 25%) and chemical degradation of antibiotics is avoided.
example 2
[0043] Example 2. Determination of therapeutic efficacy of antibacterial preparations and pharmaceutical compositions.
[0044] Therefore, beta-lactam antibiotics (cefazolin, cefuroxime, cefotaxime, ceftriaxone, cefoperazone, cefoperazone / sulbactam, ceftazidime, cefepime, cefoxitin, aztreonam) have been studied. , meropenem, carbenicillin) and its mechanization for 2 hours by weight ratio of 30:1 antibiotic / ВНSiO 2 Compositions of mixtures (cefazolin / ВНSiO 2 , Cefuroxime / ВНSiO 2 , Cefotaxime / ВНSiO 2 , Ceftriaxone / ВНSiO 2 , Cefoperazone / ВНSiO 2 , Cefoperazone / Sulbactam / ВНSiO 2 , Ceftazidime / ВНSiO 2 , Cefepime / ВНSiO 2 , Cefoxitin / ВНSiO 2 , Aztreonam / ВНSiO 2 , Meropenem / ВНSiO 2 , Carbenicillin / ВНSiO 2 ).
[0045] For the determination of β-lactams and their inclusion ВНSiO 2 The therapeutic efficacy of the pharmaceutical composition, according to [22, 23], using an experimental sepsis model and for the obtained data (χ 2 ) statistical processing metho...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More