New zwitter-ionic polymer containing multiple phosphonic acid end groups, preparation method and application thereof
A zwitterionic and polymer technology, applied in the field of zwitterionic polymers, can solve the problems of complex surface modification methods, difficult to find groups, etc., to achieve excellent anti-bacterial adhesion ability and surface lubrication ability, low concentration requirements, modified layer stabilization effect
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0035] Example 1: Functional Self-Assembly of Titanium Oxide Surface
[0036] (1) Preparation of polymer
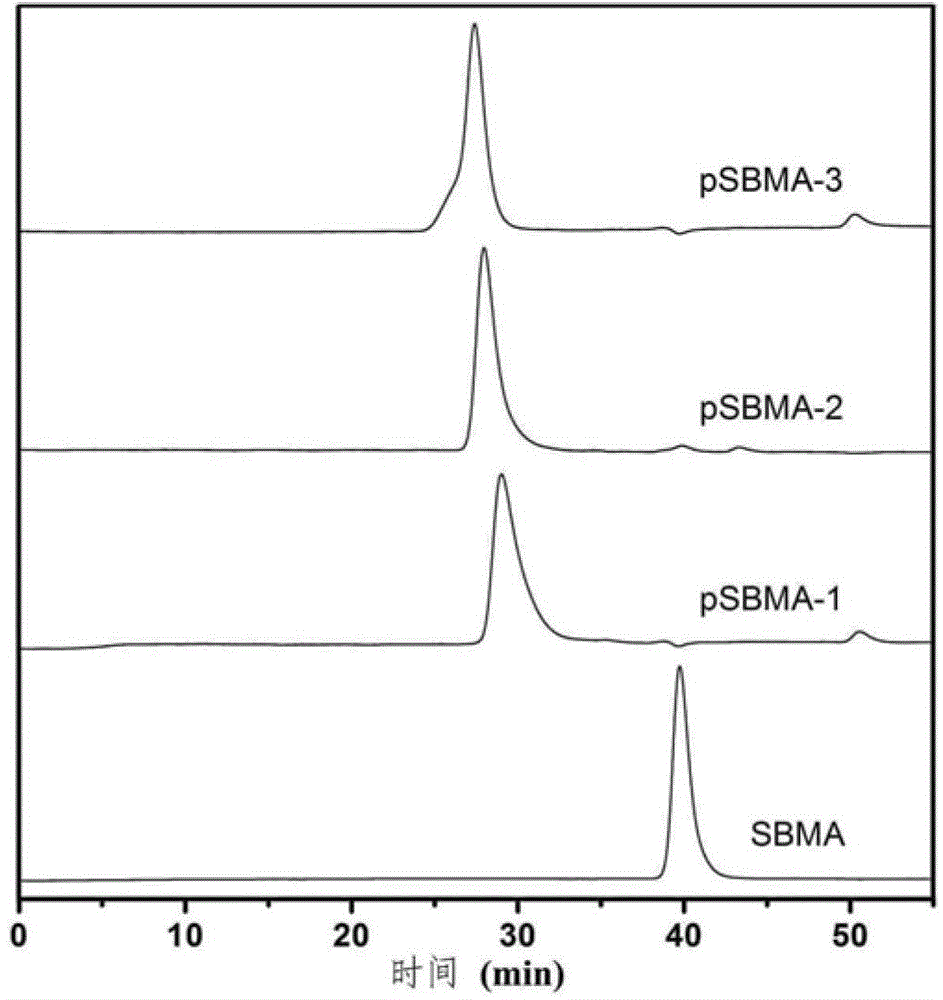
[0037] 1) Using the reversible addition‐fragmentation chain transfer (RAFT) polymerization method, take a 10 mL eggplant-shaped reaction bottle with a branch tube, add 4‐cyanovaleric acid dithiobenzoic acid, 4,4' ‐Azobis(4‐cyanovaleric acid), monomeric thiobetaine methyl acrylate (SBMA). Then add a 10:1 (volume ratio) mixture of methanol and deionized water, put it into a stirrer, dissolve the solid quickly, seal the reaction bottle, and pass nitrogen gas for 30 minutes under ice bath conditions. The oxygen-free reaction system was placed in an oil bath at 60°C to react to obtain a polymer (named pSBMA). During the whole operation, pay attention to protect from light with tin foil.
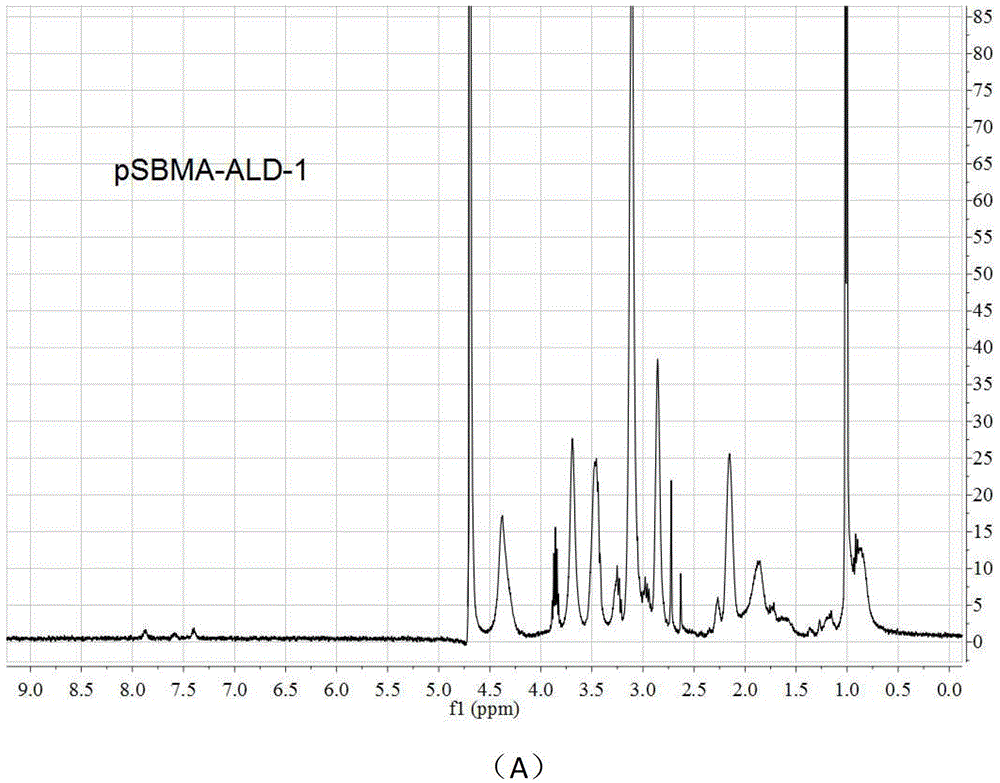
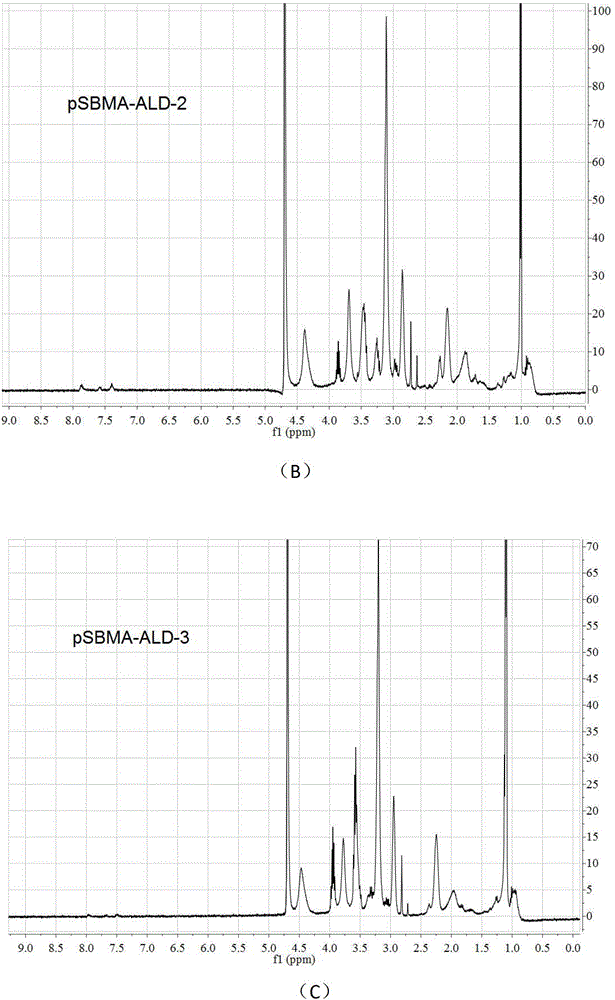
[0038] 2) Condensation acylation reaction of carboxylic acid and amine is adopted, and a 1 mL centrifuge tube is used as a reaction vessel. First, pSBMA was dissolved in 2‐(N‐morpholine)e...
Embodiment 2
[0052] Example 2: Functionalized self-assembly of stainless steel surfaces
[0053] (1) Preparation of polymer
[0054] 1) Using the reversible addition‐fragmentation chain transfer (RAFT) polymerization method, take a 10 mL eggplant-shaped reaction bottle with a branch tube, add 4‐cyanovaleric acid dithiobenzoic acid, 4,4' ‐Azobis(4‐cyanovaleric acid), monomeric thiobetaine methyl acrylate (SBMA). Then add a 10:1 (volume ratio) mixture of methanol and deionized water, put it into a stirrer, dissolve the solid quickly, seal the reaction bottle, and pass nitrogen gas for 30 minutes under ice bath conditions. The oxygen-free reaction system was placed in an oil bath at 60°C to react to obtain a polymer (named pSBMA). During the whole operation, pay attention to protect from light with tin foil.
[0055] 2) Condensation acylation reaction of carboxylic acid and amine is adopted, and a 1 mL centrifuge tube is used as a reaction vessel. First, pSBMA was dissolved in 2‐(N‐morpho...
Embodiment 3
[0070] Example 3: Fe 3 o 4 Surface Modification of Magnetic Nanoparticles
[0071] (1) Preparation of polymer
[0072] 1) Using the reversible addition‐fragmentation chain transfer (RAFT) polymerization method, take a 10 mL eggplant-shaped reaction bottle with a branch tube, add 4‐cyanovaleric acid dithiobenzoic acid, 4,4' ‐Azobis(4‐cyanovaleric acid), monomeric thiobetaine methyl acrylate (SBMA). Then add a 10:1 (volume ratio) mixture of methanol and deionized water, put it into a stirrer, dissolve the solid quickly, seal the reaction bottle, and pass nitrogen gas for 30 minutes under ice bath conditions. The oxygen-free reaction system was placed in an oil bath at 60°C to react to obtain a polymer (named pSBMA). During the whole operation, pay attention to protect from light with tin foil.
[0073] 2) Condensation acylation reaction of carboxylic acid and amine is adopted, and a 1 mL centrifuge tube is used as a reaction vessel. First, pSBMA was dissolved in 2‐(N‐morph...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More