Method for measuring phenylhydrazine compound residues in crude drugs through HPLC (high performance liquid chromatography)
A technology for compounds and APIs, applied in the field of pharmaceutical analysis
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0041] Instrument: Agilent1260 high performance liquid chromatography, 1260 ultraviolet detector
[0042] Chromatographic column: chromatographic column with phenyl bonded silica gel as filler (250×4.6mm, 5μm);
[0043] Mobile phase A: 0.01mol / L ammonium acetate buffer (pH5.1)
[0044] Mobile Phase B: Acetonitrile
[0045] See the table below for gradient elution;
[0046] time (min)
Mobile Phase A(%)
Mobile phase B(%)
0
90
10
3
90
10
14
80
20
15
10
90
25
10
90
30
90
10
35
90
10
[0047] Flow rate: 1.0mL / min
[0048] Detection wavelength: 260nm
[0049] Column temperature: 30°C
[0050] Injection volume: 10μL
[0052] experiment procedure:
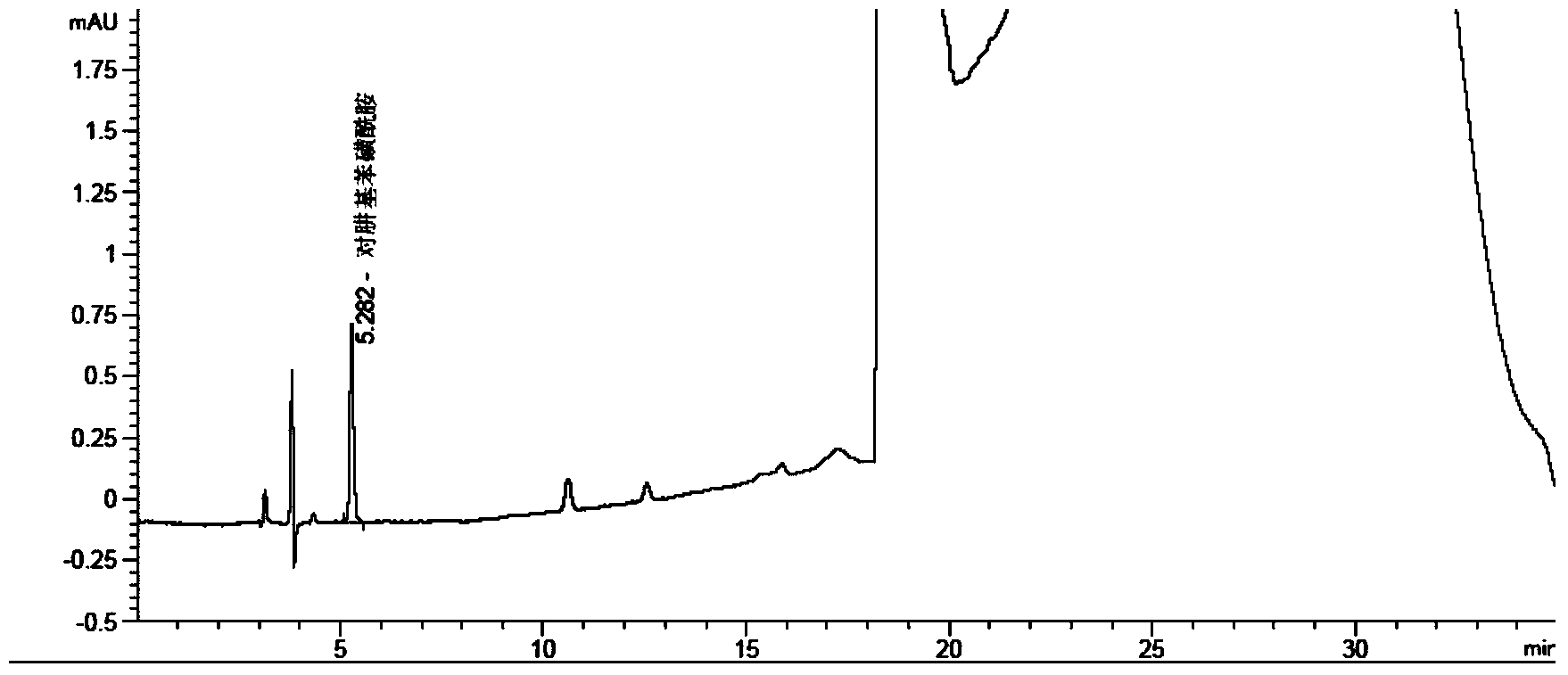
[0053] Need test solution: Weigh 1.0014g of celecoxib sample, put it in a 10mL measuring bottle, dissolve it with diluent and dilute to the mark, shake well.
[0054] Reference s...
Embodiment 2
[0058] Instrument: Agilent1260 high performance liquid chromatography, 1260 ultraviolet detector
[0059] Chromatographic column: chromatographic column with phenyl bonded silica gel as filler (250×4.6mm, 5μm);
[0060] Mobile phase A: 0.02mol / L ammonium acetate buffer (pH4.9)
[0061] Mobile Phase B: Acetonitrile
[0062] See the table below for gradient elution;
[0063] time (min)
Mobile Phase A(%)
Mobile phase B(%)
0
90
10
3
90
10
14
80
20
15
10
90
25
10
90
30
90
10
35
90
10
[0064] Flow rate: 1.0mL / min
[0065] Detection wavelength: 260nm
[0066] Column temperature: 25°C
[0067] Injection volume: 10μL
[0069] experiment procedure:
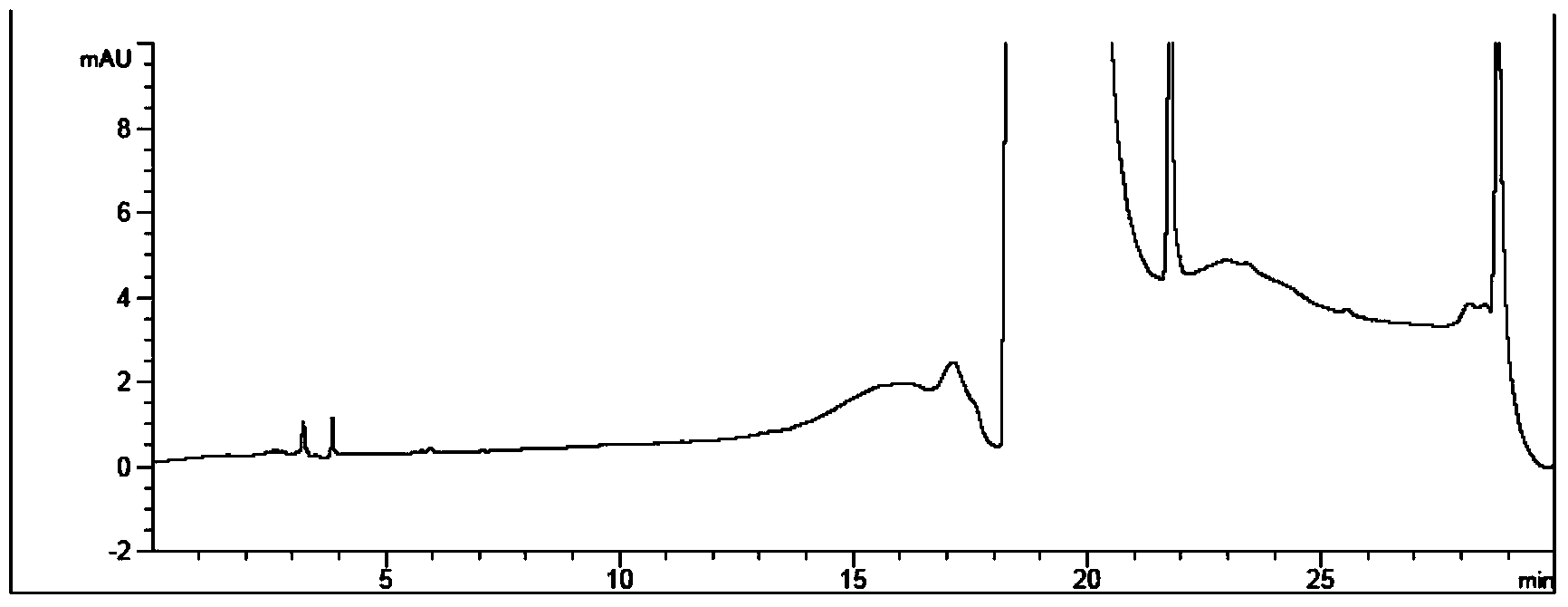
[0070] Reference substance solution: Accurately weigh 37.48mg of p-hydrazinobenzenesulfonamide hydrochloride reference substance, put it in a 100mL measuring bottle, dissolve it...
Embodiment 3
[0075] Instrument: Agilent1260 high performance liquid chromatography, 1260 ultraviolet detector
[0076] Chromatographic column: chromatographic column with phenyl bonded silica gel as filler (250×4.6mm, 5μm);
[0077] Mobile phase A: 0.01mol / L ammonium acetate buffer (pH3.0)
[0078] Mobile Phase B: Acetonitrile
[0079] See the table below for gradient elution;
[0080] time (min)
Mobile Phase A(%)
Mobile phase B(%)
0
90
10
3
90
10
14
80
20
15
10
90
25
10
90
30
90
10
35
90
10
[0081] Flow rate: 1.0mL / min
[0082] Detection wavelength: 260nm
[0083] Column temperature: 25°C
[0084] Injection volume: 20μL
[0085] Diluent: Acetonitrile
[0086] experiment procedure:
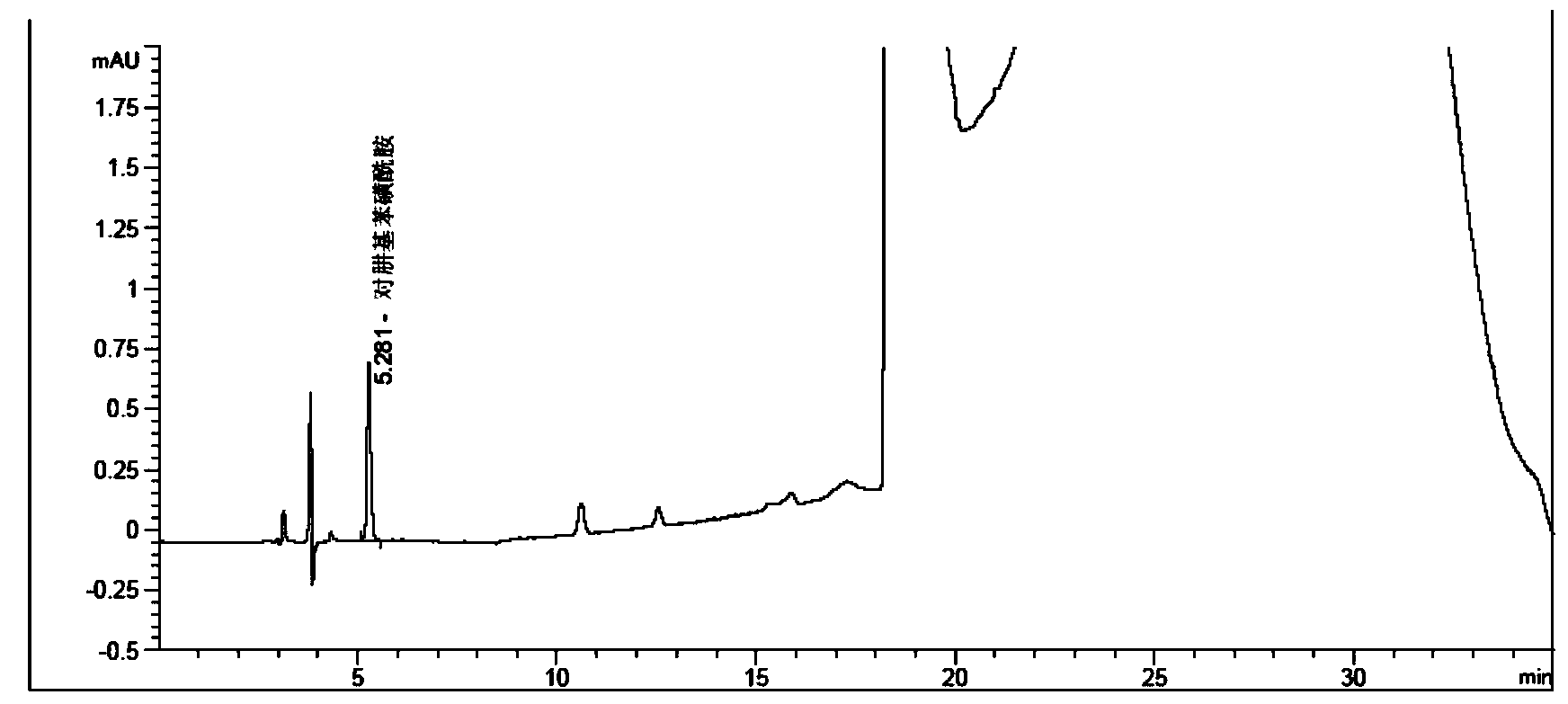
[0087] The test solution: Weigh 0.50261 g of the crude product of deferasirox, put it in a 10 mL measuring bottle, dissolve it with a diluent and dilute to the mark, and sha...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Sensitivity | aaaaa | aaaaa |
| mobile phase | aaaaa | aaaaa |
| wavelength | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More