Method for fast and efficient separation and purification of tumor cells from malignant pleural effusion and ascites
A technology for the separation and purification of tumor cells, which is applied in the field of fast and efficient separation and purification of tumor cells from malignant pleural effusion, which can solve the problems of separation and purification steps, the inability to conduct further research, and the small amount of cell collection, so as to achieve small cell damage , Tumor cells are in good condition, and the effect is reproducible
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0031] 1. Main experimental materials
[0032] (1) Source of pleural effusion: pleural effusion in patients with lung adenocarcinoma.
[0033] (2) PBS solution: Potassium dihydrogen phosphate (KH 2 PO 4 ): 0.27g, disodium hydrogen phosphate (Na 2 HPO 4 ): 1.42g, Sodium chloride (NaCl): 8g, Potassium chloride (KCl) 0.2g, add about 800mL of deionized water and stir to dissolve, then add concentrated hydrochloric acid to adjust the pH to 7.2-7.4, and finally dilute to 1L. It was prepared 24 hours before the experiment, sterilized under high temperature and high pressure, and then sealed and stored at room temperature.
[0034] (3) Calf serum: Hangzhou Sijiqing Company.
[0035] (4) 50ml centrifuge tube: American BD Company.
[0036] (5) Sterile glass Pasteur pipette: American Fasher Company
[0037] (6) Centrifuge: Eppendrof, Germany
[0038] 2. Operation steps
[0039] (1) Aseptically extract 500ml of pleural effusion from lung cancer patients, take 200ml from it, p...
Embodiment 2
[0043] The difference with embodiment 1 is:
[0044] 2. Operation steps
[0045] (1) Aseptically extract 500ml of pleural effusion from lung cancer patients, take 200ml from it, put it into four 50ml centrifuge tubes, and centrifuge at 1500r / min for 5 minutes at room temperature to obtain cell pellets.
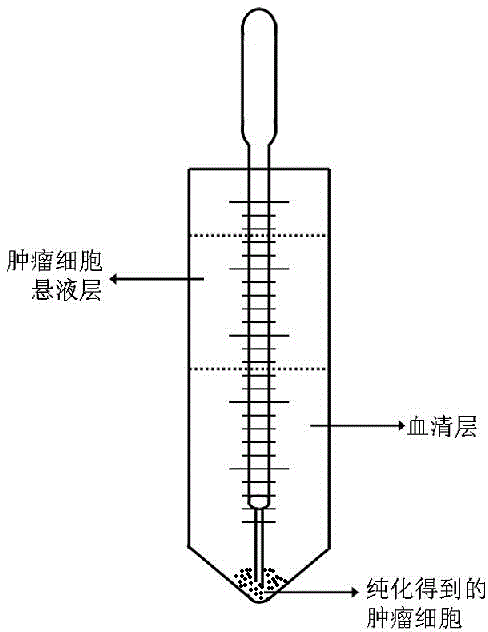
[0046](2) Collect and mix the cell pellet with 20ml of PBS liquid to obtain a suspension of tumor cells. Take a sterile 50ml centrifuge tube and add 30ml of calf serum. The specific viscosity of the serum (relative to distilled water) is 1.8. Gently add the mixed tumor cell suspension to the serum along the tube wall with a Pasteur pipette to make it into two layers, the tumor cell suspension layer is on top, and the serum layer is on the bottom, with a height of not less than 4cm.
[0047] (3) Let the centrifuge tube added to the tumor cell suspension layer stand still for 5 minutes, and use a Pasteur pipette to absorb 5ml of serum from the bottom of the centrifuge tube....
Embodiment 3
[0049] The difference with embodiment 1 is:
[0050] 2. Operation steps
[0051] (1) Aseptically extract 500ml of pleural effusion from lung cancer patients, take 200ml from it, put it into four 50ml centrifuge tubes, and centrifuge at 1500r / min for 5 minutes at room temperature to obtain cell pellets.
[0052] (2) Collect and mix the cell pellet with 20ml of PBS liquid to obtain a suspension of tumor cells. Take a sterile 50ml centrifuge tube and add 20ml of calf serum. The specific viscosity of the serum (relative to distilled water) is 1.6. Gently add the mixed tumor cell suspension to the serum along the tube wall with a Pasteur pipette to make it into two layers, the tumor cell suspension layer is on top, and the serum layer is on the bottom, with a height of not less than 4cm.
[0053] (3) Let the centrifuge tube added to the tumor cell suspension layer stand still for 6 minutes, and use a Pasteur pipette to absorb 5ml of serum from the bottom of the centrifuge tube...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More