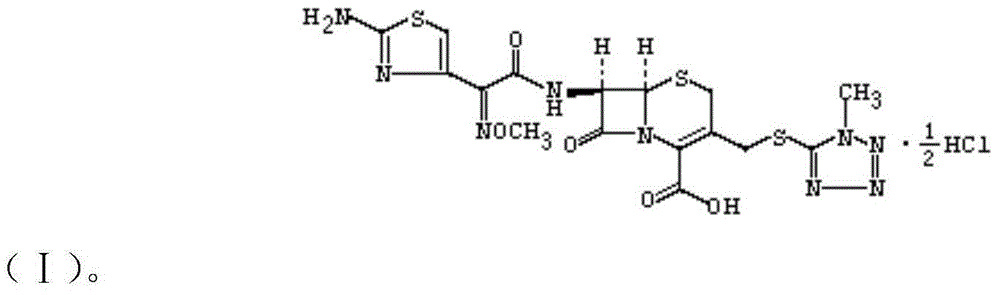
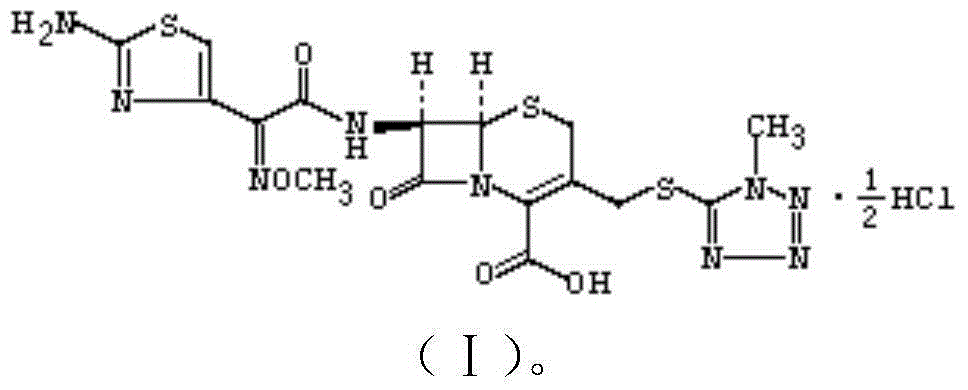
Cefmenoxime hydrochloride compound entity used for children and preparation thereof
A technology of cefmenoxime hydrochloride and compound, which is applied in the field of cefmenoxime hydrochloride compound entity and its preparation for children, can solve the problems such as difficulty in dissolving cefmenoxime hydrochloride, increase in impurities, poor stability, etc. good stability
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0022] (1) Weigh 100g of cefmenoxime crude product, add 1000ml of water, stir and slowly add 4% sodium bicarbonate solution dropwise until clarification, add 10ml of ethyl acetate under stirring, transfer to a 1000ml pressure-resistant container, ensure that it is full and air bubbles are removed, and seal Container, shake, temperature control -18 ℃ and freeze for 3 hours, then take out;
[0023] (2) Remove the organic phase, and after the ice melts, add 10 g of activated carbon, stir and decolorize, and filter;
[0024] (3) Transfer the filtrate to a crystallization tank, control the temperature at 10-15°C, adjust the pH to 1.5 with dilute hydrochloric acid, continue to grow crystals for 1 hour, filter with suction, wash with water, and dry in vacuum at 40°C to obtain the final product. The maximum impurity is 0.13%, and the total impurity is 0.23%.
Embodiment 2
[0026] (1) Weigh 100 g of cefmenoxime crude product, add 1000 ml of water, stir and slowly add 4% sodium bicarbonate solution dropwise until clarification, add 10 ml of chloroform under stirring, transfer to a 1000 ml pressure-resistant container, ensure that it is full and air bubbles are removed, seal the container, Shake, and freeze at -18°C for 3 hours before taking out;
[0027] (2) Remove the organic phase, and after the ice melts, add 10 g of activated carbon, stir and decolorize, and filter;
[0028] (3) Transfer the filtrate to a crystallization tank, control the temperature at 10-15°C, adjust the pH to 1.7 with dilute hydrochloric acid, continue growing crystals for 1 hour, filter with suction, wash with water, and dry in vacuo at 40°C. The maximum simple impurity is 0.19%, and the total impurity is 0.29%.
Embodiment 3
[0030] (1) Weigh 100g of cefmenoxime crude product, add 1000ml of water, stir and slowly add 4% sodium bicarbonate solution dropwise until clarification, add 7ml of ethyl acetate under stirring, transfer to a 1000ml pressure-resistant container, ensure that it is full and air bubbles are removed, and seal Container, shake, temperature control -18 ℃ and freeze for 3 hours, then take out;
[0031] (2) Remove the organic phase, and after the ice melts, add 10 g of activated carbon, stir and decolorize, and filter;
[0032] (3) Transfer the filtrate to a crystallization tank, control the temperature at 10-15°C, adjust the pH to 1.5 with dilute hydrochloric acid, continue to grow crystals for 1 hour, filter with suction, wash with water, and dry in vacuum at 40°C. The maximum simple impurity is 0.17%, and the total impurity is 0.24%.
PUM
| Property | Measurement | Unit |
|---|---|---|
| concentration | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More