Medical grinding wheel for grinding insulin needles and manufacturing method thereof
A technology for insulin and grinding wheels, which is applied in the manufacture of tools, abrasives, grinding devices, etc. It can solve the problems of small insulin puncture force, large needle tip puncture force, and high grinding efficiency, so as to reduce puncture force, reduce grinding scratches, The effect of improving grinding efficiency
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
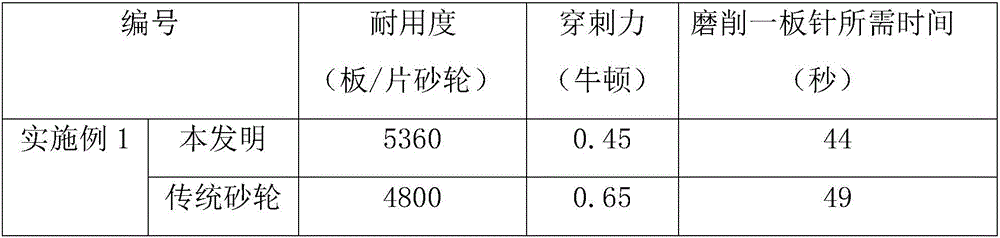
[0045] A grinding wheel for grinding insulin needles for medical use, its raw material components and weight percentages are: 65% of 2500 mesh silicon carbide, 11% of 1200 mesh ceramic corundum, 12% of modified resin, 7% of glass microspheres, 2% of cryolite powder, oxidized Zinc 3%. The raw material components and weight percentages of the modified resin are: 91% of phenolic resin and 9% of nitrile rubber. The particle size of the cryolite powder is 280 mesh, and the diameter of the glass microsphere is 0.5 mm.
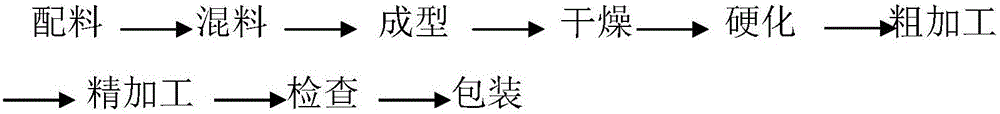
[0046] The manufacturing process steps of the present embodiment are as follows:
[0047] 1) Mixing: pass the weighed ceramic corundum abrasive and silicon carbide abrasive through a 150# sieve, pour into the mixing pot and mix for 10 minutes, add modified resin and mix for 30 minutes, add fillers such as glass beads and mix for 10 minutes , through a 46# sieve;
[0048] 2) Turn on the preheating device of the molding equipment, set the pressure head temperature t...
Embodiment 2
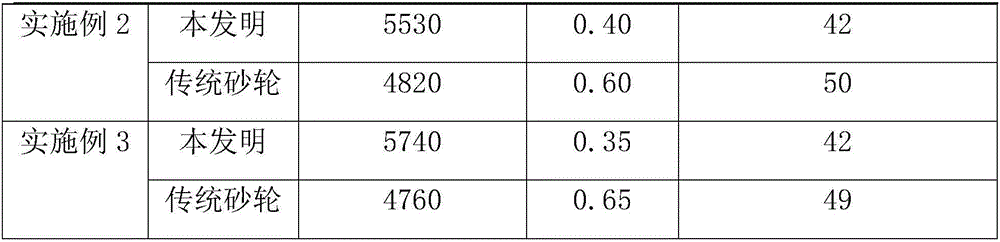
[0060] A needle-tip grinding wheel for grinding insulin for medical use, its raw material components and weight percentages are: 2500 mesh silicon carbide 64%, 1200 mesh ceramic corundum 12%, modified resin 12%, glass microspheres 7%, cryolite powder 2%, zinc oxide 3%. The raw material components and weight percentages of the modified resin are: 91% of phenolic resin and 9% of nitrile rubber. The particle size of the cryolite powder is 280 mesh, and the diameter of the glass microsphere is 0.5 mm.
[0061] The manufacturing process steps of the present embodiment are as follows:
[0062] 1) Mixing: pass the weighed ceramic corundum abrasive and silicon carbide abrasive through a 150# sieve, pour into the mixing pot and mix for 10 minutes, add modified resin and mix for 30 minutes, add fillers such as glass beads and mix for 10 minutes , through a 46# sieve;
[0063] 2) Turn on the preheating device of the molding equipment, set the pressure head temperature to 100-120°C, an...
Embodiment 3
[0075] A needle-tip grinding wheel for grinding insulin for medical use, its raw material components and weight percentages are: 2500 mesh silicon carbide 60%, 1200 mesh ceramic corundum 14%, modified resin 12%, glass microspheres 9%, cryolite powder 2%, zinc oxide 3%. The raw material components and weight percentages of the modified resin are: 91% phenolic resin, 9% NBR, the particle size of the cryolite powder is 280 mesh, and the diameter of the glass microsphere is 0.5 mm.
[0076] The manufacturing process steps of embodiment 3 are as follows:
[0077] 1) Mixing: pass the weighed ceramic corundum abrasive and silicon carbide abrasive through a 150# sieve, pour into the mixing pot and mix for 10 minutes, add modified resin and mix for 30 minutes, add fillers such as glass beads and mix for 10 minutes , through a 46# sieve;
[0078] 2) Turn on the preheating device of the molding equipment, set the pressure head temperature to 100-120°C, and set the turntable temperature...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Granularity | aaaaa | aaaaa |
| Diameter | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More