Astragaloside peritoneal dialysis solution
An astragaloside IV and glycoside peritoneal technology, which is applied in the field of medicine, can solve the problems of many adverse reactions, many impurities, complex components, etc., and achieve the effects of wide clinical application value, improving cell metabolism and reducing renal fibrosis.
Inactive Publication Date: 2017-01-04
SHANGHAI TREEFUL PHARMA
View PDF0 Cites 7 Cited by
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
As the total extract of Astragalus can not effectively control the impurity content, its quality changes with the content of total Astragalus saponins and Astragaloside IV in the original medicinal materials, and most of them have many impurities, which may easily cause indirect inaccurate clinical dosage and unstable curative effect , serious adverse reactions
[0004] For traditional Chinese medicine preparations, the total extracts of traditional Chinese medicine have complex components, and the product quality is largely affected by the extraction process conditions and the origin of traditional Chinese medicine, which makes the dosage of active ingredients in pharmaceutical preparations unstable, and there are many impurities, resulting in unstable drug efficacy. Many adverse reactions
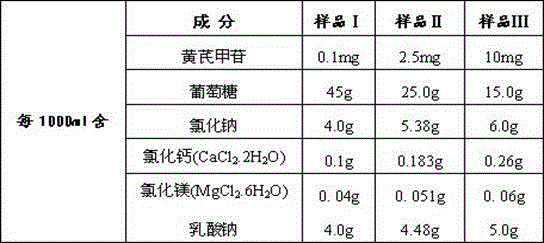
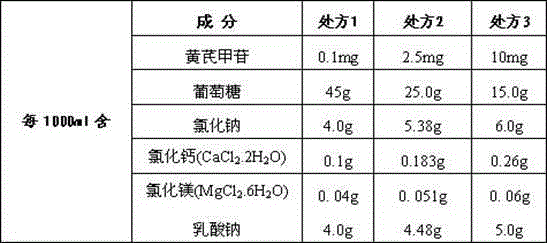
[0005] The key to the curative effect of the drug lies in the amount of effective monomers contained in traditional Chinese medicine. This application uses the effective monomer extracted from Astragalus - astragaloside IV as the main active ingredient, with glucose, sodium chloride, calcium chloride, magnesium chloride, and sodium lactate as auxiliary components. Components, dissolved in an appropriate amount of water for injection, to make astragaloside peritoneal dialysis solution, which has unique advantages in the treatment of renal failure diseases, so far there is no similar report on the study of astragaloside peritoneal dialysis solution
Method used
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
View moreImage
Smart Image Click on the blue labels to locate them in the text.
Smart ImageViewing Examples
Examples
Experimental program
Comparison scheme
Effect test
Embodiment 2
[0041] Embodiment two: Observing the effects of astragaloside IV peritoneal dialysis solution and conventional peritoneal dialysis solution on the efficacy of peritoneal dialysis in rats through animal experiments
[0042] sample: The astragaloside IV peritoneal dialysate sample prepared by the method of embodiment one Ⅰ ,sample Ⅱ ,sample Ⅲ :
[0043] Control: Peritoneal Dialysis Solution (Lactate-G1.5%) Shanghai Changzheng Fumin Jinshan Pharmaceutical Co., Ltd.
[0044] ReagentUrea Oxygen (BUN), Creatinine (Cr) and Protein Determination Kit Beckman Company, USA.
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More PUM
 Login to View More
Login to View More Abstract
The invention relates to an astragaloside peritoneal dialysis solution preparation prescription and a preparation method thereof, and is characterized in that an effective monomer-astragaloside extracted from astragalus membranaceus and used as an essential component, and glucose, sodium chloride, calcium chloride, magnesium chloride and sodium lactate as auxiliary components are dissolved in an appropriate amount of water for injection, and an astragaloside peritoneal dialysis solution is prepared and is dedicated to treatment of renal failure.
Description
technical field [0001] The invention belongs to the technical field of medicine and relates to an astragaloside IV peritoneal dialysis solution. Background technique [0002] Astragalus is the dried root of the leguminous plant Astragalus mongolica or Astragalus membranaceus. Produced in Inner Mongolia, Shanxi, Gansu, Heilongjiang and other places, it is a national third-level protected plant. It has the effects of invigorating qi and solidifying the surface, detoxifying and discharging pus, diuresis, reducing swelling and promoting muscle growth. The main chemical components contained are: astragaloside IV, saponins, polysaccharides, quercetin, flavonoids, choline, betaine, amino acids and trace amounts of folic acid. Modern pharmacological research has found that astragalus has positive effects on the cardiovascular system, blood and hematopoietic system, liver and kidney organs, and can enhance immunity, anti-cancer, anti-virus, and anti-aging. In particular, there are...
Claims
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More Application Information
Patent Timeline
 Login to View More
Login to View More Patent Type & Authority Applications(China)
IPC IPC(8): A61K33/14A61K31/7048A61P7/08A61P13/12
Inventor 不公告发明人
Owner SHANGHAI TREEFUL PHARMA