Production method capable of improving purity of metformin hydrochloride
一种盐酸二甲双胍、生产方法的技术,应用在医药化学品合成领域,能够解决设备材质要求苛刻、生产操作不易控制、原料所占比例大等问题,达到干燥速度快、缩短产品精制周期、原料转化率提高的效果
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
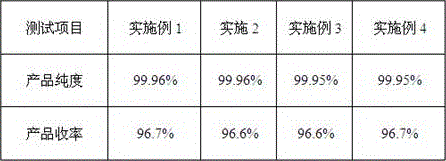
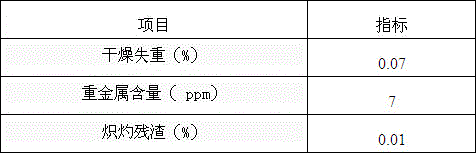
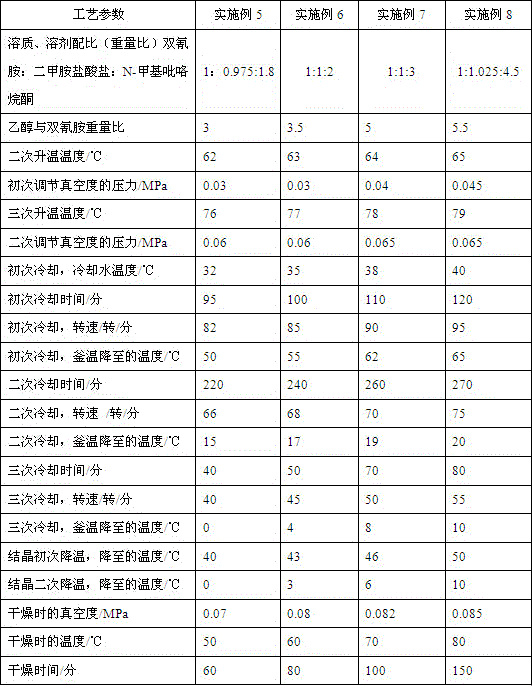
Embodiment 1
[0049] 1. Add solvent and solute to the synthesis kettle
[0050] The synthesis kettle used is a 1000L glass-lined kettle. The solvent N-methylpyrrolidone 600Kg is sucked into the synthesis kettle by vacuum, the stirring is started, and the solute dicyandiamide 250Kg and dimethylamine hydrochloride 250Kg are added.
[0051] 2. Stir
[0052] (1) Stirring for the first time
[0053] Open the steam valve, turn on the steam to raise the temperature, and use a frequency converter to adjust the stirring speed to 50-60 rpm.
[0054] (2) Second heating
[0055] Stir for 15-20 minutes to raise the temperature of the synthesis kettle to 62°C.
[0056] (3) Adjust the vacuum degree for the first time
[0057] Turn on the vacuum pump, adjust the vacuum degree in the synthesis kettle to 0.04MPa, and continue stirring for 10-20 minutes after the vacuum degree is stable.
[0058] (4) Three heating times
[0059] Open the large steam valve, continue to heat up, and stir for 15-20 minute...
Embodiment 2
[0095] The preparation steps of Example 1 are adopted, and the solvent used is the mother liquor produced in the spin-drying step of Example 1, which is put into a centrifuge from the synthesis kettle and then reused to prepare metformin hydrochloride.
Embodiment 3
[0097] Using the preparation steps of Example 1, the solvent used is the mother liquor produced in the spin-drying step of Example 2, which is put into a centrifuge from the synthesis kettle and continued to be reused to prepare metformin hydrochloride.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More