Spirobifluorene derivative and application of spirobifluorene derivative in field of organic electroluminescence
A spirobifluorene and compound technology, which is applied to spirobifluorene derivatives and their application in the field of organic electroluminescence, can solve the problems of limited application, low molecular triplet energy level, etc. The effect of long-life device performance and high stability
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
preparation example Construction
[0073]
[0074] Compounds according to the present invention can be prepared by synthetic procedures known to those of ordinary skill in the art, such as bromination, Suzuki coupling, Hartwig-Buchwald coupling, and the like.
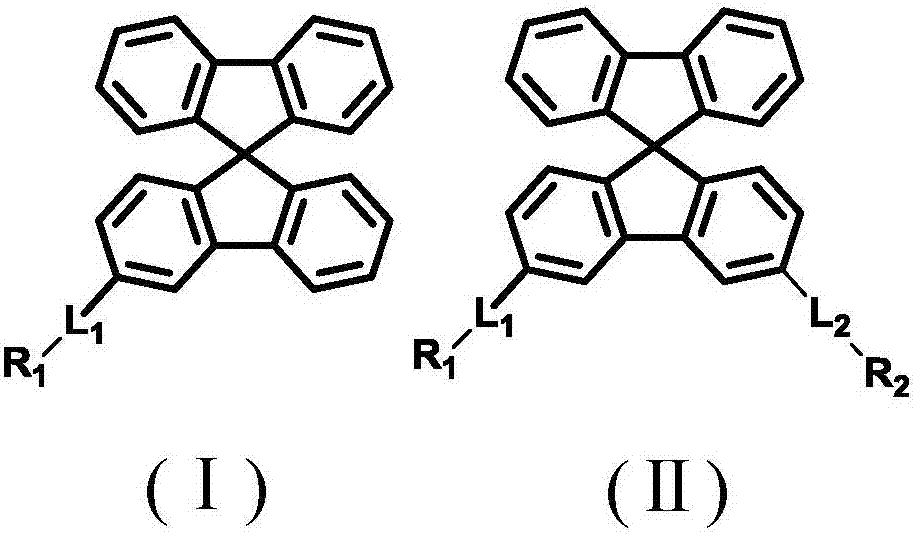
[0075] The synthesis of the compounds of the present invention usually starts with 3- and / or 6-functionalized spirobifluorene derivatives, followed by the introduction of the group -L by metal-catalyzed coupling reactions such as Suzuki coupling 1 -R 1 and / or -L 2 -R 2 .
[0076] In a preferred embodiment of the present invention, the spirobifluorene derivative is a compound functionalized with a boronic acid derivative, and the group -L 1 -R 1 and / or -L 2 -R 2 Derived from halogen-functionalized compounds. In another preferred embodiment of the present invention, the spirobifluorene derivative is a halogen-functional compound, and the group -L 1 -R 1 and / or -L 2 -R 2 Compounds derived from functionalization of boronic acid derivatives.
[...
preparation example
[0092] Synthesis of Intermediate 1:
[0093] The structural formula and synthetic route of intermediate 1 are shown in the following formula:
[0094]
[0095]The preparation method of the compound of formula M1 is: in the 250 mL there-necked flask equipped with thermometer, dropping funnel and mechanical stirring, add 10.0g (36.21mmol) 2-amino-4'-bromo-benzophenone and 80mL water, Slowly add 5.0g, 98% (50mmol) concentrated sulfuric acid dropwise under stirring, stir for 30 minutes, cool to 0°C, add dropwise a solution of 1.5g (21mmol) sodium nitrite dissolved in 5mL water, after the dropwise addition, keep the After 30 minutes, the temperature was slowly raised to 60°C. After the bubbles were completely released, it was cooled to room temperature and filtered to obtain a light yellow crystalline solid. The solid was separated by column chromatography (350 mesh silica gel, the eluent was petroleum ether:dichloromethane=20:1 (V / V)), the solvent was evaporated, and after dry...
preparation example 1
[0098] Preparation Example 1: Synthesis of Compound 1-75
[0099] The structural formula and synthetic route of compound 1-75 are shown in the following formula:
[0100]
[0101] In a 200mL Schlenk bottle, add 1.50g (4.16mmol) of 3-boronic acid-9,9' spirobifluorene, 0.47g (2.08mmol) of 2,4-dichloro-6-phenyl-1,3,5-triazine ), tetrakis(triphenylphosphine)palladium 0.26g (0.23 mmol), anhydrous potassium carbonate 1.77g (12.8mmol), tetrahydrofuran 50mL, water 8mL, under the protection of argon gas, stirred and reacted at 70°C for 30 hours, and the reaction was completed. Evaporate the solvent, dissolve the residue with 50mL dichloromethane and 50mL water, wash with water, separate the organic layer, extract the water layer twice with 15mL dichloromethane, combine the organic layers, wash twice with 50mL water until neutral, evaporate the solvent Finally, the residue was separated by column chromatography (silica gel was 350 mesh, and the eluent was petroleum ether: chloroform...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com