Method for preparing genetically-modified t cells which express chimeric antigen receptor
A chimeric antigen receptor, gene modification technology, applied in receptor/cell surface antigen/cell surface determinant, fusion cells, cells modified by introducing foreign genetic material, etc., can solve the problem of low gene transfer efficiency and cell damage , cell viability, decreased cell proliferation rate, etc.
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
preparation example Construction
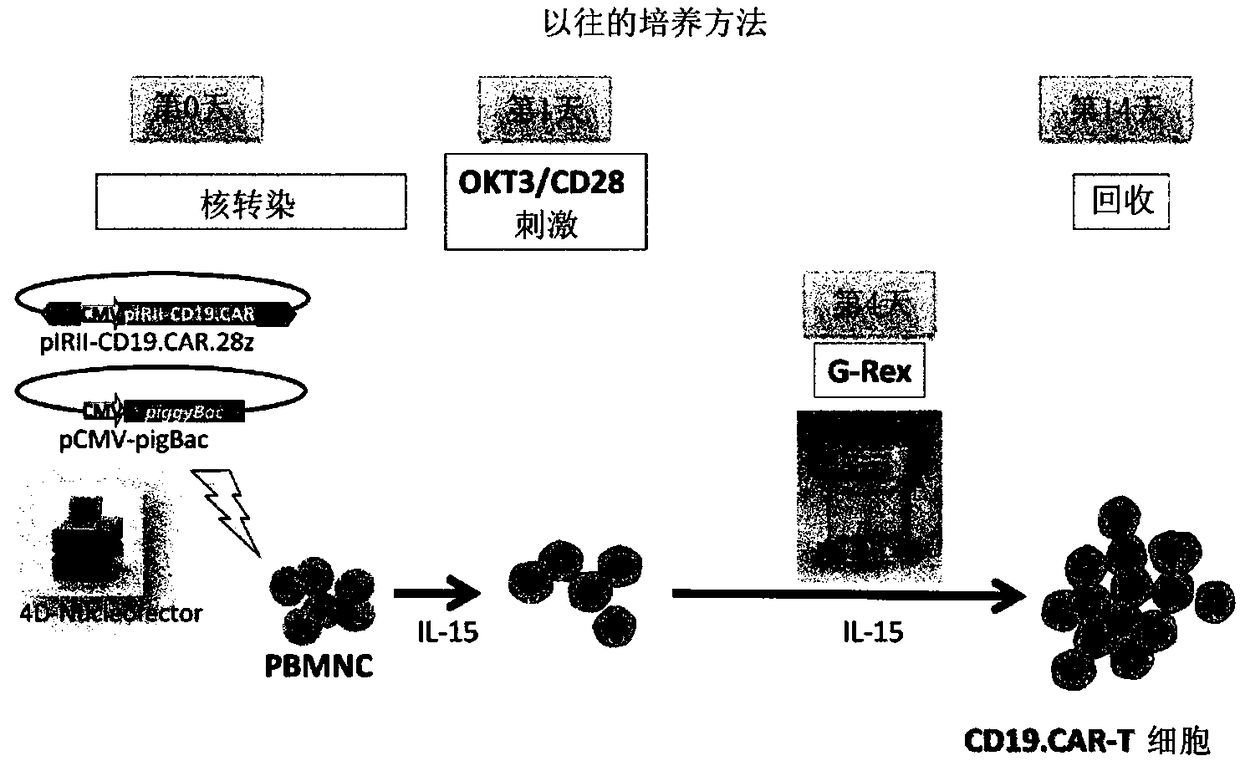
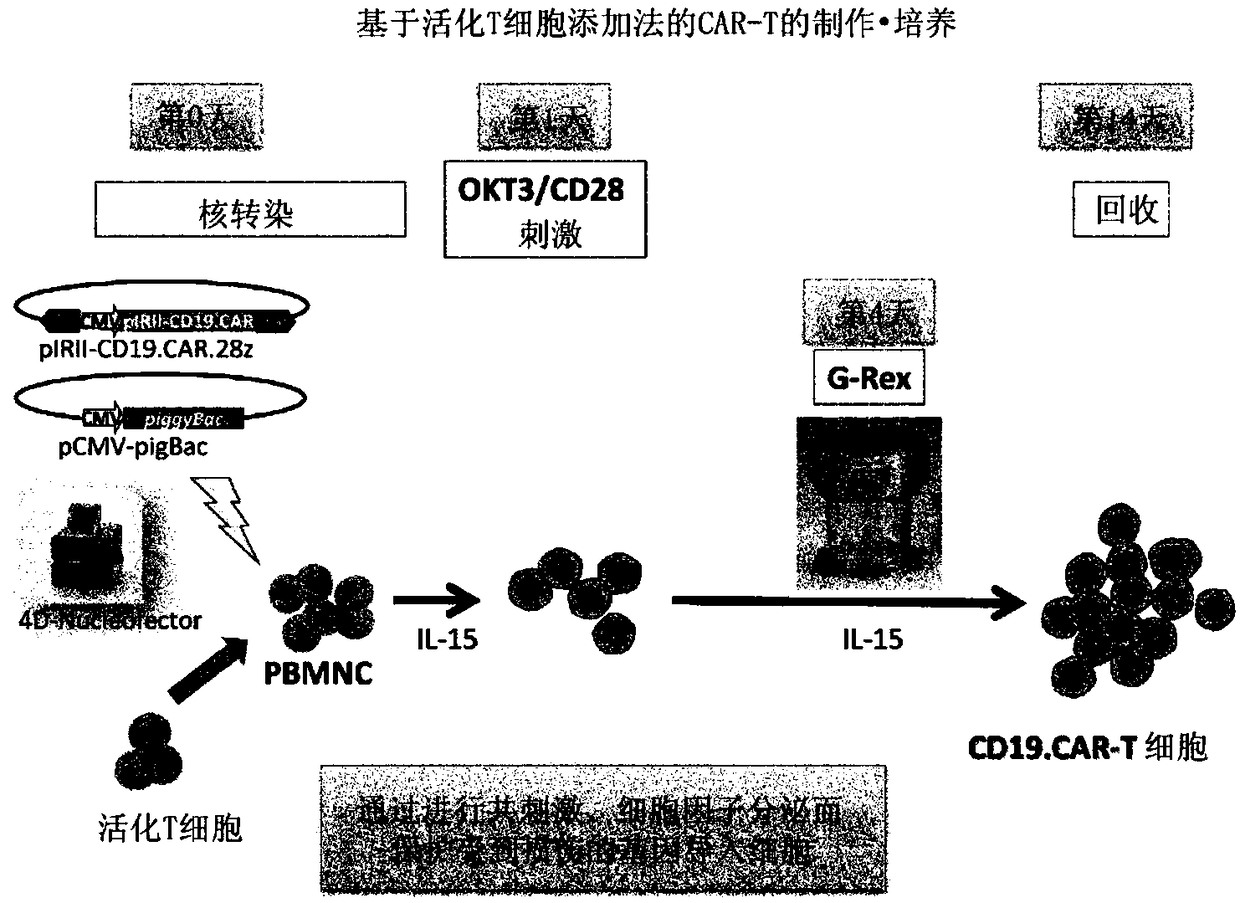
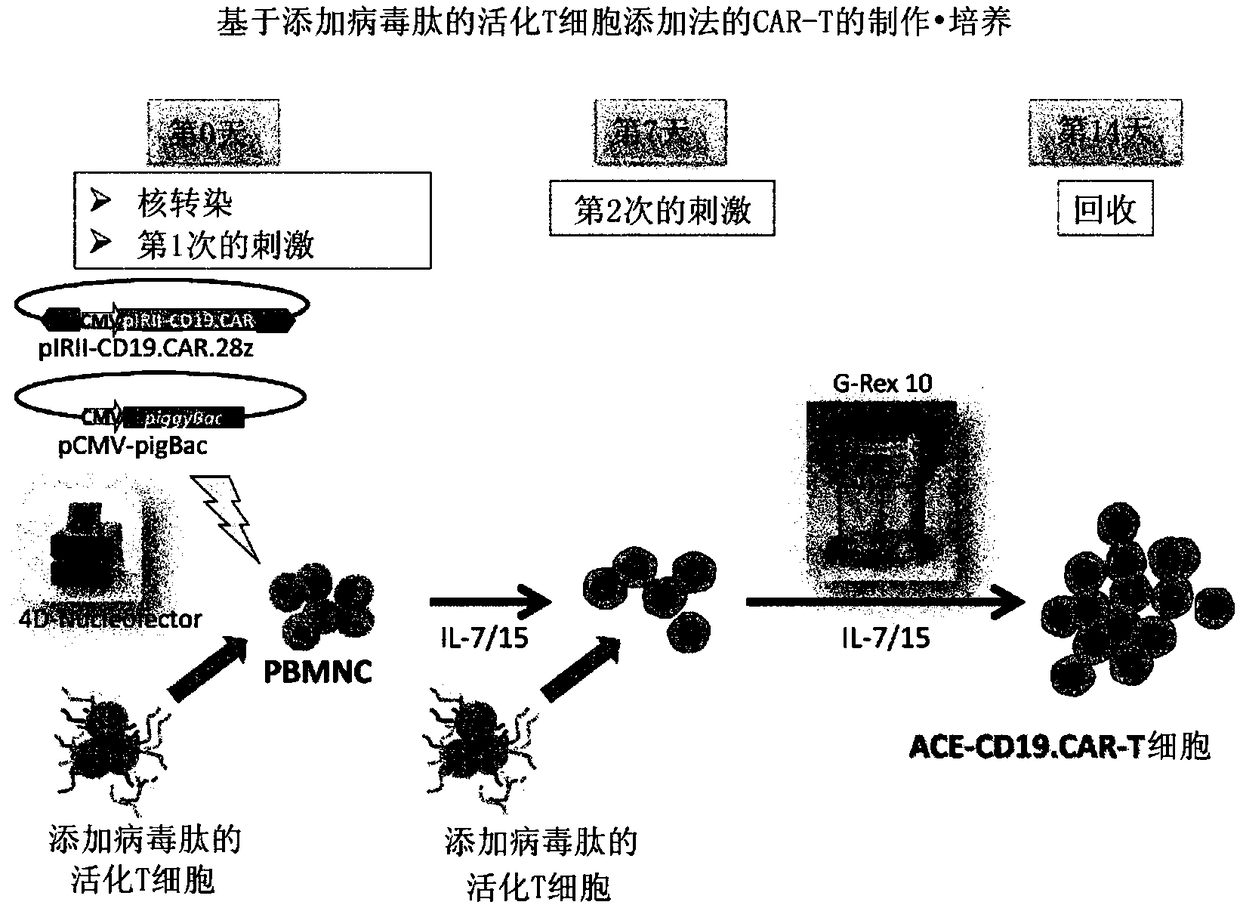
[0095] The preparation method of this mode includes the following steps (i) to (iv). In addition, regarding matters not mentioned (for example, methods for preparing cell populations containing T cells, basic manipulations of stimulation by anti-CD3 The operation of gene introduction, the basic operation of co-cultivation, the recovery method of cells, etc.) are the same as the above-mentioned first production method, therefore, redundant descriptions are omitted, and corresponding descriptions are cited.
[0096] (i) a step of preparing non-proliferative cells retaining viral peptide antigens by stimulating a T cell-containing cell population with an anti-CD3 antibody and an anti-CD28 antibody , obtained by culturing in the presence of viral peptide antigens and incapacitating the proliferative ability
[0097] (ii) A step of obtaining a genetically modified T cell into which a target antigen-specific chimeric antigen receptor gene is introduced by the transposon method
[...
Embodiment
[0115]
[0116] Compared with the case of using a viral vector, the CAR therapy using the transposon method is particularly advantageous in terms of safety. On the other hand, low gene transfer efficiency, easy damage to cells during gene transfer operations (such as electroporation), and a small number of obtained cells are problems. In order to overcome these problems, the following studies have been conducted.
[0117] 1. Materials
[0118] (1) Antibody
[0119] Anti-CD3 antibody (Miltenyi Biotec)
[0120] Anti-CD28 antibody (Miltenyi Biotec)
[0121] (2) culture medium
[0122] TexMACS (Miltenyi Biotec)
[0123] (3) Cytokines
[0124] Recombinant human IL-7 (Miltenyi Biotec company)
[0125] Recombinant human IL-15 (Miltenyi Biotec)
[0126] (4) Viral peptide mixture
[0127] PepTivator (registered trademark) CMV pp65-premium grade, human (Miltenyi Biotec)
[0128] PepTivator (registered trademark) AdV5Hexon-premium grade, human (Miltenyi Biotec company)
[01...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More