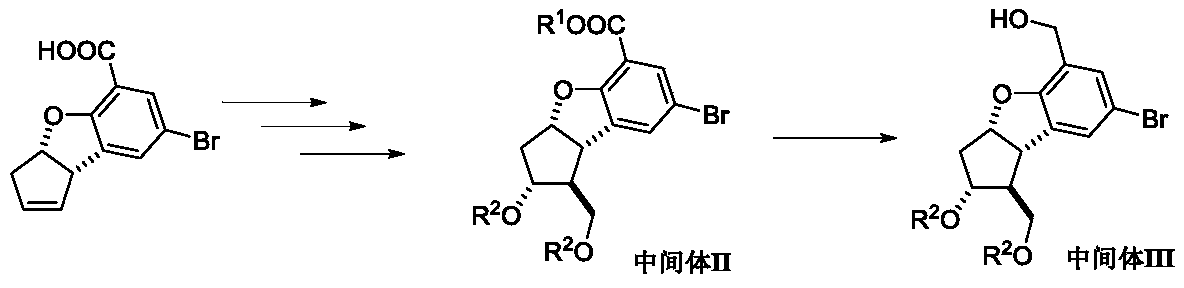
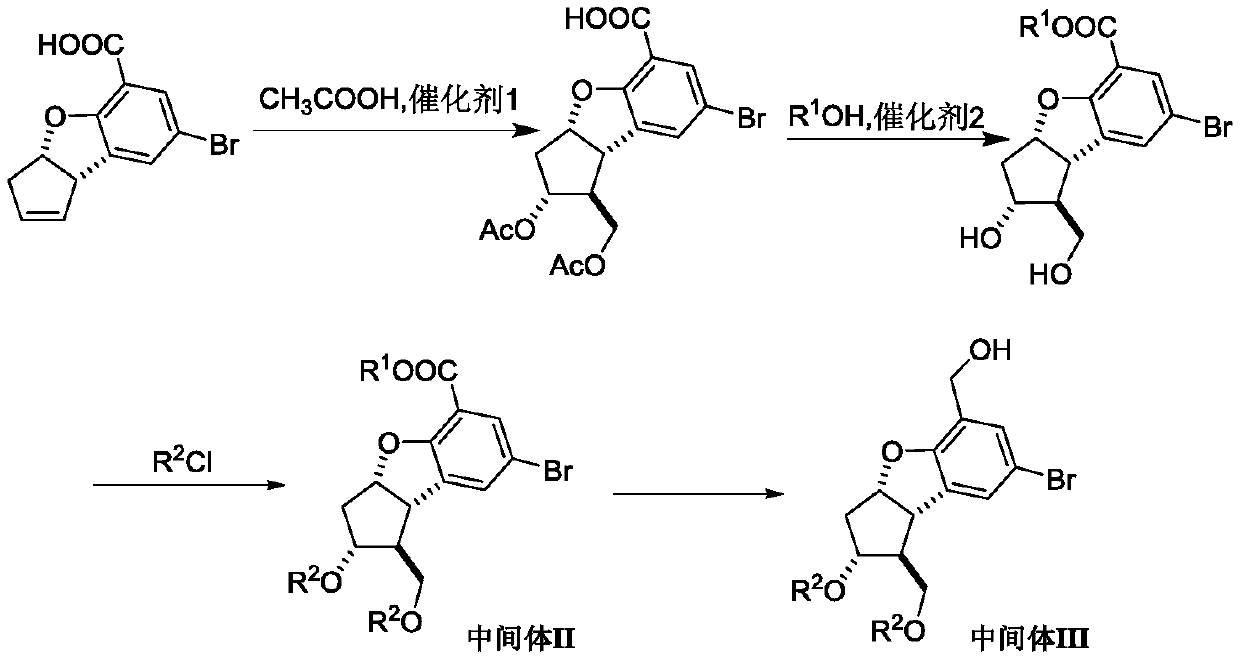
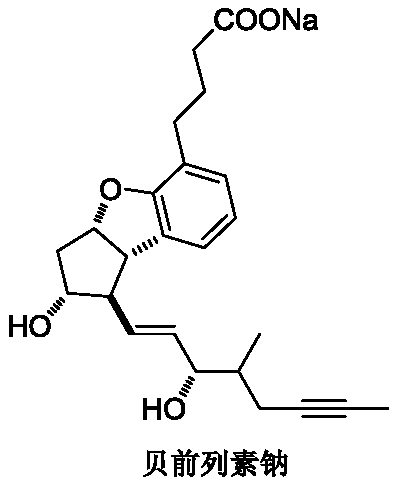
Synthesis method of key intermediate of beprost sodium
A technology of beraprost sodium and its intermediates, which is applied in the field of compound preparation and can solve the problems of severe exothermic post-processing and difficulties
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0077] Embodiment one: the synthesis of intermediate II (R 1 is methyl, R 2 bit TBDMS)
[0078]
[0079] Add 3a,8b-cis-dihydro-3H-5-carboxy-7-dibromocyclopentadiene[b]benzofuran (28 g), acetic acid, paraformaldehyde, concentrated sulfuric acid, and The mass ratio to raw materials is 10:1, the molar ratio of paraformaldehyde to raw materials is 1:1, the molar ratio of concentrated sulfuric acid to raw materials is 2:1, and the system is reacted for 6 hours at 75-80°C under nitrogen atmosphere. Most of the acetic acid was distilled off under reduced pressure, and an aqueous sodium carbonate solution was slowly added to the system. After the bubbles were completely released, the aqueous phase was extracted twice with ethyl acetate, and the ethyl acetate layers were combined, dried, filtered, and the organic phase was rotary evaporated to obtain the crude product ( 1S,2R,3aS,8bS)-2-Acetoxy-1-(acetoxymethyl)-7-bromo-2,3,3a,8b-tetrahydro-1H-cyclopentadiene[b] Benzofuran-5-carb...
Embodiment 2
[0082] Embodiment two: the synthesis of intermediate II (R 1 is methyl, R 2 bit TBDMS)
[0083]
[0084] Add 3a,8b-cis-dihydro-3H-5-carboxy-7-dibromocyclopentadiene[b]benzofuran (28 g), acetic acid, paraformaldehyde, concentrated sulfuric acid, and The mass ratio to raw materials is 4:1, the molar ratio of paraformaldehyde to raw materials is 2:1, the molar ratio of concentrated sulfuric acid to raw materials is 2:1, and the system is reacted for 4 hours at 85-90°C under nitrogen atmosphere. Most of the acetic acid was distilled off under reduced pressure, and an aqueous sodium carbonate solution was slowly added to the system. After the bubbles were completely released, the aqueous phase was extracted twice with ethyl acetate, and the ethyl acetate layers were combined, dried, filtered, and the organic phase was rotary evaporated to obtain the crude product ( 1S,2R,3aS,8bS)-2-Acetoxy-1-(acetoxymethyl)-7-bromo-2,3,3a,8b-tetrahydro-1H-cyclopentadiene[b] Benzofuran-5-carbo...
Embodiment 3
[0087] Embodiment three: the synthesis of intermediate II (R 1 is methyl, R 2 bit TBDMS)
[0088]
[0089] Add 3a,8b-cis-dihydro-3H-5-carboxy-7-dibromocyclopentadiene[b]benzofuran (28 g), acetic acid, paraformaldehyde, concentrated sulfuric acid, and The mass ratio to raw materials is 8:1, the molar ratio of paraformaldehyde to raw materials is 2:1, the molar ratio of concentrated sulfuric acid to raw materials is 3:1, and the system is reacted at 75-80°C for 8 hours under nitrogen atmosphere. Most of the acetic acid was distilled off under reduced pressure, and an aqueous sodium carbonate solution was slowly added to the system. After the bubbles were completely released, the aqueous phase was extracted twice with ethyl acetate, and the ethyl acetate layers were combined, dried, filtered, and the organic phase was rotary evaporated to obtain the crude product ( 1S,2R,3aS,8bS)-2-Acetoxy-1-(acetoxymethyl)-7-bromo-2,3,3a,8b-tetrahydro-1H-cyclopentadiene[b] Benzofuran-5-car...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More