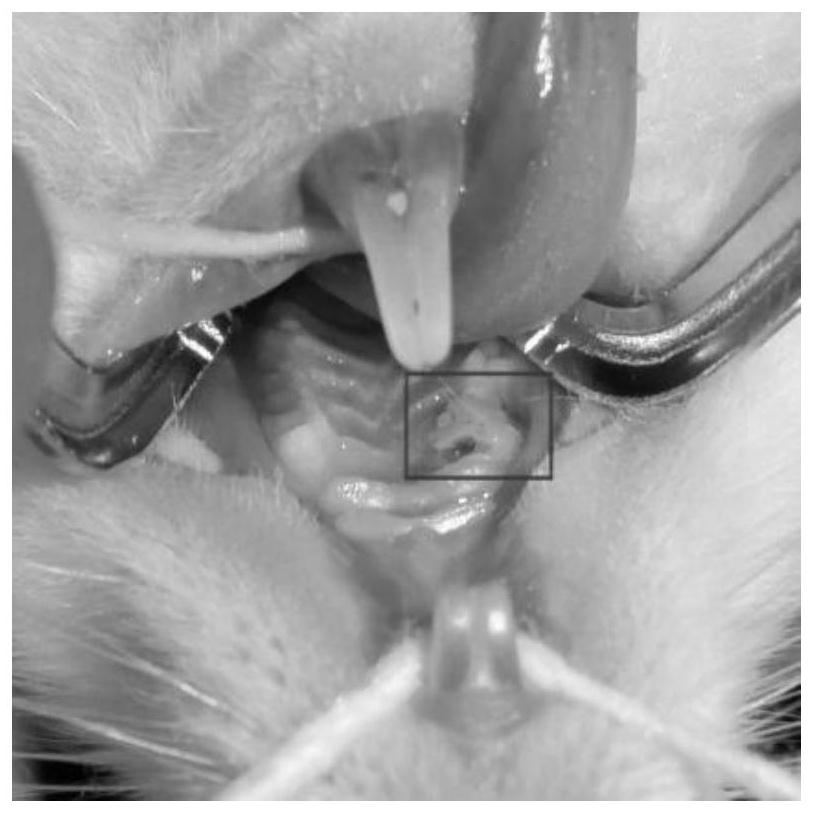
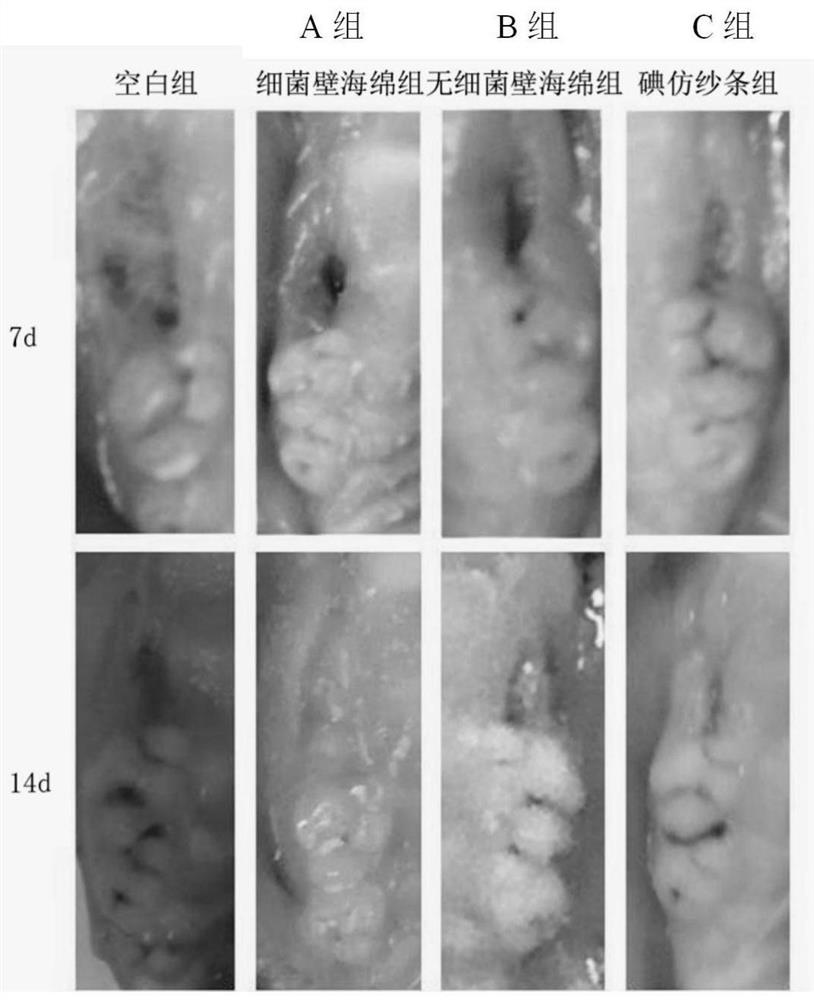
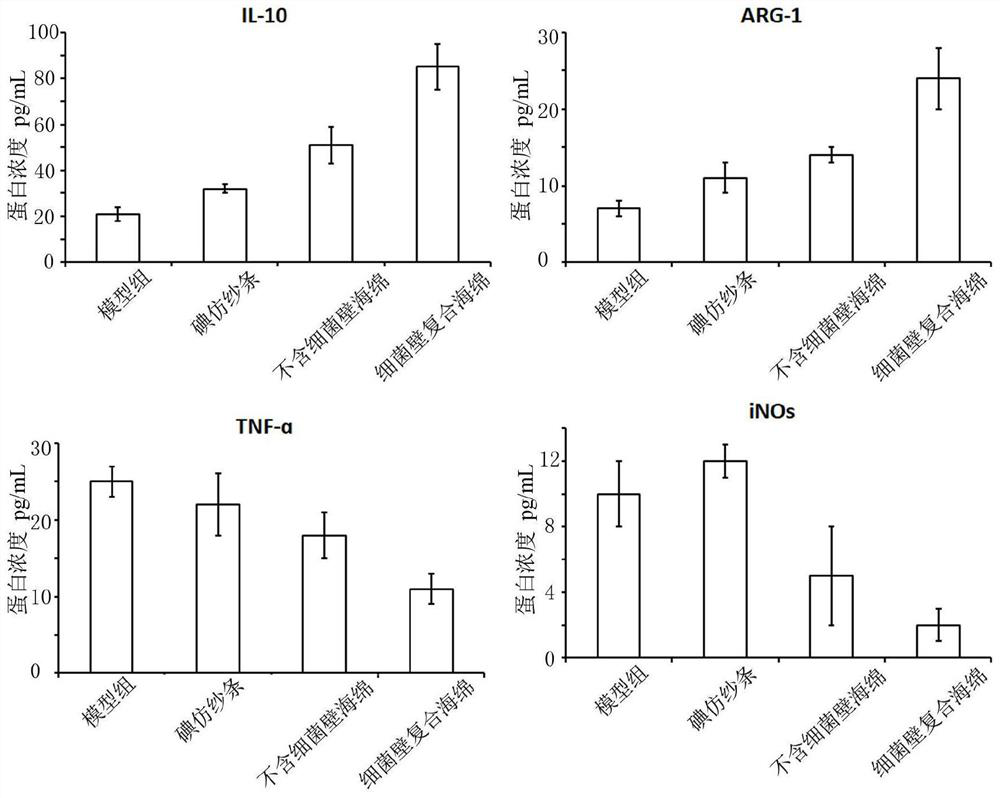
Method for preparing composite sponge for treating dry socket
A composite sponge, dry socket technology, applied in the direction of drug combination, pharmaceutical formulation, medical raw materials derived from bacteria, etc., can solve the problems of taking out the gauze again, slow wound healing, etc., to avoid inflammatory reaction, good therapeutic effect, good The effect of clinical application value
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment Construction
[0022] In order to make the above objects, features and advantages of the present invention more comprehensible, the specific implementation of the present invention will be described in detail below in conjunction with specific examples.
[0023] In the following description, a lot of specific details are set forth in order to fully understand the present invention, but the present invention can also be implemented in other ways different from those described here, and those skilled in the art can do it without departing from the meaning of the present invention. By analogy, the present invention is therefore not limited to the specific examples disclosed below.
[0024] Second, "one embodiment" or "an embodiment" referred to herein refers to a specific feature, structure or characteristic that may be included in at least one implementation of the present invention. "In one embodiment" appearing in different places in this specification does not all refer to the same embodime...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More