Insecticidal and antibacterial 5-imino substituted derivative as well as preparation method and application thereof
A derivative and imino technology, applied in the field of 5-imino substituted derivatives and preparation
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
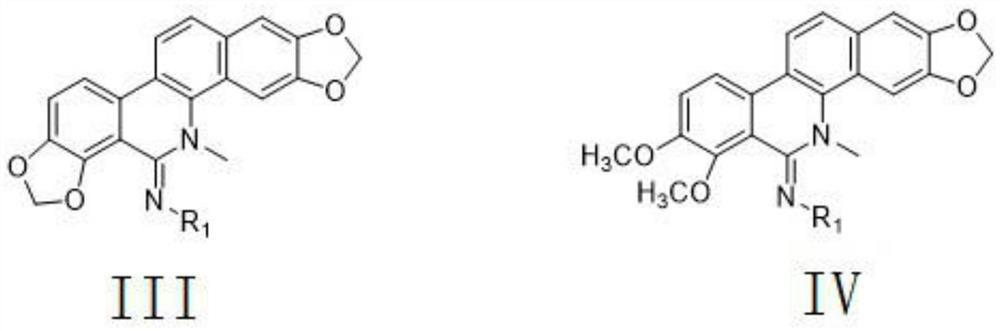
[0069] A 5-imino substituted derivative with insecticidal and antibacterial properties. The 5-imino substituted derivative is a polysubstituted-1H-1,2,4-triazole amidine compound, and its general structural formula is:
[0070]
[0071] Further, R in the general structural formula 1 Selected from H, the general formula is C n h 2n+1 or C m h 2m-1 Straight-chain or branched aliphatic hydrocarbon group, substituted or unsubstituted phenyl, substituted or unsubstituted benzyl, substituted or unsubstituted pyridyl, substituted or unsubstituted furyl, substituted or unsubstituted triazolyl, Wherein, n is an integer of 1-18, and m is an integer of 3-13; R in the general structural formula 1 One or more of substituted or unsubstituted 1,2,4-triazolyl, chloropyridyl, chlorothiazolyl; the substituents are F, Cl, Br, I, hydroxyl, C1 -One or more of 4 alkyl, C1-4 alkoxy, trifluoromethyl, amino, nitro, cyano, carboxyl, propionyloxy, acetoxy, acetyl, formyl; The substituent is mul...
Embodiment 2
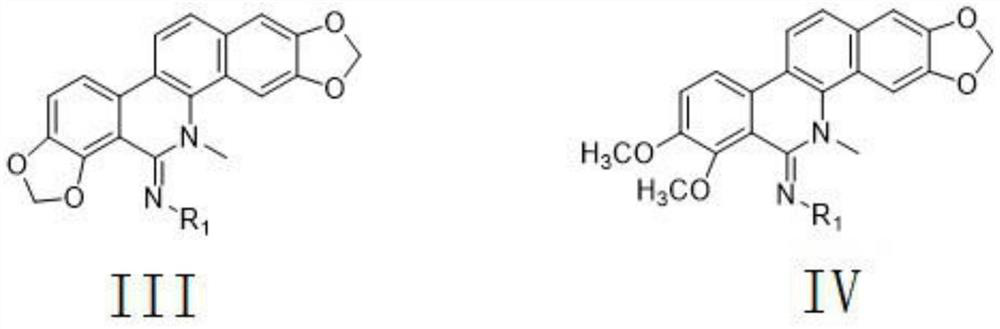
[0083] A 5-imino substituted derivative with insecticidal and antibacterial properties. The 5-imino substituted derivative is a polysubstituted-1H-1,2,4-triazole amidine compound, and its general structural formula is:
[0084]
[0085] Further, R1 in the general structural formula is selected from H, and the general formula is C n h 2n+1 or C m h 2m-1 Straight-chain or branched aliphatic hydrocarbon group, substituted or unsubstituted phenyl, substituted or unsubstituted benzyl, substituted or unsubstituted pyridyl, substituted or unsubstituted furyl, substituted or unsubstituted triazolyl, Among them, n is an integer of 1-18, m is an integer of 3-13; R1 in the general structure formula is a substituted or unsubstituted 1,2,4-triazolyl, chloropyridyl, chlorothiazolyl One or more than one; the substituents are F, Cl, Br, I, hydroxyl, C1-4 alkyl, C1-4 alkoxy, trifluoromethyl, amino, nitro, cyano, carboxyl One or more of , propionyloxy, acetyloxy, acetyl, and formyl; the ...
Embodiment 3
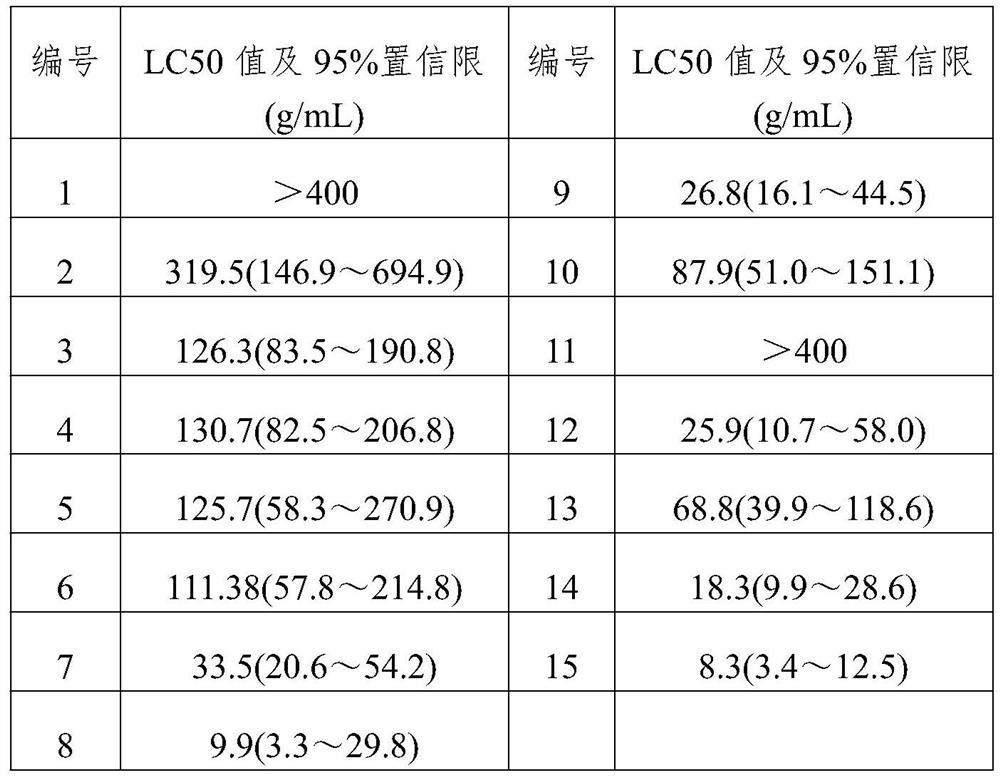
[0097] A 5-imino substituted derivative with insecticidal and antibacterial properties. The 5-imino substituted derivative is a polysubstituted-1H-1,2,4-triazole amidine compound, and its general structural formula is:
[0098]
[0099] Further, R1 in the general structural formula is selected from H, and the general formula is C n h 2n+1 or C m h 2m-1 Straight-chain or branched aliphatic hydrocarbon group, substituted or unsubstituted phenyl, substituted or unsubstituted benzyl, substituted or unsubstituted pyridyl, substituted or unsubstituted furyl, substituted or unsubstituted triazolyl, Among them, n is an integer of 1-18, m is an integer of 3-13; R1 in the general structure formula is a substituted or unsubstituted 1,2,4-triazolyl, chloropyridyl, chlorothiazolyl One or more than one; the substituents are F, Cl, Br, I, hydroxyl, C1-4 alkyl, C1-4 alkoxy, trifluoromethyl, amino, nitro, cyano, carboxyl One or more of , propionyloxy, acetyloxy, acetyl, and formyl; the ...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More