Process for catalytic synthesis of 2-cyano-4'-methyl biphenyl
A methyl biphenyl and process technology, applied in the field of catalytic synthesis of 2-cyano-4'-methyl biphenyl, can solve the problem of large amount of metal palladium and the like, achieve easy recovery and reuse, simple reaction conditions and high activity Effect
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
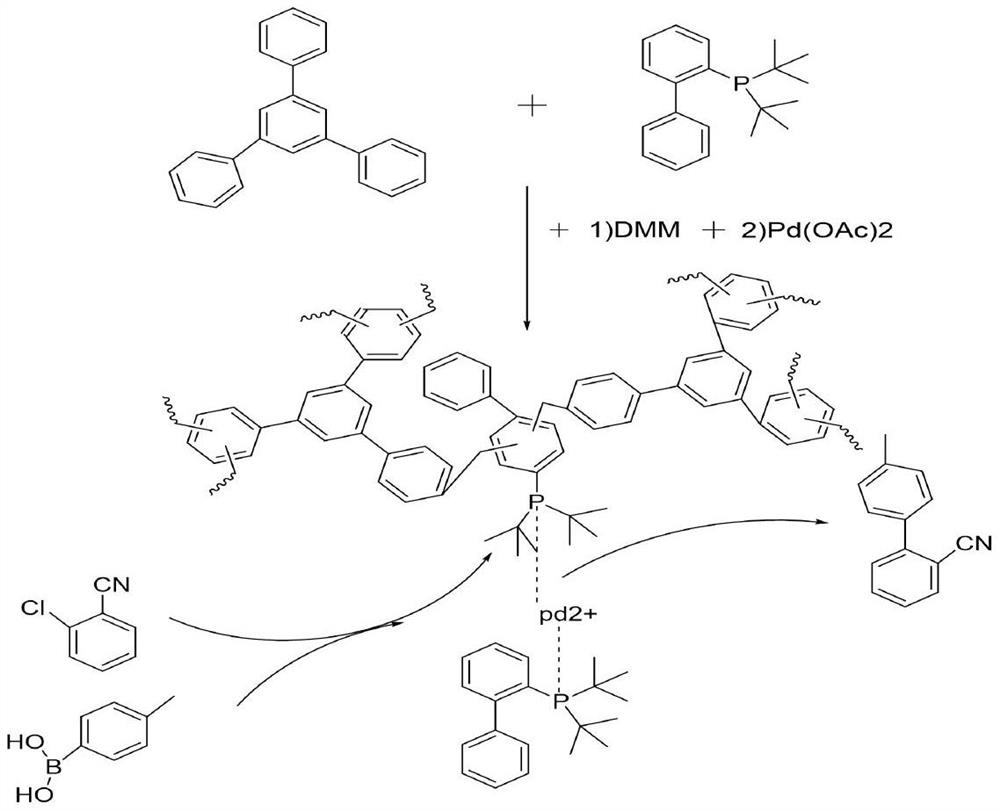
[0019] Add 2-(di-tert-butylphosphine)biphenyl and triphenylbenzene 1:1 (2-(di-tert-butylphosphine)biphenyl dosage 100mg) into the reactor, then add 2g dimethoxymethane and 1g of phosphotungstic acid was quickly flushed into nitrogen, and reacted at 150°C for 8 hours to obtain a phosphorus-containing palladium catalyst support, which was used to prepare 2-cyano-4'-methylbiphenyl from o-chlorobenzonitrile.
[0020] In the pressure tube, add 137.5mg (1mmoL) of o-chlorobenzonitrile, and 163.1mg (1.2mmoL) of p-tolueneboronic acid and 2 equivalents of CSCO 3 , 100 mg containing 2-(di-tert-butylphosphine) biphenyl polymer carrier and palladium acetate (1%) and solvent dioxane 2 mL. Fill the pressure tube with argon gas and tighten the pressure tube, put it in an oil bath at 130°C for 5 hours, after the reaction, take out the pressure tube and cool it down to room temperature, take a sample, and check the conversion rate and selectivity by gas chromatography. The reaction results are ...
Embodiment 2
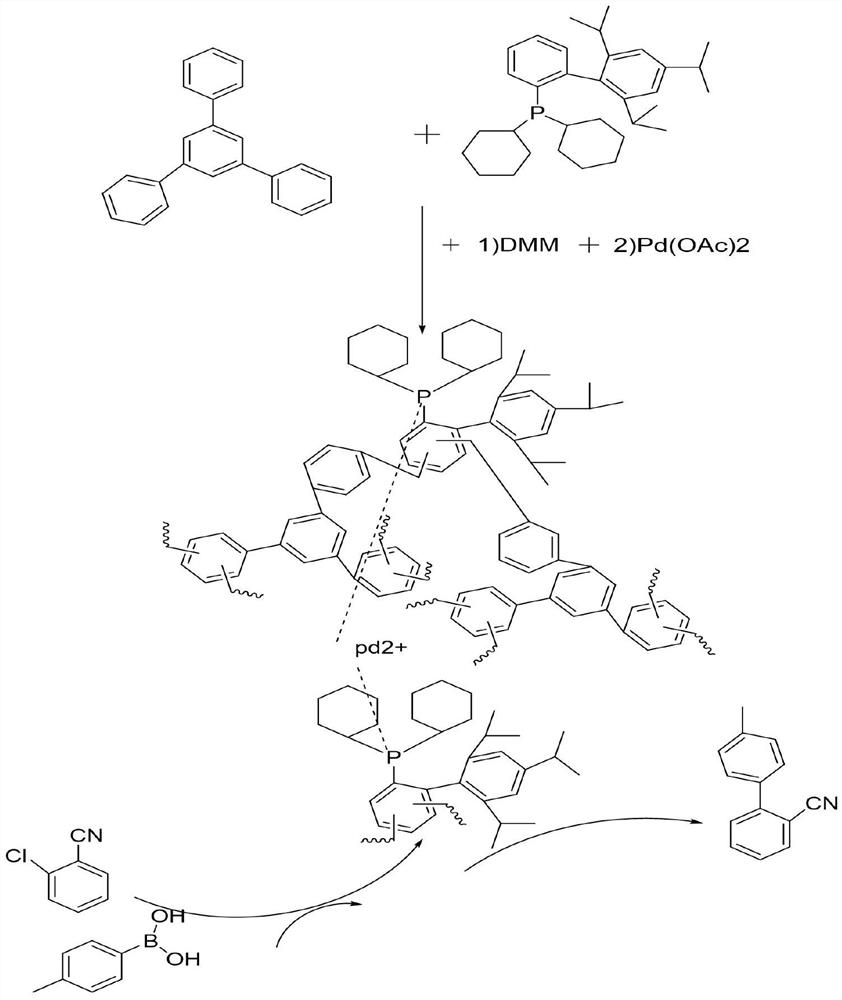
[0024] Add XPhos and triphenylbenzene 1:2 (the dosage of XPhos is 100mg) into the reaction kettle, then add 3g of dimethoxymethane and 2g of phosphotungstic acid, quickly flush with argon, react at 170°C for 10h, and get Phosphine-containing palladium catalyst support for the preparation of 2-cyano-4'-methylbiphenyl from o-chlorobenzonitrile.
[0025] In the pressure tube, add 137.5mg (1mmoL) of o-chlorobenzonitrile, and 163.1mg (1.2mmoL) of p-tolueneboronic acid and 2 equivalents of CSCO 3 , 100 mg containing XPhos polymer carrier and palladium acetate (1%) and solvent dioxane 2 mL. Fill the pressure tube with nitrogen and tighten it, put it in an oil bath at 130°C and react for 10 hours. After the reaction, take out the pressure tube and cool it down to room temperature, take a sample, and use gas chromatography to detect the conversion rate and selectivity. The reaction results are shown in Table 1. As shown in Example 2;
[0026]
[0027] Preparation of 2-cyano-4'-met...
Embodiment 3
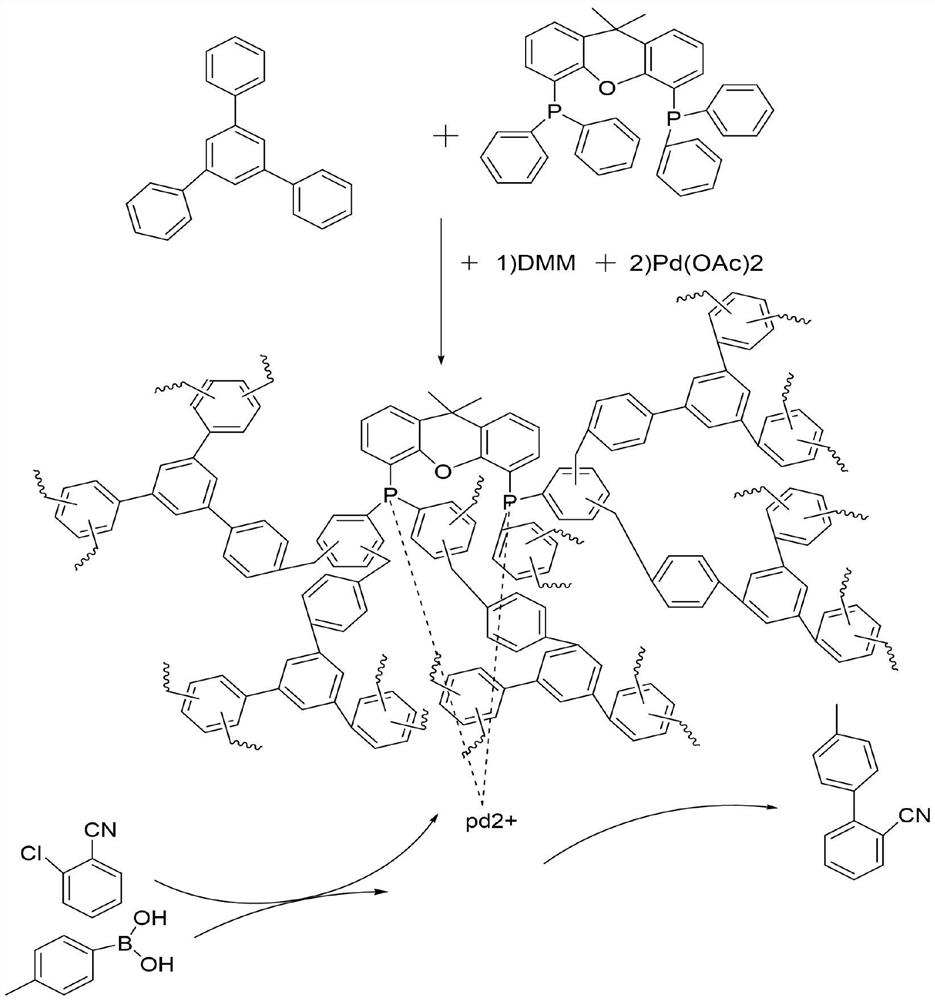
[0029] Add XantPhos and triphenylbenzene 1:3 (XantPhos dosage 100mg) into the reaction kettle, then add 4g of dimethoxymethane and 3g of phosphotungstic acid, quickly flush with argon, react at 180°C for 15h, and get Phosphine-containing palladium catalyst support for the preparation of 2-cyano-4'-methylbiphenyl from o-chlorobenzonitrile.
[0030] In the pressure tube, add 137.5mg (1mmoL) of o-chlorobenzonitrile, and 163.1mg (1.2mmoL) of p-tolueneboronic acid and 2 equivalents of CSCO 3 , 100 mg containing XantPhos polymer carrier and palladium acetate (1%) and solvent dioxane 2 mL. Fill the pressure tube with argon gas and tighten the pressure tube, put it in an oil bath at 130°C for 15 hours. After the reaction, take out the pressure tube and cool it down to room temperature, take a sample, and use gas chromatography to detect the conversion rate and selectivity. The reaction results are shown in Table 1. Shown in embodiment 3;
[0031]
[0032] Preparation of 2-cyano-4...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More