Polysubstituted furo-cycloheptatriene pyrrole derivative and preparation method thereof
A technology of cycloheptatriene pyrrole and polysubstituted furan, which is applied in the field of polysubstituted furanocycloheptatriene pyrrole derivatives and preparation thereof, can solve the problems of low atom utilization rate, complicated process, severe reaction conditions and the like, and achieves The raw materials and reagents are stable and easy to obtain, the preparation method is simple, and the water and oxygen tolerance is good.
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
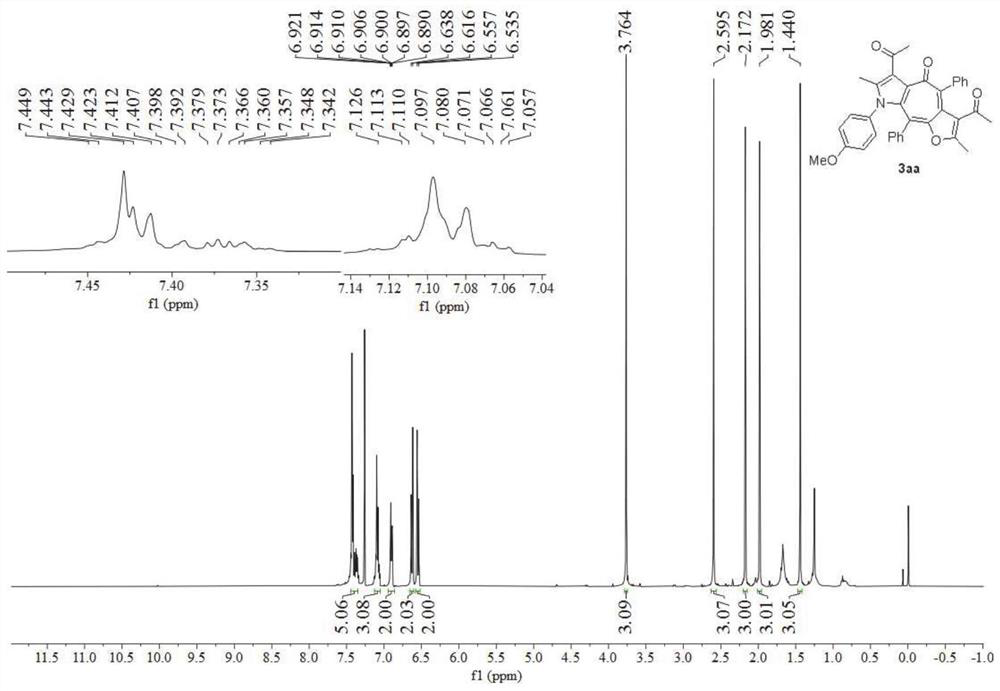
[0049] Preparation of polysubstituted furocycloheptatriene pyrrole derivatives 3aa:
[0050]
[0051] Add p-methoxyphenylisonitrile 1a (0.3mmol), 3-(3-phenylpropyl-2-yn-1-ylidene)-2,4-pentanedione 2a (0.75 mmol), and dissolve with dichloromethane (2mL), add stirring bar, add zinc chloride (0.3mmol), after stirring at room temperature for 10 minutes, add m-chloroperoxybenzoic acid (0.3mmol) in the reaction system, Continue to stir the reaction system at room temperature for 18 h. At this time, TLC monitors that 3aa is completely formed. Then, aqueous sodium bisulfite solution (0.1N×5 mL) is added to the system to quench the reaction, and dichloromethane (10 mL×3) Extract the aqueous phase, mix the obtained organic phases and add anhydrous sodium sulfate to dry, then evaporate the organic solvent through a rotary evaporator, and finally, separate the final product through silica gel column chromatography, and obtain the final product through nuclear magnetic hydrogen spectrum...
Embodiment 2
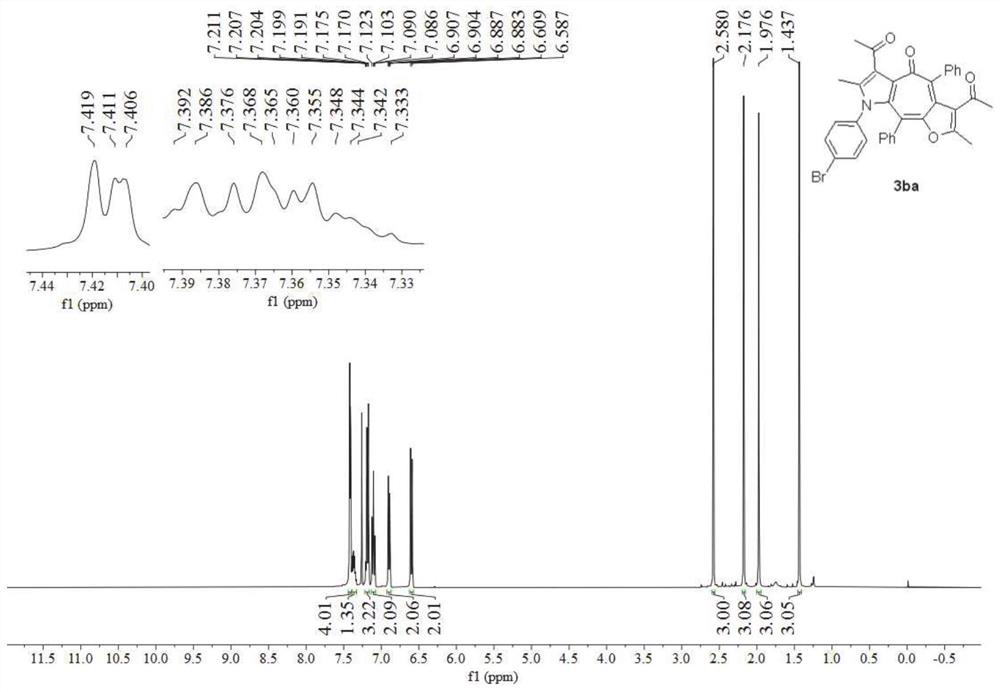
[0055] Preparation of multi-substituted furocycloheptatrienyl pyrrole derivatives 3ba:
[0056]
[0057] P-bromophenylisonitrile 1b was used to replace p-methoxyphenylisonitrile 1a in Example 1, and other conditions were the same as in Example 1 to obtain a multi-substituted furocycloheptatrienyl pyrrole derivative 3ba with a yield of 74% .
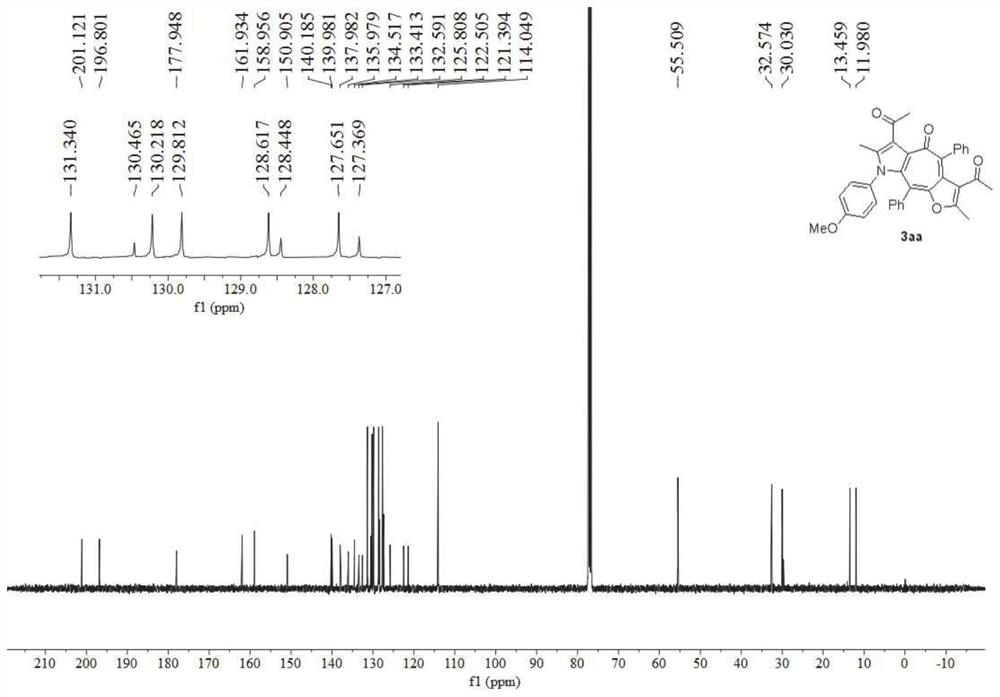
[0058] image 3 It is the nuclear magnetic hydrogen spectrogram of the furo cycloheptatrienyl pyrrole derivative obtained in Example 2 of the present invention, Figure 4 Its nuclear magnetic carbon spectrum, spectrum analysis data:
[0059] 1 H NMR (400MHz, CDCl 3 )δ7.44–7.39(m,4H),7.39–7.33(m,1H),7.22–7.16(m,3H),7.10(t,J=7.5Hz,2H),6.92–6.87(m,2H) ,6.60(d,J=8.6Hz,2H),2.58(s,3H),2.18(s,3H),1.98(s,3H),1.44(s,3H). 13 C NMR (101MHz, CDCl 3 )δ200.8,196.6,177.8,162.0,150.9,139.8,139.3,138.2,136.8,136.1,134.2,133.3,132.7,131.9,131.2,130.4,130.3,128.6,128.5,127.8,127.6,126.2,122.5,122.3,120.8 ,32.5,30.0,13.4,11.9.HRMS(ESI-TOF)m / z calc...
Embodiment 3
[0061] Preparation of polysubstituted furocycloheptatriene pyrrole derivatives 3ca:
[0062]
[0063] Use p-ethoxycarbonylphenyl isonitrile 1c to replace p-methoxyphenyl isonitrile 1a in Example 1, and other conditions are the same as in Example 1 to obtain multi-substituted furocycloheptatriene pyrrole derivatives 3ca with a yield of 72%.
[0064] Spectral analysis data:
[0065] 1 H NMR (400MHz, CDCl 3 )δ7.74(d,J=8.4Hz,2H),7.43–7.33(m,5H),7.12–7.06(m,1H),7.05–7.00(m,2H),6.94–6.88(m,2H) ,6.81(d,J=8.4Hz,2H),4.38(q,J=7.1Hz,2H),2.58(s,3H),2.18(s,3H),1.95(s,3H),1.44(s, 3H), 1.40(t, J=7.1Hz, 3H). 13 C NMR (101MHz, CDCl 3 )δ200.7,196.6,177.9,165.3,162.1,150.9,141.7,139.8,139.2,138.3,136.1,134.1,133.3,132.9,131.2,130.5,130.0,129.9,128.8,128.6,128.5,127.8,127.7,126.3,122.5 , 120.8, 61.3, 32.5, 30.0, 14.2, 13.4, 11.9. HRMS (ESI-TOF) m / z calculated for C 38 h 31 NNaO 6 + ([M+Na] + )620.2044, found 620.2023.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com