Synthesis method of diafenthiuron impurity C
A technology of diafenthiuron and impurities, applied in the field of medicinal chemistry to achieve the effect of improving quality standards
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
preparation example Construction
[0029] The invention provides the preparation method of diafenthiuron impurity C, such as figure 1 , including the following steps:
[0030] Step S1: Dissolve 2,6-isopropylaniline in pyridine, then add 4-toluenesulfonyl chloride to obtain the first reaction solution, heat to reflux until the reaction is complete, mix the cooled first reaction solution with a sufficient amount of HCl The solution was mixed uniformly, and the first solid was obtained by suction filtration, and the first solid was dissolved in ethanol, and compound 3 was obtained by recrystallization;
[0031] Step S2: HNO 3 , the compound 3, AcOH, NaNO 2 sequentially added to H 2 Obtain the second reaction solution in 0, stir and react at the first temperature for a period of time, then raise the temperature to the second temperature and stir the reaction until the reaction is complete, mix the cooled first reaction solution with a sufficient amount of HCl solution, and pump Solid compound 4 was obtained by ...
Embodiment 1
[0039] This embodiment provides the preparation method of compound 3 (i.e. step S1):
[0040] Compound 1 (2,6-isopropylaniline, 0.2mol, 35.4g) was dissolved in 75ml of pyridine (Pyridine), and compound 2 (4-toluenesulfonyl chloride, 0.22mol, 41.91g) was added to obtain the first reaction solution, Heated to reflux for 4h, TLC detected that the reaction was complete, cooled, poured into 2mol / L HCl solution, stirred for 0.5h, and filtered to obtain a solid, which was dissolved in ethanol (EA) and recrystallized to obtain compound 3 (50g, yield : 79%).
[0041] The prepared compound 3 was detected by proton nuclear magnetic spectrum:
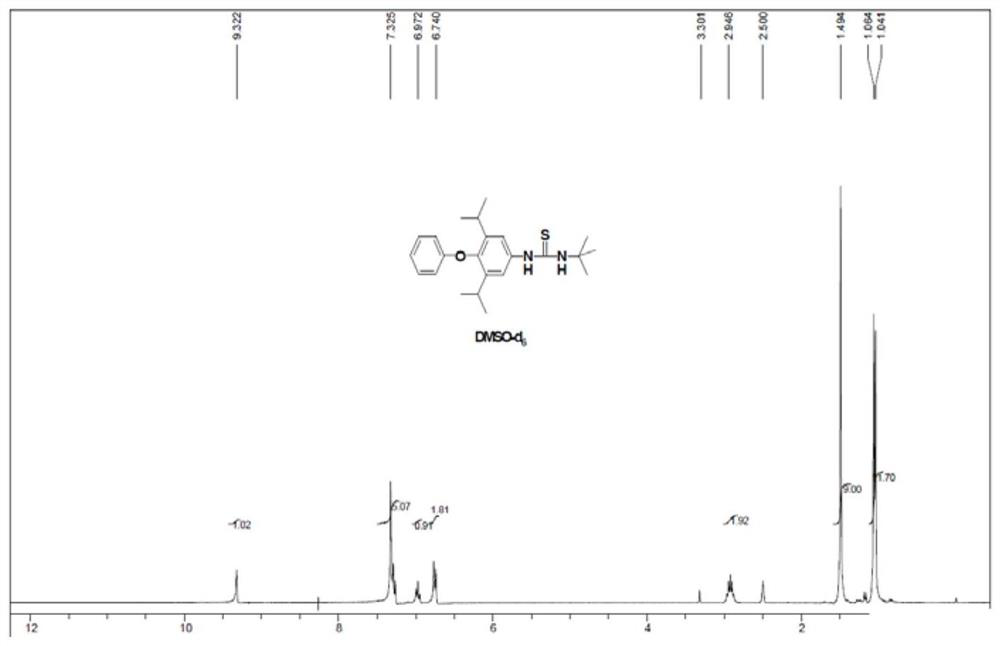
[0042] 1 HNMR (300MHz, CDCl 3 )δ0.91(d,12H),2.37(s,3H),3.08-3.11(m,2H), 7.10(d,2H),7.20(t,1H),7.38(d,2H),7.60(d ,2H),9.35(s,1H)
Embodiment 2
[0044]This embodiment provides the preparation method of compound 4 (i.e. step S2):
[0045] 30ml concentrated HNO 3 (concentration 65%) was added to 160ml H 2 O, then sequentially added compound 3 (7g, 0.021mol), 140ml AcOH, NaNO 2 (2.23g, 0.032mol) to obtain the second reaction solution, stirred at room temperature for 24h, then heated up to 100°C for 4h, TLC detected that the reaction was complete, cooled, poured into 500ml H 2 O, stirred for 0.5 h, and was filtered with suction to obtain solid compound 4 (4 g, yield: 50%).
[0046] The prepared compound 4 was carried out by proton nuclear magnetic spectrum detection:
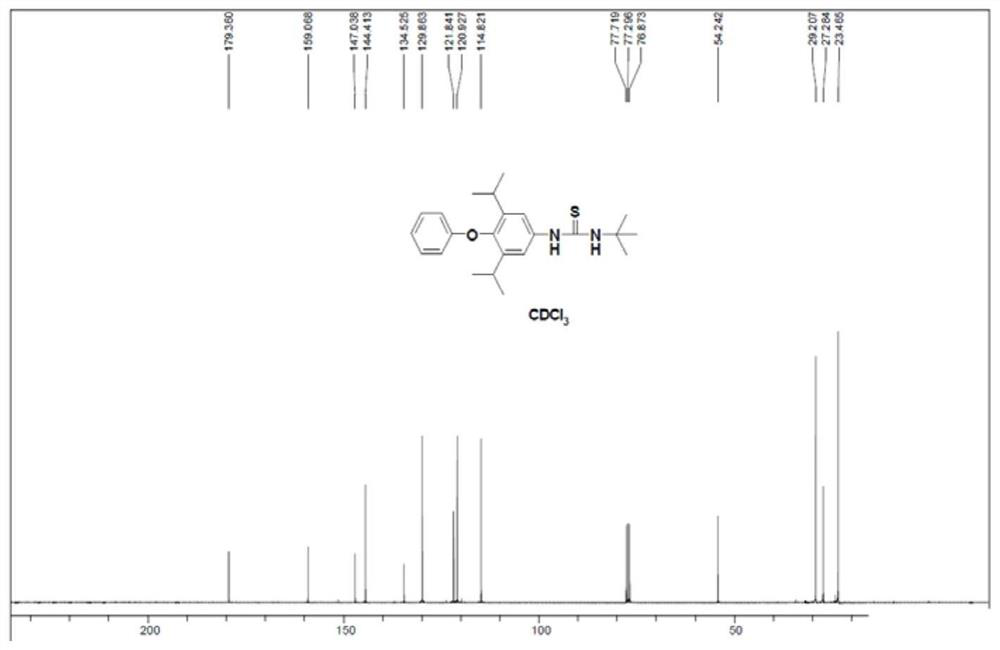
[0047] 1 HNMR (300MHz, CDCl 3 )δ1.02(d,12H),2.44(s,3H),3.10-3.22(m,2H), 6.24(s,2H),7.28(d,2H),7.61(d,2H),7.98(s ,1H)
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More