Adenosine-coated liposome
A technology of liposome and adenosine, applied in the manufacture and use of the injectable preparation, the field of injectable preparation
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0074] This embodiment describes the disclosure of the lipid body.
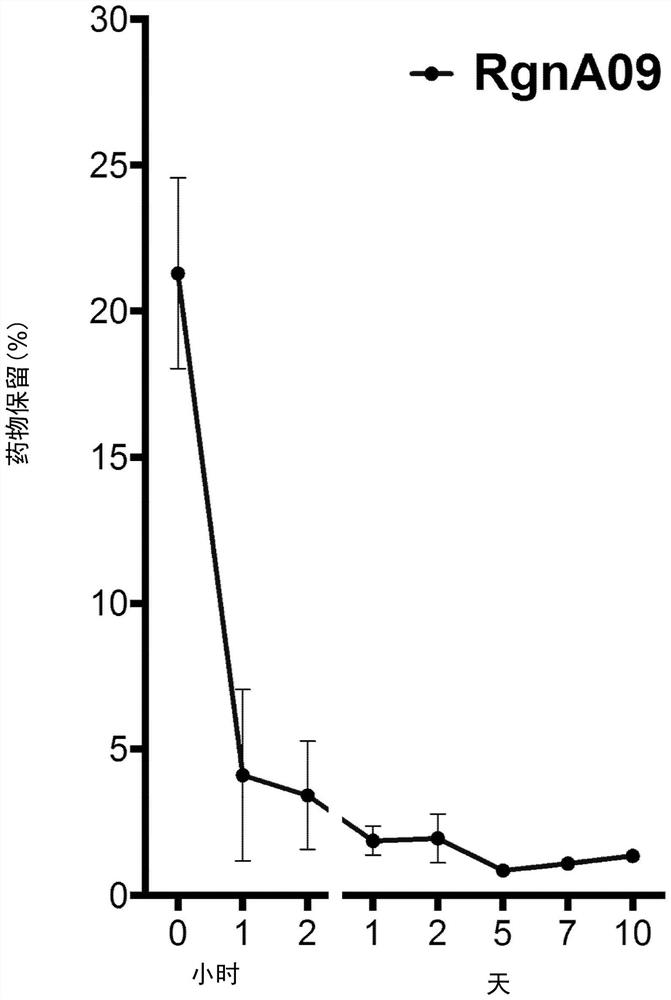
[0075] Because the half-life of adenosine is only a few seconds, the lipid-adenosine used in the above studies is freshly prepared every day. It developed and evaluated a series of storage and stable preparation solutions based on lipid group division, dissolving efficiency, and retaining characteristics. Determine whether it should include cholesterol / stabilizer and optimize these variables. The percentage of liposome-bound adenosine in the total formulation was measured and could be increased or decreased by reducing or increasing the amount of adenosine solution used to hydrate a fixed amount of pre-liposomal lyophilizate. The ratio of adenosine to (adenosine + lipid) in the resulting liposomal particles formed by hydrating the preliposomal lyophilizate with adenosine solution depends on the adenosine concentration in the solution rather than the used volume.
[0076] All preparations are hydrated with ...
Embodiment 2
[0088] This embodiment describes the disclosure of the lipid body.
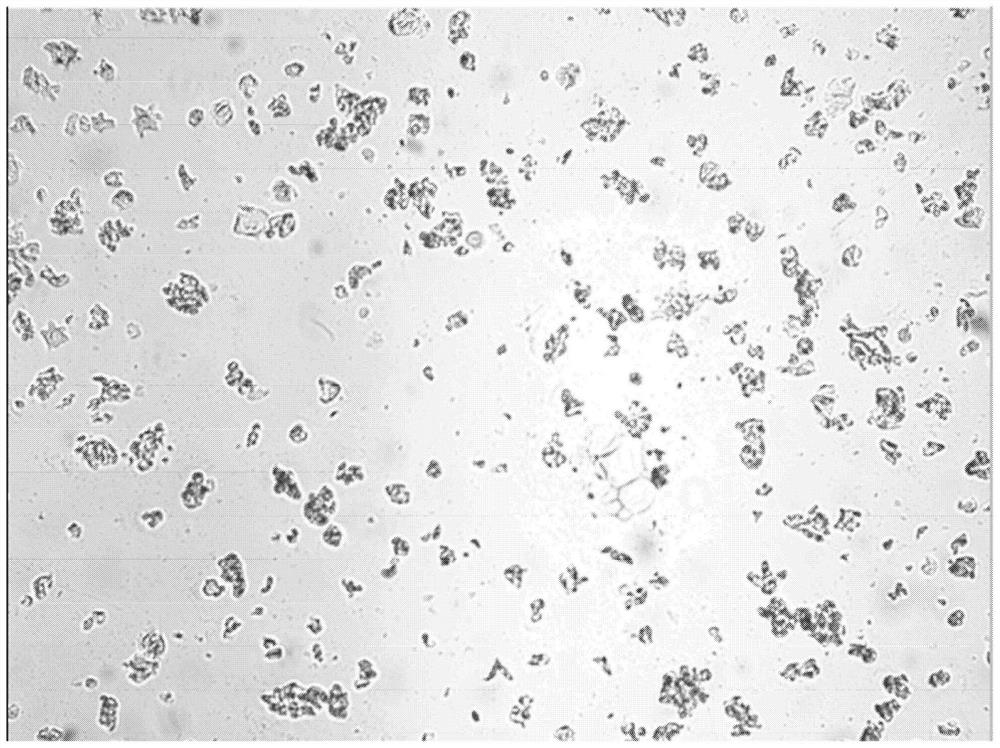
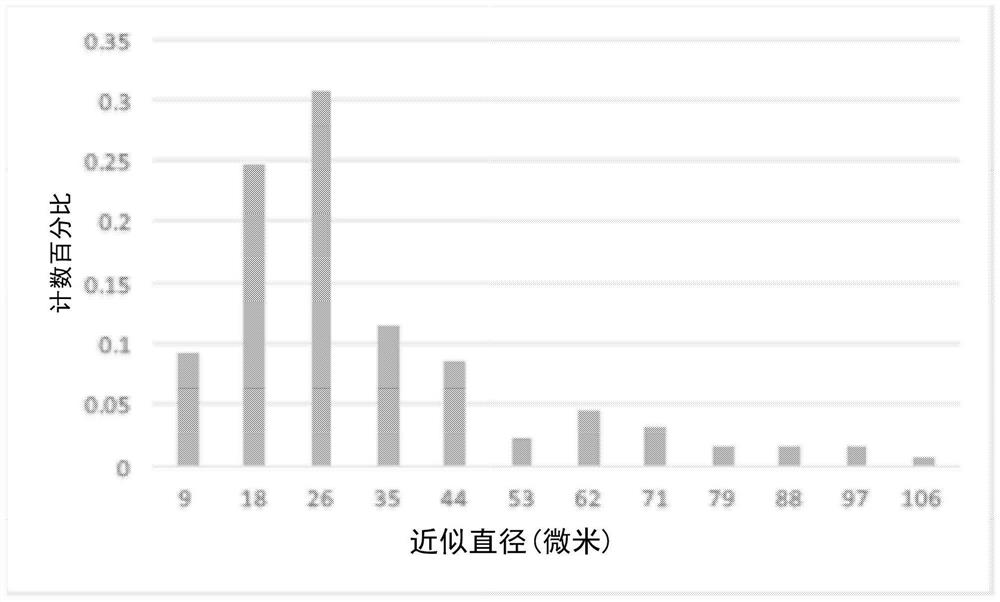
[0089] The laboratory preparations provided by cross-polarized microscope to the provided lipid-adenosine were analyzed. The results are shown in Figure 8 .
[0090]When viewed under a microscope, the liposome suspension showed signs of clearly crystallized adenosine. It is believed that the liposomal suspension may not contain substantial liposomal content, mainly an emulsion. The spherical object in the image is probably oil. The composition of the formulation contains a substantial proportion (60%) of soybean oil, which would not be a component of the liposomal formulation, but would be a component of the emulsion.
[0091] Taking into account the solubility of adenosine in water, it was 7 mg / mL. The laboratory formulation calls for 300 mg of adenosine to be added to 10 mL of normal saline. Presumably, only 70 mg of adenosine will dissolve, leaving 230 mg of adenosine as crystals. It is thought tha...
Embodiment 3
[0113] This example describes methods of using the injectable formulations of the present disclosure.
[0114] Rats with established OA received intra-articular injections of saline (100 μL), while the other 8 groups of animals received Ade in 2 different liposomal formulations at doses of 3, 1, 0.3 and 0 mg / mL. The first injection was given 4 weeks after the ACL ruptured. Animals received 6 injections every 10 days. Knee joint swelling was measured prior to each injection as a measure of joint inflammation. Rats were tested for pain at baseline (before the first injection), 5 days after the third injection, and finally at day 57, just before sacrifice (7 days after the last injection). Post-execution joints were analyzed by histology and uCT.
[0115] 10 treatment groups
[0116] 2 preparations x 4 doses = 8 treatment groups
[0117] 1 positive control (Rgn01)
[0118] 1 negative control (normal saline)
[0119] RgnA01 The preparation is according to Corciulo et al. ...
PUM
| Property | Measurement | Unit |
|---|---|---|
| diameter | aaaaa | aaaaa |
| diameter | aaaaa | aaaaa |
| diameter | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More