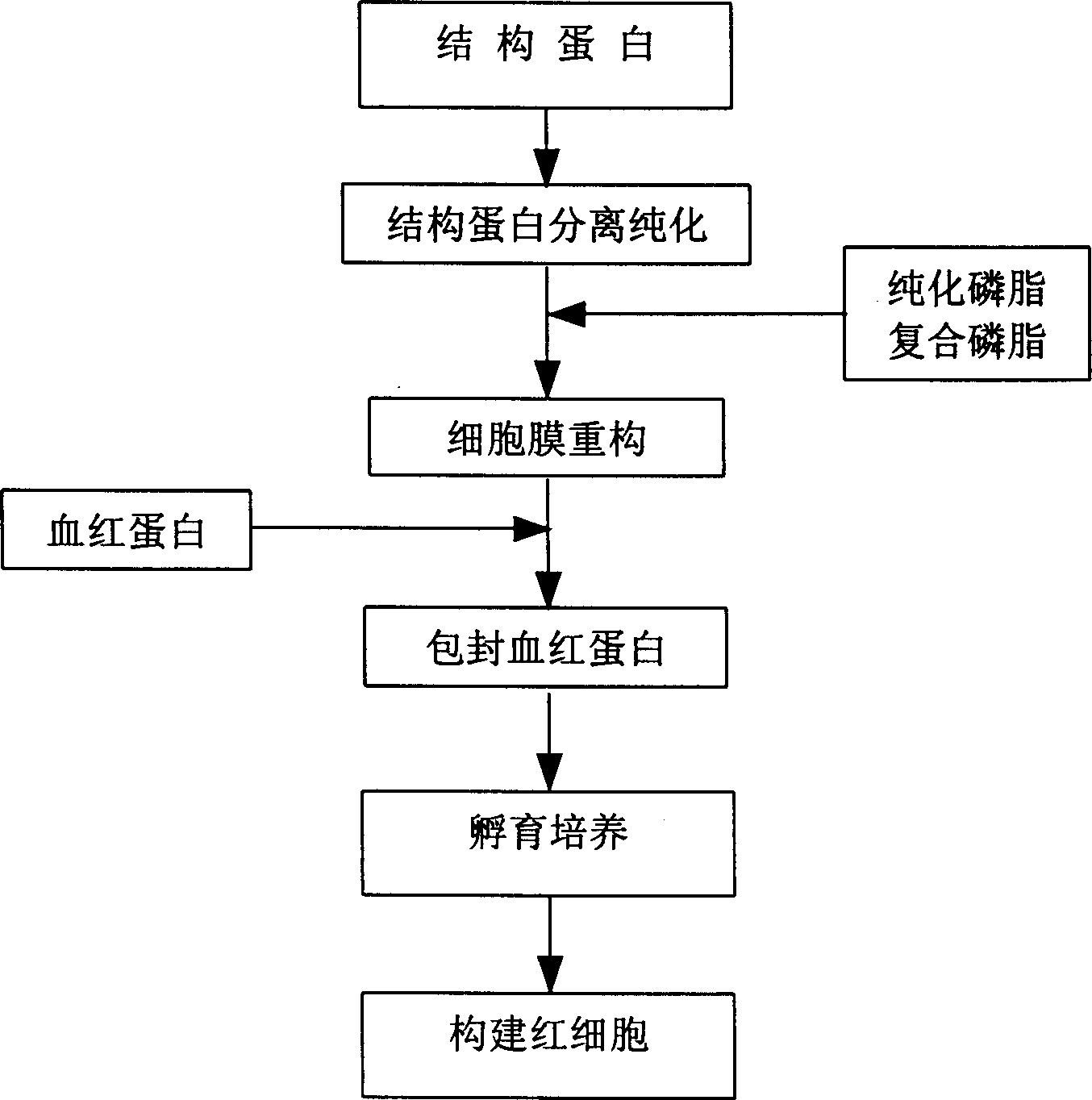
In vitro reconstituted human erythrocyte, its preparation and application in blood substitute material
A human erythrocyte and erythrocyte technology, applied in animal cells, vertebrate cells, blood/immune system cells, etc., can solve the problems of fragile membranes, incalculable demand and market, difficult storage and transportation of natural blood, etc. Effect
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 2
[0031] The structural protein crude product is obtained through dialysis, desalting, purification and freeze-drying to obtain the structural protein product. The plant structural proteins obtained by this method are globulins, usually αβ dimers, tetramers, and hexamers. In the experiment, more than 70% of the products are αβ dimer globulins through the regulation of pH and ionic strength. Example 2 Separation and Purification Method II of Structural Protein
[0032] (1), extract structural protein
[0033] Use animal blood containing anticoagulants (per 30ml of blood plus about 5ml of heparin pH7.4 isotonic phosphate buffer) In order to minimize the denaturation and enzymatic hydrolysis of structural proteins, the entire operation should be performed at a low temperature of 0-4°C conduct. Take 5ml of the above-mentioned anticoagulated blood, refrigerated and centrifuged (3000rpm, ~4°C, 15-30 minutes) to precipitate red blood cells, and separate the plasma supernatant and buf...
Embodiment 5
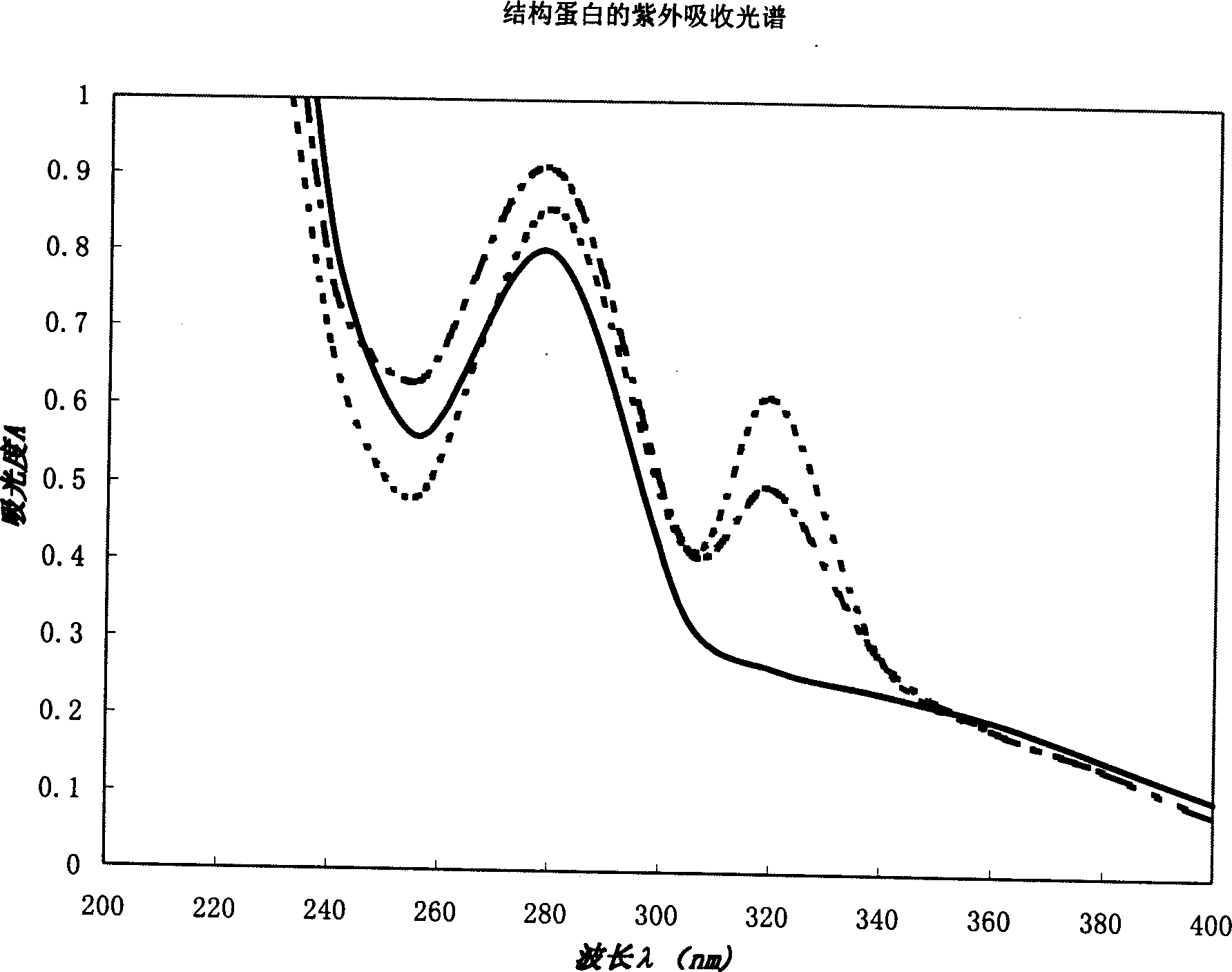
[0038] The structural protein product obtained by separation and purification is dissolved in physiological saline at a concentration of 200-800 ppm, and subjected to ultraviolet spectrophotometric analysis. Instrument: PE-λ900, scanning wavelength range: 200-400nm. For spectrogram see figure 2 . The solid lines in the figure represent the ultraviolet absorption peaks of structural proteins derived from animal blood, and the dotted lines represent the ultraviolet absorption peaks of structural proteins derived from plant proteins. The three spectral curves all show the maximum absorption peak at 280nm. Example 3 Structural Protein Gel Electrophoresis UV Scanning Spectrum Detection
Embodiment 3
[0038] The structural protein product obtained by separation and purification is dissolved in physiological saline at a concentration of 200-800 ppm, and subjected to ultraviolet spectrophotometric analysis. Instrument: PE-λ900, scanning wavelength range: 200-400nm. For spectrogram see figure 2 . The solid lines in the figure represent the ultraviolet absorption peaks of structural proteins derived from animal blood, and the dotted lines represent the ultraviolet absorption peaks of structural proteins derived from plant proteins. The three spectral curves all show the maximum absorption peak at 280nm. Example 3 Structural Protein Gel Electrophoresis UV Scanning Spectrum Detection
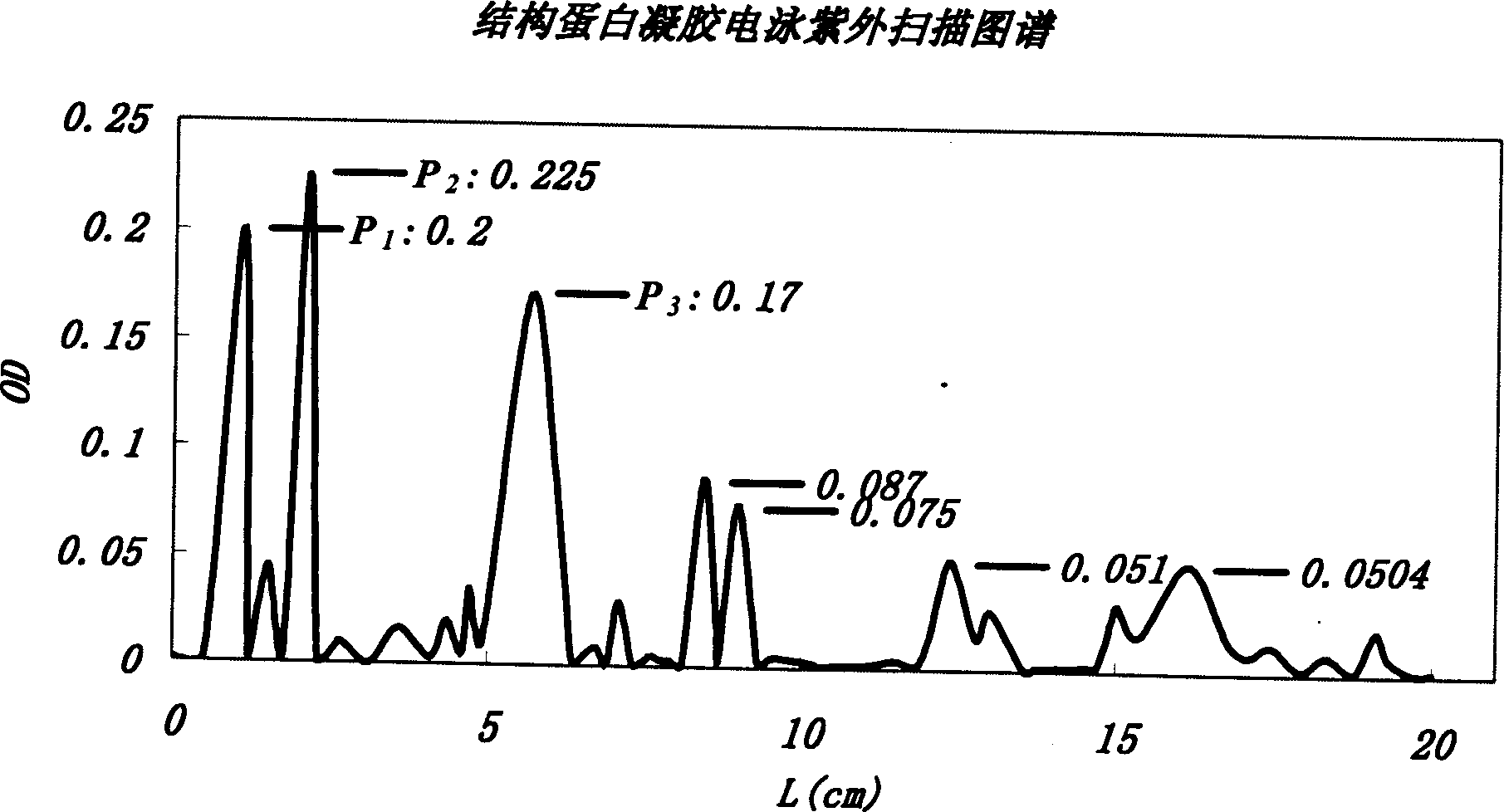
[0039] The structural protein product obtained by separation and purification is dissolved in physiological saline at a concentration of 10-200 μg / ml, and analyzed by polyacrylamide gel electrophoresis. Instrument: electrophoresis instrument BIO-RAD300; image system: IMAGE SYSTEM SX-100 scanni...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More