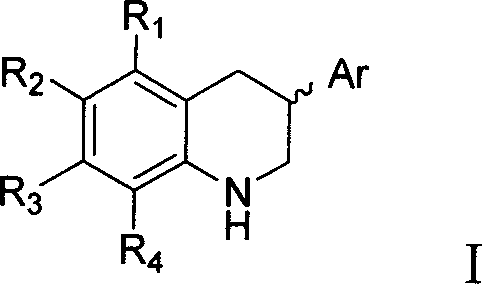
3-substituted 1,2,3.4-tetrahydro-quinazine derivative, its synthesis and use
A technology of tetrahydroquinoline and derivatives, which is applied in the field of 3-substituted 1, can solve the problems of limited synthesis conditions of quinoline derivatives, difficulty in preparing 3-aryl groups, and difficult operation, so as to avoid hydrogenation and reduction response, treatment of cerebral ischemia, effects of mild conditions
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
preparation example Construction
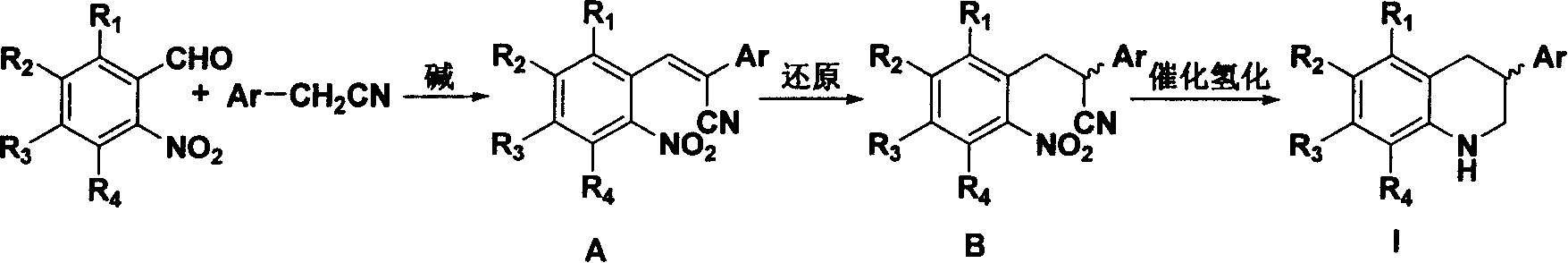
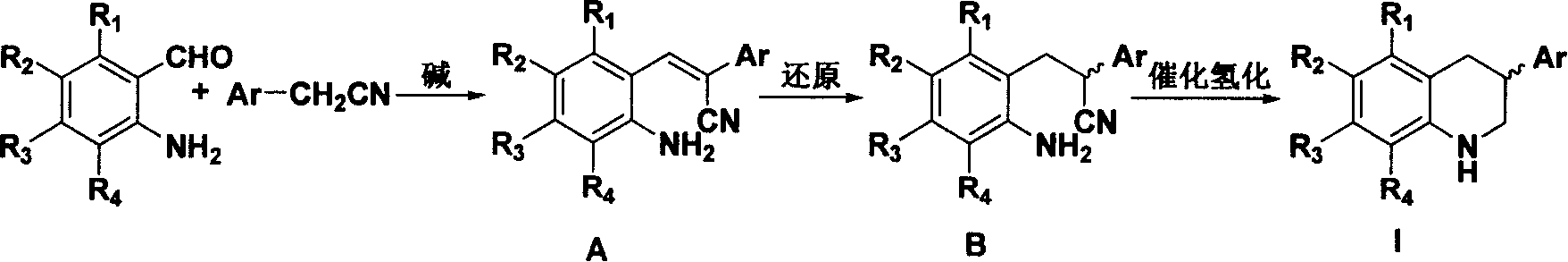
[0024] The synthesis method comprises: under alkaline conditions, substituted o-nitrobenzaldehyde or o-aminobenzaldehyde and aryl acetonitrile derivatives are condensed into an acrylonitrile compound of structural formula A; the acrylonitrile compound of structural formula A is The reduction reaction reduces to the propionitrile compound of the structural formula B; then the ring is closed under catalytic hydrogenation conditions to generate 3-substituted 1,2,3,4-tetrahydroquinoline derivatives.
[0025] Wherein, in the substituted o-nitrobenzaldehyde or o-aminobenzaldehyde and aryl acetonitrile derivatives condensed into the acrylonitrile compound of structural formula A, the base used can be an inorganic base, such as potassium carbonate; it can also be an organic base, Such as organic amines, sodium or potassium alcoholate. The amount of base is 10%-100% molar ratio of any reactant.
[0026] The acrylonitrile compound of structural formula A is reduced into the propionitri...
Embodiment 1
[0031] The preparation of the acrylonitrile compound of structural formula A
[0032] (1) Preparation of (Z)-3-(2-nitrophenyl)-2-phenylacrylonitrile (A-1)
[0033] Completely dissolve 0.23g (10mmol) of sodium in 50mL of absolute ethanol, add 15.12g (100mmol) of 2-nitrobenzaldehyde and 11.71g (100mmol) of phenylacetonitrile under stirring, stir at room temperature for 4h, filter, and wash with absolute ethanol to obtain Bright yellow solid 23.00g, yield 92.0%.
[0034] 1 HNMR (300MHz, CDCl 3 )δ7.42-7.53 (m, 3H), 7.64 (t, J=8.2, 1H), 7.73 (dd, J=1.7, 8.1, 2H), 7.80 (t, J=7.6, 1H), 7.95 (d , J=7.6, 1H), 8.06 (s, 1H), 8.26 (d, J=8.3, 1H); MS (EI): m / e (%) 250 (16M + ), 119(100), 92(84).
[0035] (2) Preparation of (Z)-2-(4-methoxyphenyl)-3-(2-nitrophenyl)-acrylonitrile (A-2)
[0036] 2-nitrobenzaldehyde and 4-methoxyphenylacetonitrile were carried out according to the method described in Example 1.(1) to obtain compound A-2 with a yield of 74.2%.
[0037] 1 HNMR (300MHz, C...
Embodiment 2
[0096] The preparation of the propionitrile compound of structural formula B
[0097] (1) Preparation of 3-(2-nitrophenyl)-2-phenylpropionitrile (B-1)
[0098] Dissolve 5.00g (20mmol) (Z)-3-(2-nitrophenyl)-2-phenyl-acrylonitrile in 20mL of anhydrous tetrahydrofuran, and add 1.13g (30mmol) sodium borohydride in batches under an ice-water bath , stirred at room temperature; after TLC monitoring of no raw materials, quenched with 1mol / L HCl, concentrated under reduced pressure to remove tetrahydrofuran, then extracted with ethyl acetate, washed with saturated sodium chloride solution, dried the organic phase with anhydrous magnesium sulfate, filtered, and concentrated under reduced pressure to obtain yellow Solid 4.80g, yield 92.0%. The next reaction was carried out directly without purification.
[0099] 1 HNMR (300MHz, CDCl 3 )δ 3.22 (dd, J=10.3, 13.3, 1H), 3.54 (dd, J=5.2, 13.1, 1H), 4.39 (dd, J=5.1, 10.8, 1H), 7.32-7.53 (m, 7H) , 7.60-7.66 (m, 1H), 8.08 (d, J=8.1, 1H); M...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More