Tissue products derived from animals lacking any expression of functional alpha 1,3 galactosyltransferase
A galactosyl and transferase technology, which can be used in bone implants, skin transplantation, medical science, etc., and can solve problems such as allele effects and phenotype changes.
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
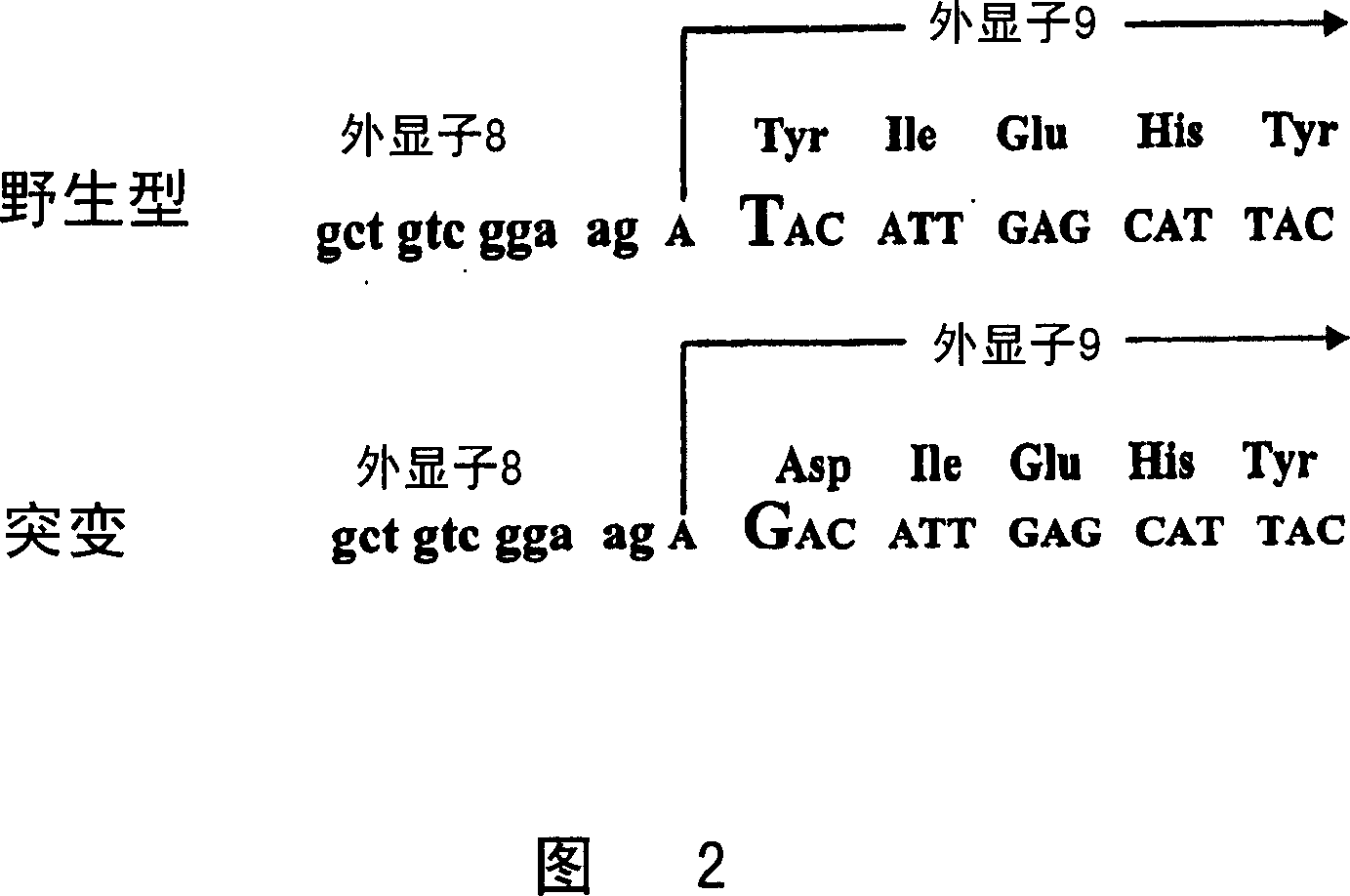
[0284] Generating pig cells that are heterozygous for the α-1,3-GT gene
[0285] Isolation and Transfection of Primary Pig Fetal Fibroblasts
[0286] Fetal fibroblasts (PCFF4-1 to PCFF4-10) were isolated from 10 fetuses of the same pregnancy on the 33rd day of pregnancy. After removing the head and internal organs, the fetus was washed with Hanks balanced salt solution (HBSS; Gibco-BRL, Rockville, MD), placed in 20ml HBSS, and cut into pieces with small surgical scissors. The tissue was pelleted and resuspended in a 50ml test tube containing 40ml DMEM and 100 U / ml collagenase per fetus (Gibco-BRL). The tube was incubated in a shaking water bath at 37°C for 40 minutes. The digested tissue is allowed to settle for 3-4 minutes, and the cell-rich supernatant is transferred to a new 50 ml test tube and pelleted. The cells were then resuspended in 40 ml DMEM containing 10% fetal calf serum (FCS), 1X non-essential amino acids, 1 mM sodium pyruvate and 2ng / ml bFGF, and inoculated into a 1...
Embodiment 2
[0298] Preparation of pig cells heterozygous for α-1,3-GT gene
[0299] Heterozygous α-1,3-GT knockout fetal fibroblasts (657A-I11 1-6) cells were isolated from the fetus on the 32nd day of pregnancy as described above (see also Dai et al. Nature Biotechnology 20: 451 (2002)). After removing the head and internal organs, some fetuses were washed with Hanks balanced salt solution (HBSS; Gibco-BRL, Rockville, MD), placed in 20ml HBSS, and cut into pieces with small surgical scissors. The tissue is pelleted and resuspended in a 50ml test tube containing 40ml DMEM and 100U / ml collagenase (Gibco-BRL) per fetus. The test tube was incubated in a shaking water bath at 37°C for 40 minutes. The digested tissue is allowed to settle for 3-4 minutes, and the cell-rich supernatant is transferred to a new 50 ml test tube and pelleted. Then resuspend the cells in 40ml containing 10% fetal calf serum (FCS), 1X non-essential amino acids, 1mM sodium pyruvate (Gibco-BRL) and 2ng / ml basic fibroblast g...
Embodiment 3
[0301] Selection of pig cells homozygous for α-1,3-GT gene with Clostridium difficile toxin A
[0302] Toxin A cytotoxicity curve
[0303] Porcine cells (PCFF4-6) were exposed to a 10-fold serial dilution of toxin A (0.00001μg / ml-10μg / ml) for 1 hour or overnight. Culture the cells in a 24-well plate and incubate with toxin at 37°C for 1 hour or overnight. The results of this exposure are detailed in Table 2. Obviously, exposure to 1 μg / ml toxin A for 1 hour resulted in a cytotoxic effect on >90% of cells. Therefore, a toxin A concentration of 1 μg / ml or slightly higher than 1 μg / ml is selected to select genetically altered cells.
[0304] Table 2. Toxin A toxicity after 1 hour and overnight exposure
[0305] [Toxin A], μg / ml
Incubate for 1 hour
Incubate overnight
0
100% full
100% full
.00001
100% full
100% full
.0001
100% full
100% full
.001
100% full
100% full
.01
100% full...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Diameter | aaaaa | aaaaa |
| Length | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More