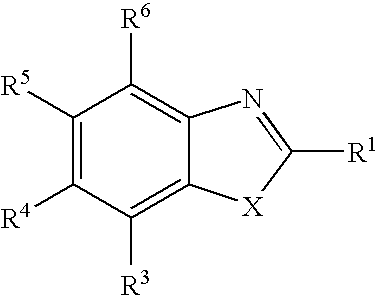
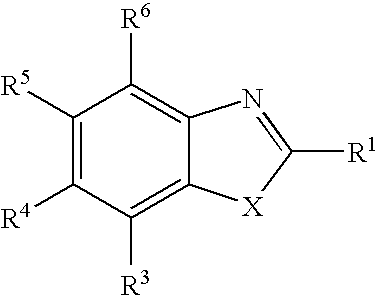
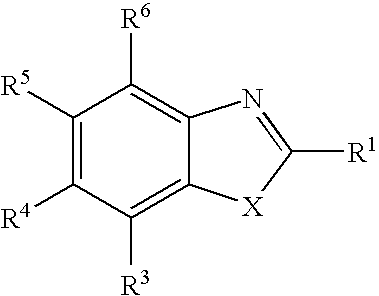
Therapeutic benzothiazole compounds
a technology of benzothiazole and compound, which is applied in the field of ligands, can solve problems such as limiting its us
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
4-Chloro-6-hydroxy-2-(4-hydroxyphenyl)benzoxazole
1) Synthetic method A: Synthesis of 2-amino-3-chloro-5-methoxyphenol
[0098] A solution of 3-chloro-5-methoxy-2-nitrophenol [1] (450 mg) and tin(II) chloride dihydrate (2 g, 4 eq.) in ethyl acetate (30 mL) was heated under reflux for 4 h. The mixture was cooled, diluted with ethyl acetate and aqueous potassium fluoride. The mixture was filtered through celite. The organic layer was washed with brine and dried over MgSO4. Evaporation of the solvent afforded the title compound (280 mg) as a pale solid. NMR (DMSO-d6): 9.74 (m, 1H), 6.33 (d, 1H, J=2.4 Hz), 6.30 (d, 1H, J=2.4 Hz), 4.19 (m, 2H), 3.60 (s, 3H).
[0099] Reference 1: Hodgson, Wignall, J. Chem. Soc., 1928, 330. Prepared from 1-chloro-3,5-dimethoxybenzene by nitration with concentrated nitric acid in acetic anhydride below 10° C. to give 1-chloro-3,5-dimethoxy-2-nitrobenzene, and subsequent reaction with boron tribromide (1 eq.) in dichloromethane from −78° C. to 0° C.
2) Synthe...
example 2
[0100] A solution of 2-amino-3-chloro-5-methoxyphenol (270 mg) and ethyl 4-hydroxybenzimidate hydrochloride (376 mg, 1.2 eq.) in absolute ethanol (5 mL) was heated under reflux for 4 h. The mixture was cooled, partitioned between ethyl acetate and water. The organic layer was dried over MgSO4. After evaporation of the solvent, the residue was triturated with methanol to give the title compound (130 mg) as a light orange solid. NMR (DMSO-d6): 10.35 (s, 1H), 8.00 (d, 2H, J=8.7 Hz), 7.39 (d, 1H, J=2.1 Hz), 7.10 (d, 1H, J=2.1 Hz), 6.96 (d, 2H, J=8.7 Hz), 3.85 (s, 3H); MS: 276 (MH+).
3) Synthetic method C: Synthesis of 4-chloro-6-hydroxy-2-(4-hydroxyphenyl)benzoxazole
[0101] To a suspension of 4-chloro-2-(4-hydroxyphenyl)-6-methoxybenzoxazole (240 mg) in dichloromethane (5 mL) cooled at −78° C. was added boron tribromide (5 mL, 1M solution in dichloromethane, 5.7 eq.). The mixture was stirred at −78° C. for 10 min, warmed to room temperature and stirred for 3 h. The mixture was poured o...
example 3
6-Hydroxy-2-(4-hydroxyphenyl)-4-methylbenzoxazole
1) Synthesis of 2-(4-hydroxyphenyl)-6-methoxy-4-methylbenzoxazole (Example 4)
According to synthetic method B, from 2-amino-5-methoxy-3-methylphenol [2] (440 mg) was obtained the title compound (340 mg) as a light orange solid. MS: 256 (MH+).
[0102] Reference 2: Musso H; Beecken H, Chem. Ber. 1961, 94, 585; made from 3,5-dimethoxytoluene by nitration and monodeprotection of the 3-methoxy with BBr3 similarly to Ref. 1 followed by reduction of the nitro group to the amino by hydrogenation with palladium on charcoal.
[0103] 2) According to synthetic method C, the above compound (220 mg) was converted to 6-hydroxy-2-(4-hydroxyphenyl)-4-methylbenzoxazole (112 mg) as a light pinkish powder. NMR (DMSO-d6): 10.14 (s, 1H), 9.58 (s, 1H), 7.94 (d, 2H, J=8.7 Hz), 6.92 (d, 2H, J=8.7 Hz), 6.84 (s, 1H), 6.63 (s, 1H), 2.46 (s, 3H); MS: 242 (MH+).
PUM
| Property | Measurement | Unit |
|---|---|---|
| pH | aaaaa | aaaaa |
| volume | aaaaa | aaaaa |
| concentration | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More