Schisandrin B preparation
a technology of schisandrine and preparation, which is applied in the field of preparations comprising schisandrine b, can solve problems such as muscle damage, and achieve the effect of enhancing sports activities
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
Extraction and Purification of (−)Schisandrin B (Sch B)
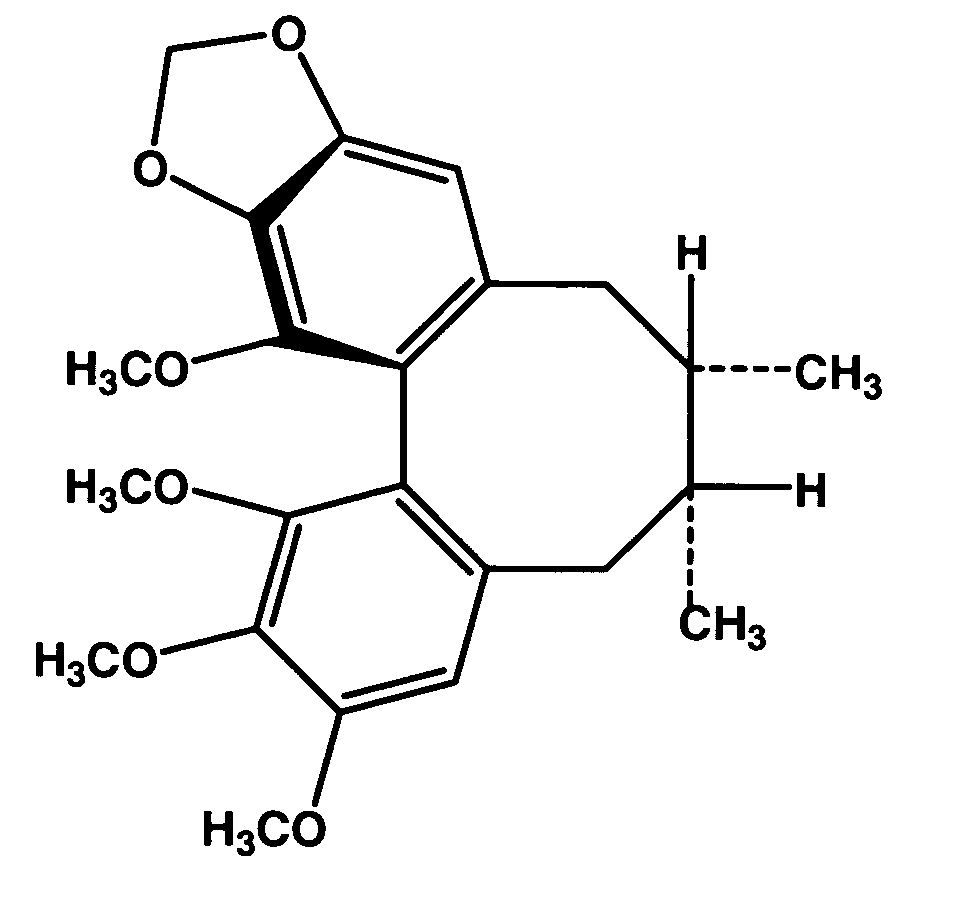
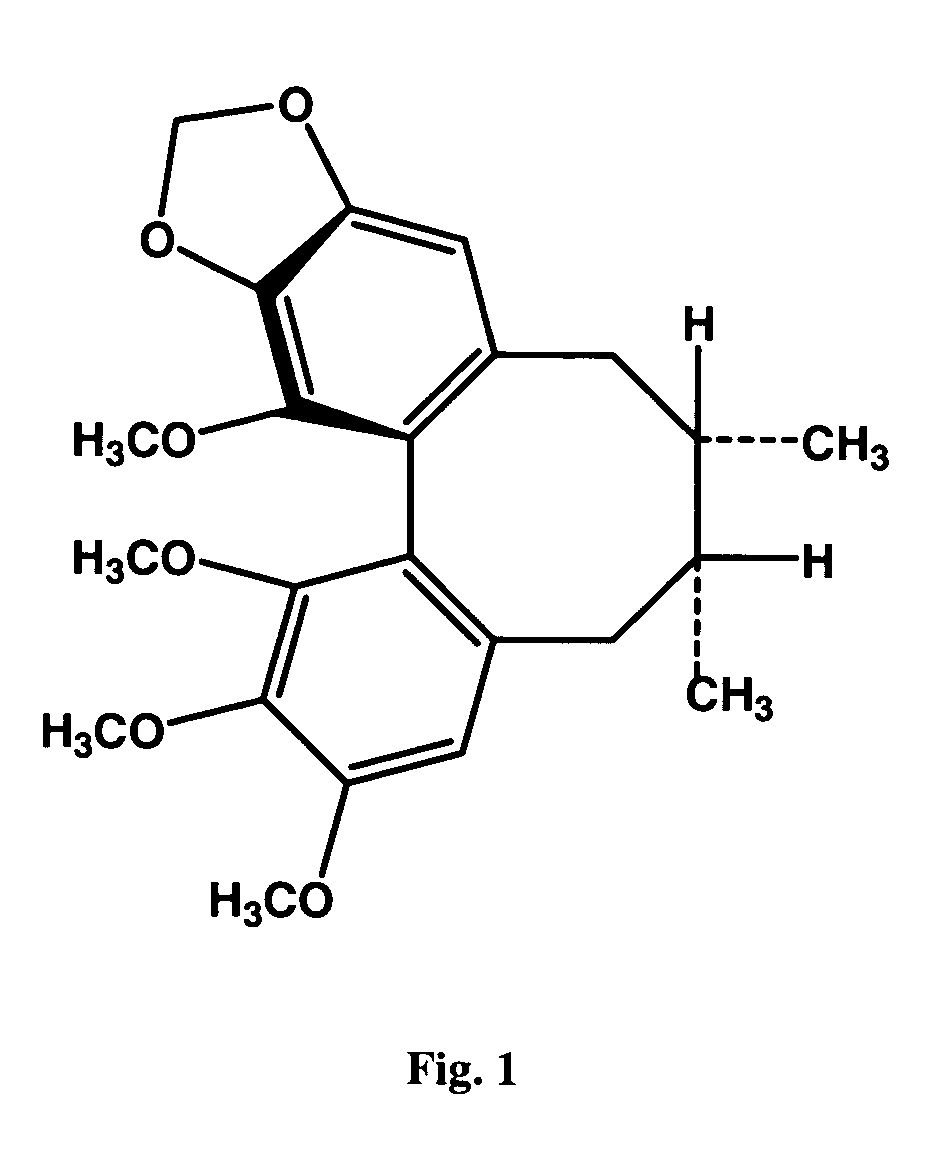
[0060] Dried powder of the Schisandra fruits (Schisandra chinensis (Turcz) Baillon) was extracted with petroleum ether. The petroleum ether extract was then subjected to silica gel column chromatography using isocratic elution with acetone:petroleum ether (5:95, v / v). The Schisandrin B-containing fractions, as detected by thin layer chromatography, were pooled, and a crude crystalline fraction was obtained. The crystalline fraction was further purified by preparative reverse-phase HPLC using Prep Nova-Pak HR C18 (19×300 mm) column eluted by methanol : H2O (75:25, v / v). Purified (−) Schisandrin B and (+) Schisandrin B were obtained, and the chirality of (−) Schisandrin B and (+) Schisandrin B (FIG. 1) was confirmed by optical rotation measurement, with the specific rotation measured in methanol at 20° C. being −47.2° and +55.3°, respectively. HPLC analysis using chiral column (CHIRACEL OD-H, 4.6×250 mm, Diacel Chemical Industri...
example 2
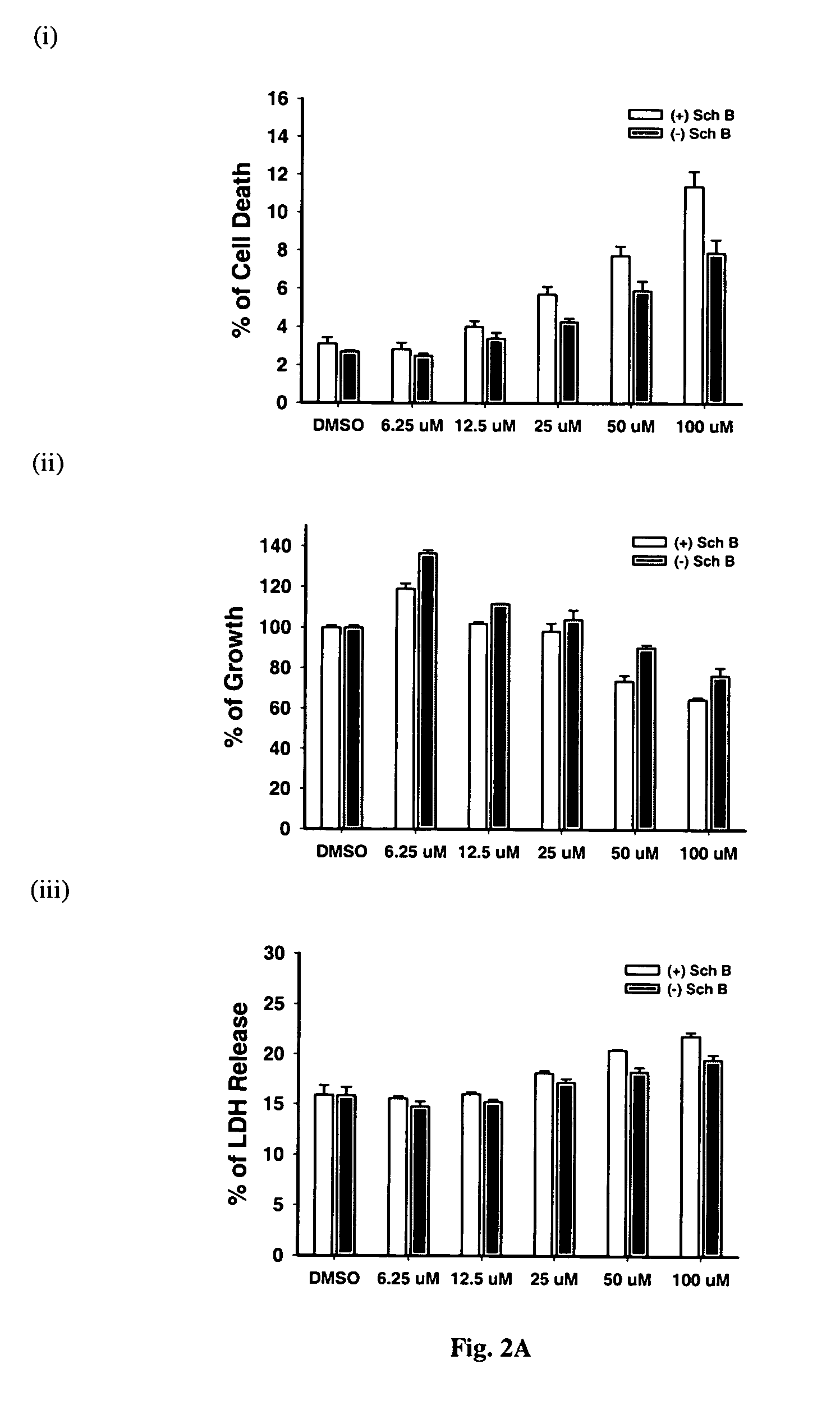
Time-Course of (−) Schisandrin B-Induced Increase In Cellular GSH Level in H9c2 Cells: Protection Against Oxidative Challenge by Xanthine / Xanthine Oxidase (X / XO)
Methods
Cell Culture
[0063] H9c2 cells, a permanent cell line derived from cardiac myoblasts of rat embryo, have early been characterized as a suitable model of myocardial cells [Hescheler et al. 1991). H9c2 cells were cultured as monolayers in Dulbecco's modified Eagle's medium (GIBCO BRL) supplemented with 10% (v / v) fetal bovine serum. The medium contained glucose (4.5 g / L) and glutamine (4.5 mM), supplemented with NaHCO3 (17 mM), penicillin (100 IU / ml), and streptomycin (100 μg / ml). Cells were grown under an atmosphere of 5% CO2 in air at 37° C. The medium was replaced by fresh medium every 2 or 3 days. A stock of cells was grown in a 75 cm culture flask and split before confluence at a subcultivation ratio of 1:10. Cells used for experiments were seeded at a density of 3.75×104 cells / well on a 12-well culture plate,...
example 3
Cytoprotective Effect of (−) Schisandrin B on Menadione-Induced Toxicity in H9c2
Methods
Cell Culture
[0070] H9c2 cells, a permanent cell line derived from cardiac myoblasts of rat embryo, have early been characterized as a suitable model of myocardial cells [Hescheler et al. 1991]. H9c2 cells were cultured as monolayers in Dulbecco's modified Eagle's medium (GIBCO BRL) supplemented with 10% (v / v) fetal bovine serum. The medium contained glucose (4.5 g / L) and glutamine (4.5 mM), supplemented with NaHCO3 (17 mM), penicillin (100 IU / ml), and streptomycin (100 μg / ml). Cells were grown under an atmosphere of 5% CO2 in air at 37° C. The medium was replaced by fresh medium every 2 or 3 days. A stock of cells was grown in a 75 cm culture flask and split before confluence at a subcultivation ratio of 1:10. Cells used for experiments were seeded at a density of 3.75×104 cells / well on a 12-well culture plate, and the cells were grown for 24 h to about 80% confluence prior to drug treatmen...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Fraction | aaaaa | aaaaa |
| Fraction | aaaaa | aaaaa |
| Fraction | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More