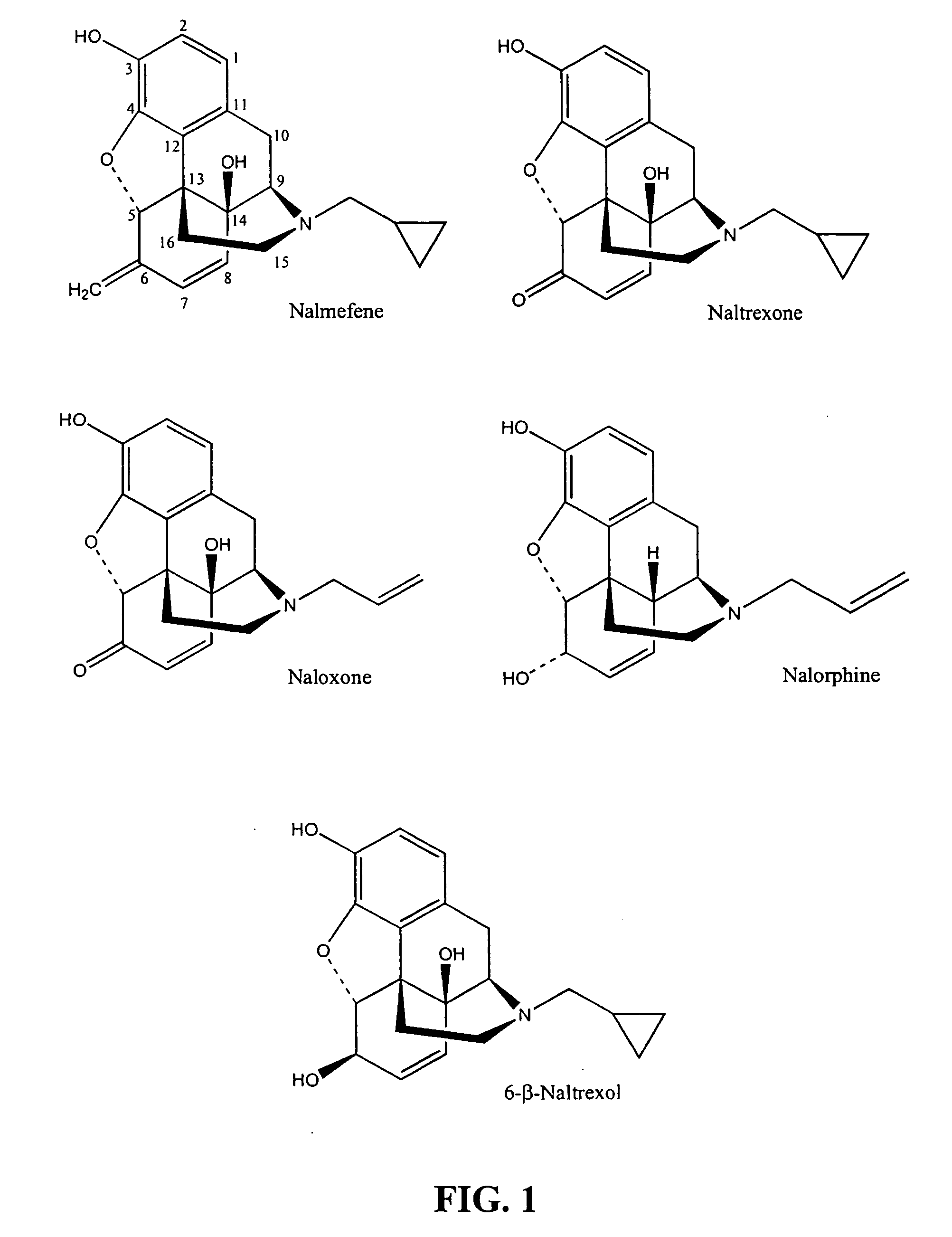
Inhibitors of ABC drug transporters at the blood-brain barrier
a technology of abc drug and bloodbrain barrier, which is applied in the direction of heterocyclic compound active ingredients, biocide, animal husbandry, etc., can solve the problems of preventing the development of therapies for a wide variety of disorders, delivering these pharmaceutical substances to their active sites in the central nervous system, and affecting the development of therapies. , to achieve the effect of enhancing the efficacy of a non-opioid cns-active agent, reducing the efficacy rate rate ra
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
Opioid Receptor Antagonists Inhibit Human PGP-Mediated Transport
[0067] Porcine kidney-derived, LLC-PK1, cells expressing human PGP cDNA (designated 15B-J) were cultured in 24 well Transwell™ culture inserts at 37° C. on an orbital shaker. Transport assays were conducted in 24 well Transwell™ culture inserts with Hanks Balanced Salt Solution (HBSS) buffered with the addition of 10 mM HEPES (pH 7.2).
[0068] The test substances, naloxone, naltrexone and nalmefene, were purchased from Sigma-Aldrich. Stock solutions of the compounds were made in DMSO, and dilutions of these in transport buffer were prepared for assay in the monolayers. The DMSO concentration (0.55%) was constant for all conditions within the experiment. All test substance and control drug solutions prepared in HBSS / HEPES buffer contained 0.55% DMSO.
[0069] The test substance was added to the donor and receiver chambers. Duplicate monolayers and thirteen test substance concentrations of 0.0001, 0.0003, 0.001, 0.003, 0.01...
example 2
6-β-Naltrexol Does Not Inhibit Human PGP-Mediated Transport
[0074] Porcine kidney-derived, LLC-PK1, cells expressing human PGP cDNA (designated 15B-J) were cultured in 24 well Transwell™ culture inserts at 37° C. on an orbital shaker. Transport assays were conducted in 24 well Transwell™ culture inserts with Hanks Balanced Salt Solution (HBSS) buffered with the addition of 10 mM HEPES (pH 7.2).
[0075] The test substance, 6-β-naltrexol, was provided by LC Resources, Inc. Stock solutions of the compounds were made in DMSO, and dilutions of these in transport buffer were prepared for assay in the monolayers. The DMSO concentration (0.55%) was constant for all conditions within the experiment. All test substance and control drug solutions prepared in HBSS / HEPES buffer contained 0.55% DMSO.
[0076] The test substance was added to the donor and receiver chambers. Duplicate monolayers and thirteen test substance concentrations of 0.0001, 0.0003, 0.001, 0.003, 0.01, 0.03, 0.1, 0.3, 1, 3, 10,...
example 3
Opioid Receptor Antagonists Inhibit PGP ATPase Activity
[0080] The test substances, naloxone, naltrexone and nalmefene, were purchased from Sigma-Aldrich. Stock solutions of the compounds were made in DMSO, and dilutions of these in transport buffer were prepared for assay in the monolayers. The DMSO concentration (0.55%) was constant for all conditions within the experiment. All test substance and control drug solutions prepared in HBSS / HEPES buffer contained 0.55% DMSO.
[0081] The test substances were incubated in the membranes and supplemented with MgATP, with and without sodium orthovanadate present. Orthovanadate inhibits PGP by trapping MgADP in the nucleotide binding site. Thus, the ATPase activity measured in the presence of orthovanadate represents non-PGP ATPase activity and was subtracted from the activity generated without orthovanadate to yield vanadate-sensitive ATPase activity.
[0082] ATPase assays were conducted in 96-well microtiter plates. A 0.06 ml reaction mixtur...
PUM
| Property | Measurement | Unit |
|---|---|---|
| pH | aaaaa | aaaaa |
| voltage | aaaaa | aaaaa |
| flow rate | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More