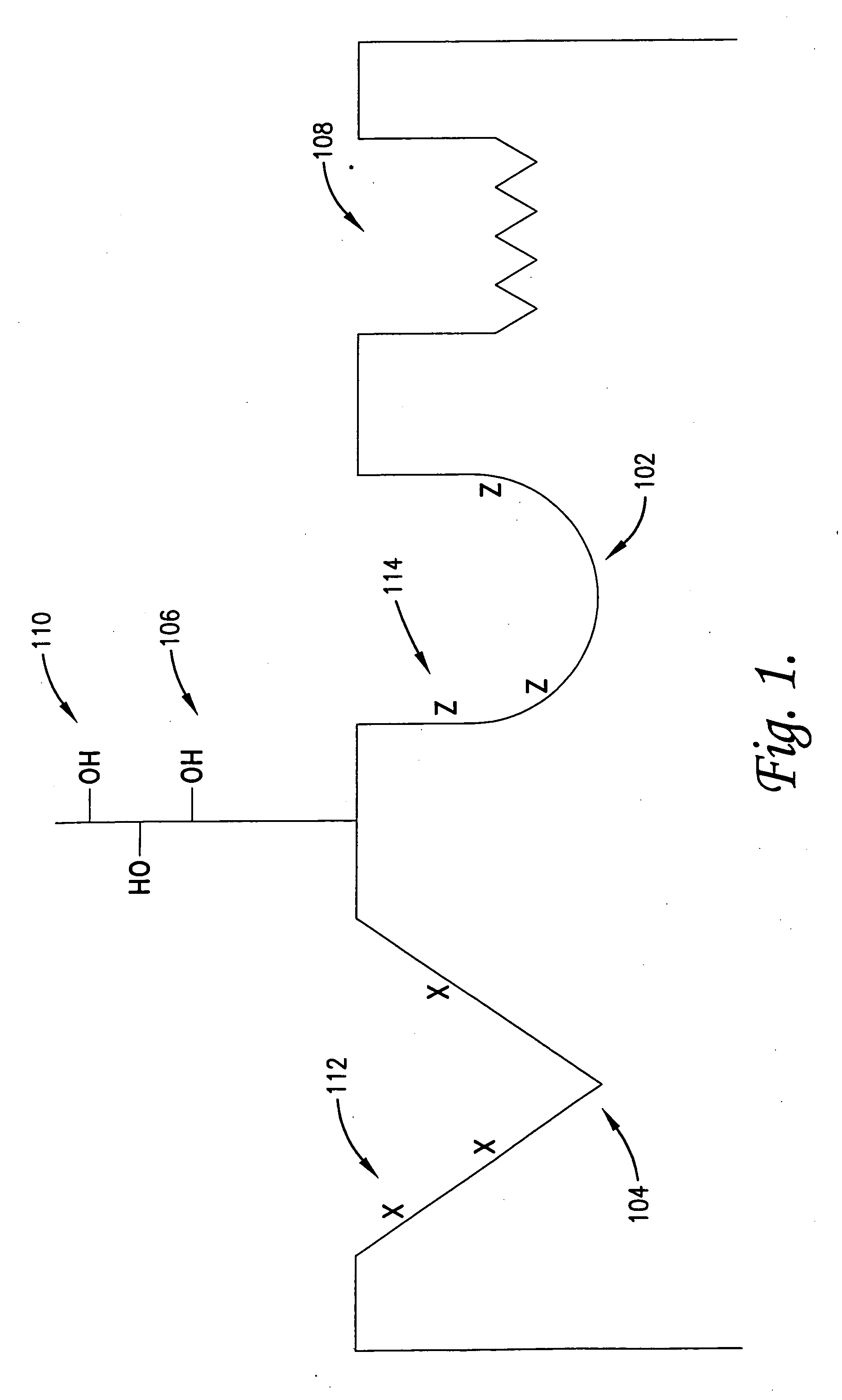
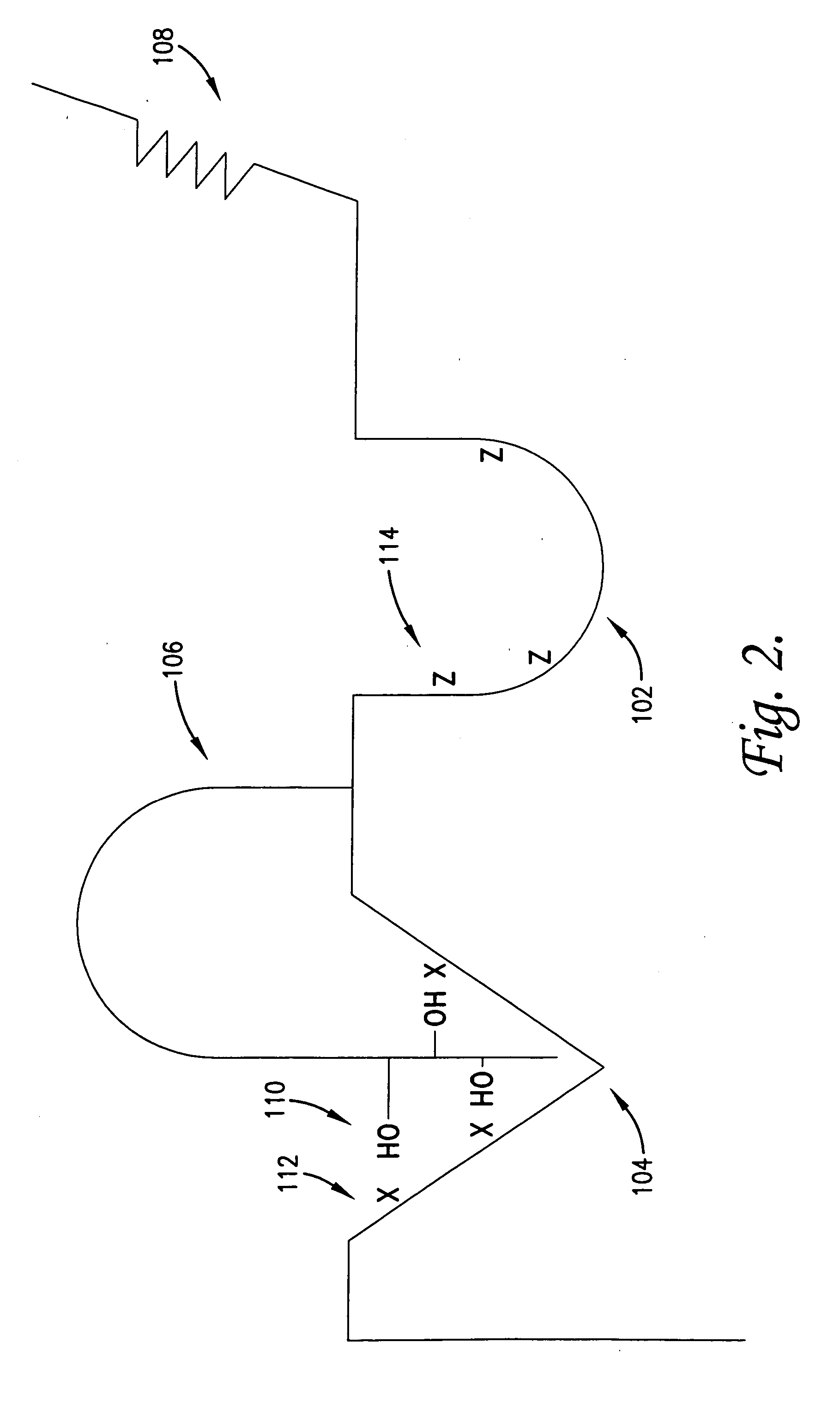
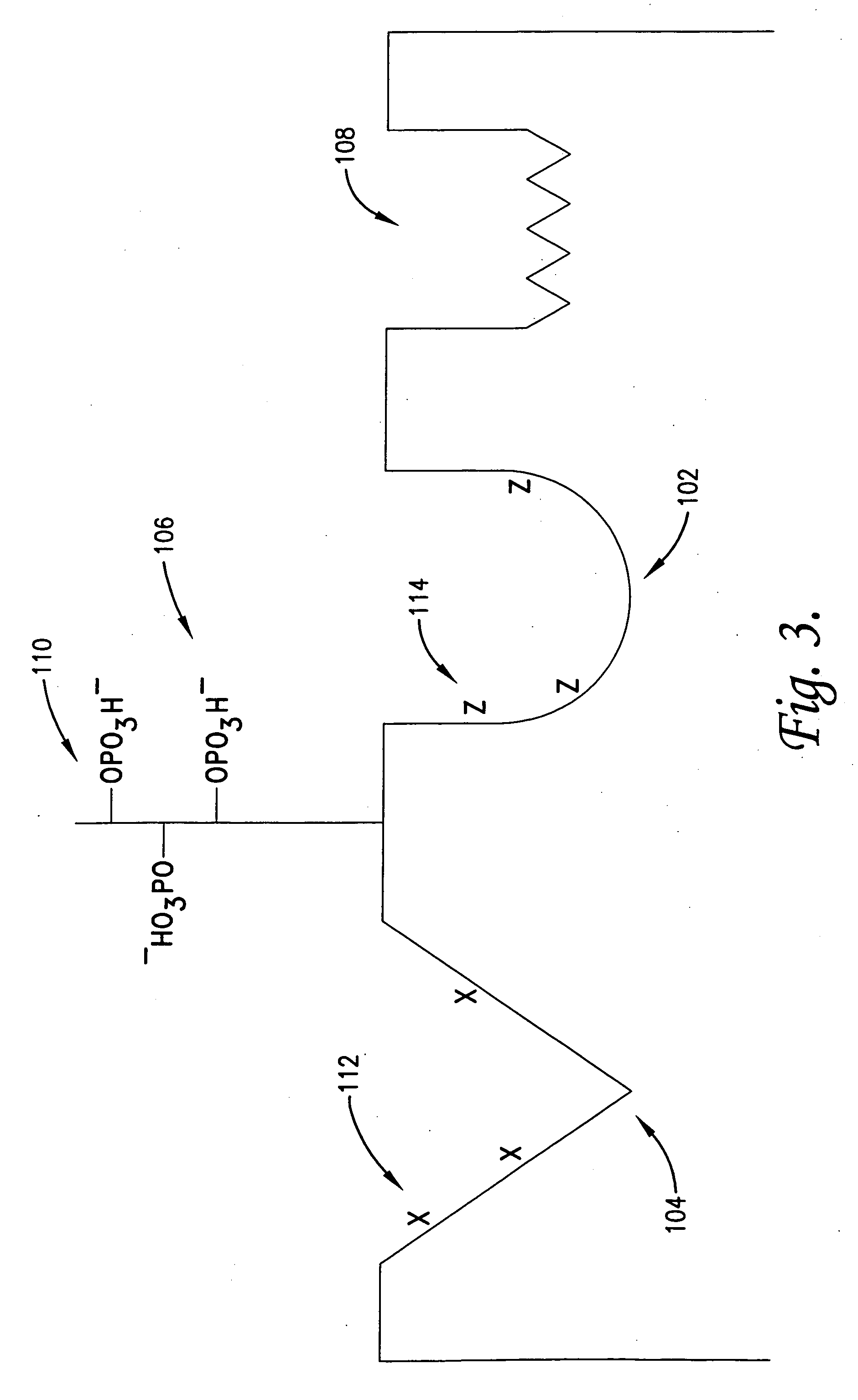
Anti-inflammatory medicaments
a technology of anti-inflammatory and anti-inflammatory drugs, which is applied in the field of anti-inflammatory medicaments, can solve the problems of lack of selectivity, insufficient therapeutic windows to achieve maximum efficacy, and the method and strategy by which the pharmaceutical industry sets about to develop small molecule therapeutics has not significantly advanced, so as to achieve greater propensity, lesser propensity to interact, and greater propensity
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
examples
[0240] The following examples set forth preferred methods in accordance with the invention. It is to be understood, however, that these examples are provided by way of illustration and nothing therein should be taken as a limitation upon the overall scope of the invention.
[0241] [Boc-sulfamide] aminoester (Reagent AA), 1,5,7,-trimethyl-2,4-dioxo-3-aza-bicyclo[3.3.1]nonane-7-carboxylic acid (Reagent BB), and Kemp acid anhydride (Reagent CC) was prepared according to literature procedures. See Askew et. al J. Am. Chem. Soc. 1989, 111, 1082 for further details.
example a
[0242]
[0243] To a solution (200 mL) of m-amino benzoic acid (200 g, 1.46 mol) in concentrated HCl was added an aqueous solution (250 mL) of NaNO2 (102 g, 1.46 mol) at 0° C. The reaction mixture was stirred for 1 h and a solution of SnCl2.2H2O (662 g, 2.92 mol) in concentrated HCl (2 L) was then added at 0° C., and the reaction stirred for an additional 2h at RT. The precipitate was filtered and washed with ethanol and ether to yield 3-hydrazino-benzoic acid hydrochloride as a white solid.
[0244] The crude material from the previous reaction (200 g, 1.06 mol) and 4,4-dimethyl-3-oxo-pentanenitrile (146 g, 1.167 mol) in ethanol (2 L) were heated to reflux overnight. The reaction solution was evaporated in vacuo and the residue purified by column chromatography to yield ethyl 3-(3-tert-butyl-5-amino-1H-pyrazol-1-yl)benzoate (Example A, 116 g, 40%) as a white solid together with 3-(5-amino-3-tert-butyl-1H-pyrazol-1-yl)benzoic acid (93 g, 36%). 1H NMR (DMSO-d6): 8.09 (s, 1H), 8.05 (brd, J...
example b
[0245]
[0246] To a solution of 1-naphthyl isocyanate (9.42 g, 55.7 mmol) and pyridine (44 mL) in THF (100 mL) was added a solution of Example A (8.0 g, 27.9 mmol) in THY (200 mL) at 0° C. The mixture was stirred at RT for 1 h, heated until all solids were dissolved, stirred at RT for an additional 3 h and quenched with H2O (200 mL). The precipitate was filtered, washed with dilute HCl and H2O, and dried in vacuo to yield ethyl 3-[3-t-butyl-5-(3-naphthalen-1-yl)ureido)-1H-pyrazol-1-yl]benzoate (12.0 g, 95%) as a white power. 1H NMR (DMSO-d6): 9.00 (s, 1H), 8.83 (s, 1H), 8.25 7.42 (m, 11H), 6.42 (s, 1H), 4.30 (q, J=7.2 Hz, 2H), 1.26 (s, 9H), 1.06 (t, J=7.2 Hz, 3H); MS (ESI) m / z: 457.10 (M+H+).
PUM
| Property | Measurement | Unit |
|---|---|---|
| volume | aaaaa | aaaaa |
| temperature | aaaaa | aaaaa |
| volume | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More