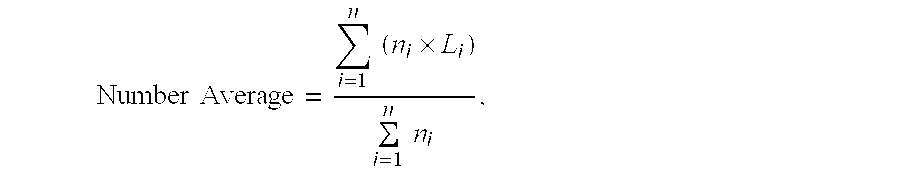Shaped binderless titanosilicate zeolite ts-1
a technology of zeolite and zeolite, which is applied in the field of process for producing crystalline titanosilicate zeolite ts1, can solve the problems of increasing the complexity of catalyst manufacturing involving zeolitic materials, affecting the catalytic performance of zeolite so bound and formed, and reducing the number of process steps.
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
example 1
Preparation of Titanosilicate Zeolite TS-1 Using Ti(butoxide)4
[0060]A reaction mixture was prepared by placing 100 grams of Ultrasil® VN3SP silica in a Baker-Perkins mixer. Sixty-five grams of 40 wt. % tetrapropylammonium hydroxide (TPAOH) was added to the mixer and mixed with the silica for 30 minutes. Eight grams of Ti(butoxide)4 was dissolved in 20 grams of isopropyl alcohol and added to the mixer, followed by 35 grams of water. The mixture was then mixed until a uniform paste was obtained. It is important that the Ti(butoxide)4 be thoroughly dispersed throughout the paste. No binder is added to the paste.
[0061]The paste was dried to an extrudable consistency and extruded with a Carver press using a 1 / 12 inch die. Half of the extrudate was dried to 47% volatiles, and the other half was dried to 43% volatiles.
[0062]Both batches were crystallized in an autoclave at autogenous pressure at 150° C. for 24 hours. The resulting products were binderless extrudates containing essentially...
example 2
Preparation of Titanosilicate Zeolite TS-1 Using Ti(butoxide)4
[0063]Titanosilicate zeolite TS-1 was prepared by the procedure of Example 1, except that the Ti(butoxide)4 was mixed with the TPAOH (without isopropyl alcohol) prior to addition to the mixer. The resulting product was binderless extrudates containing essentially 100% titanosilicate zeolite TS-1 as determined by X-ray diffraction analysis and infra-red spectroscopy (as described in aforementioned U.S. Pat. No. 4,410,501). The extrudates contain crystals of TS-1 having a crystallite size of less than about 0.4 micron, as determined by Scanning Electron Microscopy (“SEM”).
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More