Vaccine for preventing pneumococus infection
A Streptococcus pneumoniae and vaccine technology, applied in antibacterial drugs, immunoglobulins, bacterial antigen components, etc., can solve the problems of difficult treatment of pneumococcal disease and the need for enhanced vaccination
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
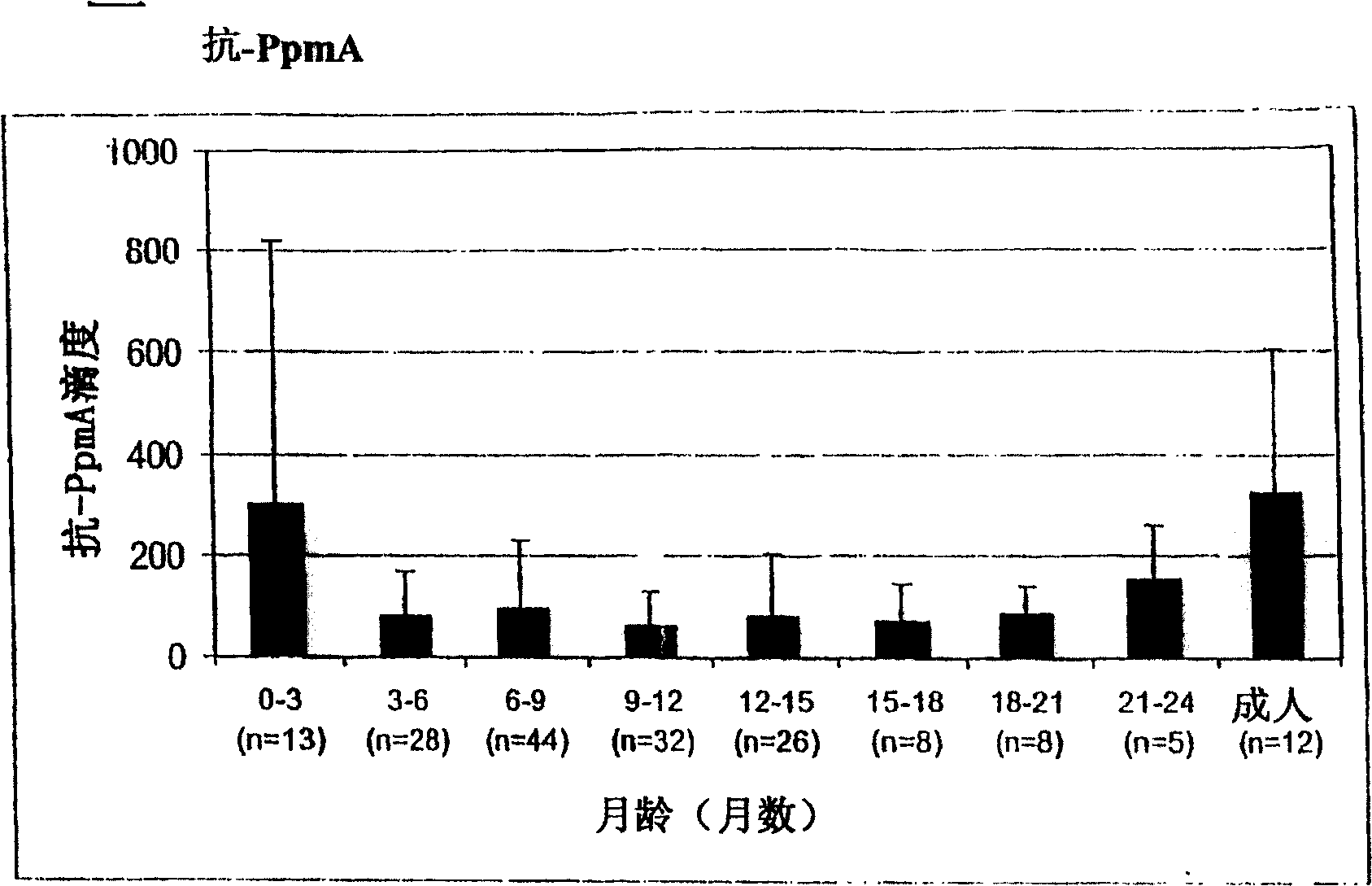
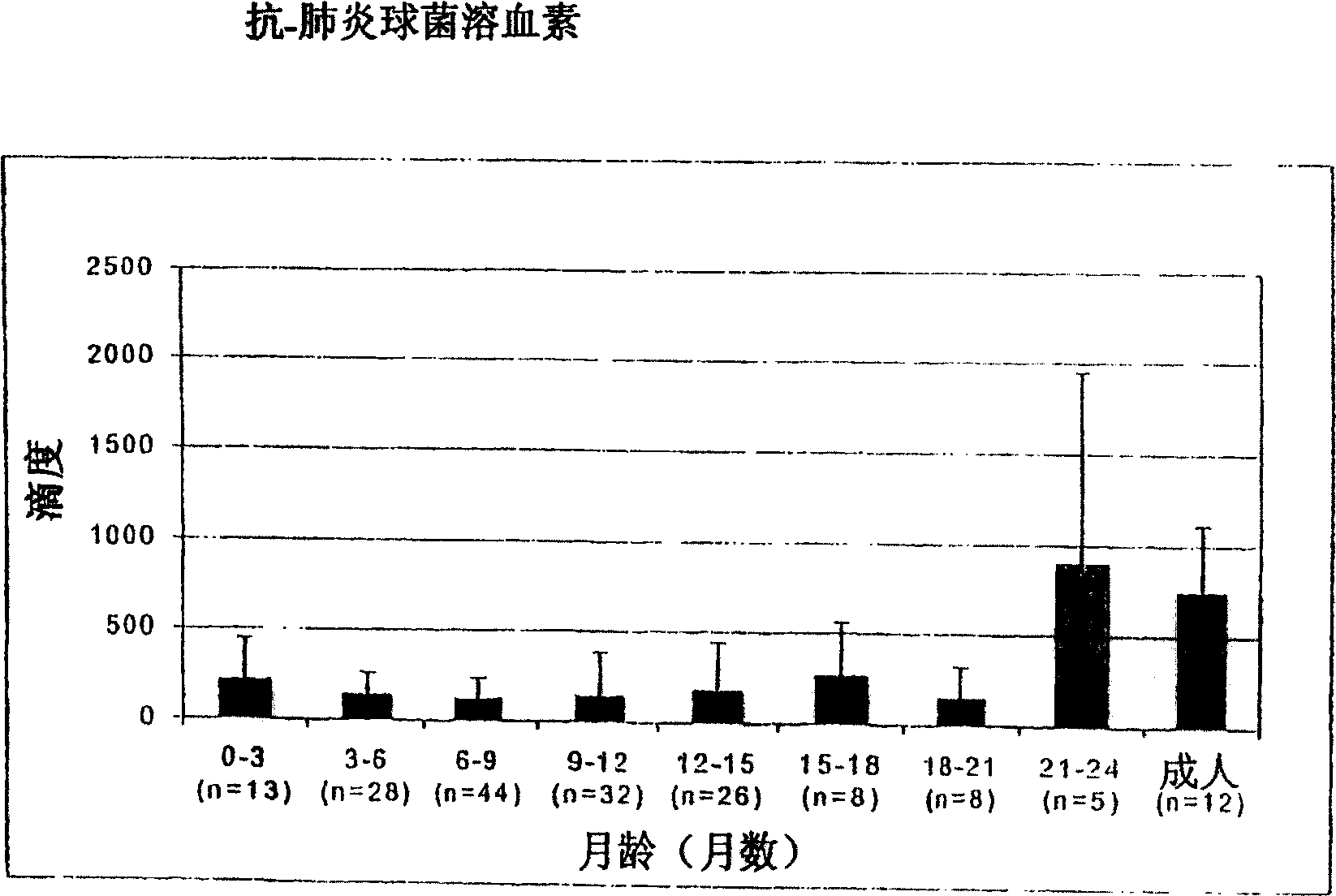
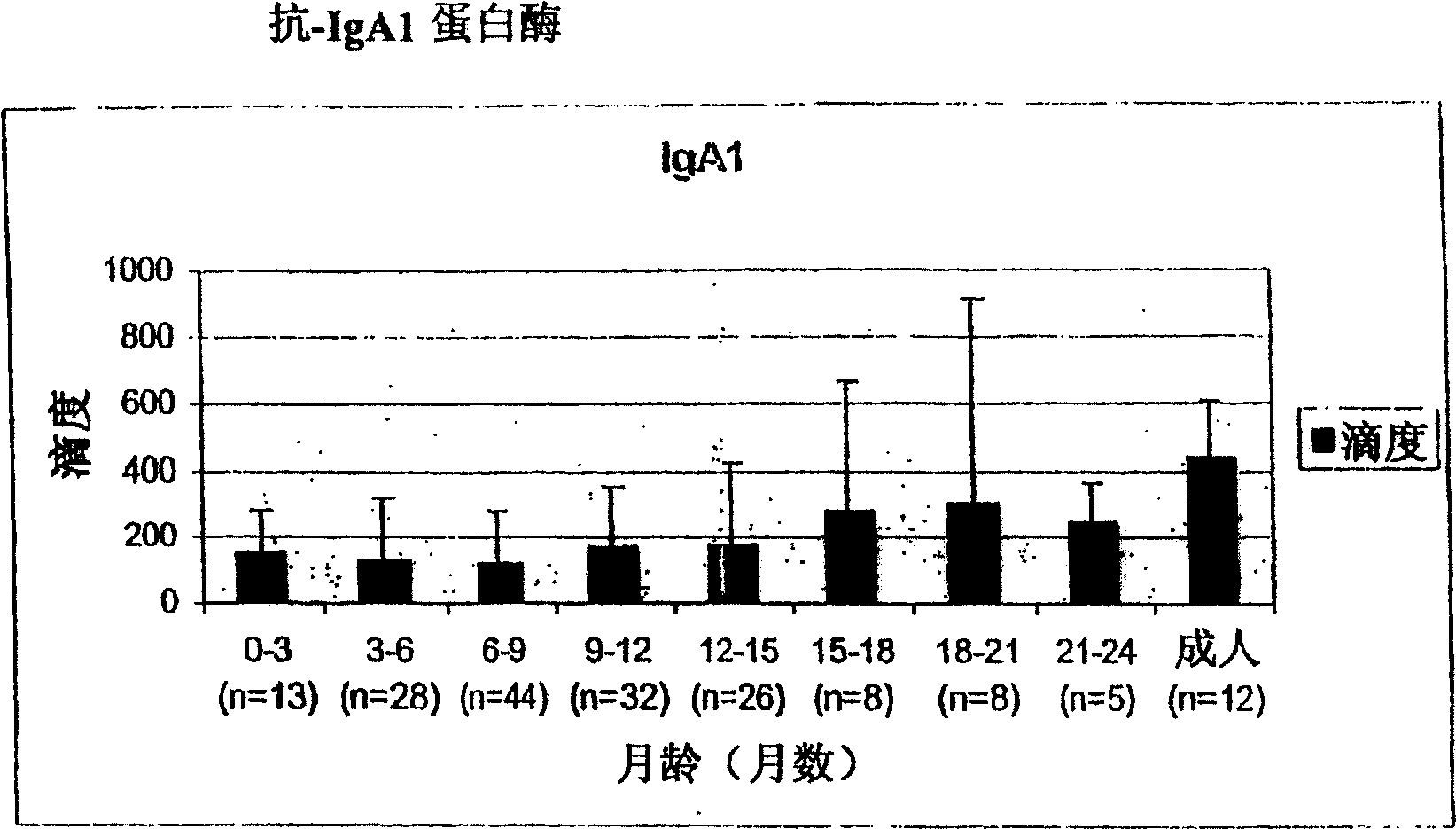
[0127] The relationship between the antibody titer against the surface protein of Streptococcus pneumoniae and the occurrence of the first acute otitis media
[0128] Survey of some groups in Finland
[0129] Such as Figure 1-5 IgG titers against PpmA, SlrA, and IgAl proteases were all lower than those against S. pneumoniae α-enolase in patients with a first onset of otitis media. Due to the lack of correlation between antibody titers and protection, SlrA and IgAl proteases were considered to have no added value for protective immunity and thus would not be selected as vaccine components.
Embodiment 2
[0131] Immunoprotective potential of surface proteins of Streptococcus pneumoniae
[0132] To determine the protective potential of S. pneumoniae surface proteins, both active and passive immunization / challenge experiments were performed.
[0133] method
[0134] mouse strain
[0135] Female mice of the MF1 cross were immunized and purchased from Harlan Olac, Bicester, UK. All mice were housed and free from specific pathogens. Active immunizations were initiated with 6-week-old mice (25-30 g) and passive immunizations with 9-week-old mice (30-35 g). strain
[0136] Streptococcus pneumoniae D39, serotype 2, was obtained from the National Center for Type Cultures (NCTC 7466; Central Public Health Laboratory, London). Streptococcus pneumoniae TIGR4, serotype 4, was obtained from the Genome Institute. Bacteria were grown on No. 2 blood agar matrix (Oxoid, Basingstoke, UK) supplemented with 5% (vol / vol) defibrinated horse blood (BAB). Stock cultures were prepared by inoculat...
Embodiment 3
[0255] Research on In silico
[0256] The nucleotide sequence of PpmA was determined by searching partial homology in the database.
[0257] The PpmA gene was amplified by PCR from Streptococcus pneumoniae D39 using the following primers:
[0258] 5' CCATGGCTAGCCACCATCACCATCACCATTCGAAAGGGTCAGAAGGTGC 3' and 5' TCATGGATCCGGACTATTCGTTTGATGTAC 3', which insert flanking NheI and BamHI restriction sites, and N-terminal His 6 mark. The amplified DNA was cloned into pETlla expression vector (Stratagene, LaJolla, CA) and electrotransfected into E. coli BL21(DE3). Ni was carried out by using HisTrap kit (Amersham Pharmacia) according to the manufacturer's suggestion. + Affinity chromatography was used to purify the recombinant protein.
[0259] Purified recombinant proteins were diafiltered with anti-10 mM HEPES buffer, pH 7.5, lyophilized and stored at -20°C.
[0260] PpmA
[0261] The ppmA Nucleotide Sequence of Streptococcus pneumoniae D39
[0262]ATGAAGAAAAAATTATTGGCAGGTGCCAT...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More