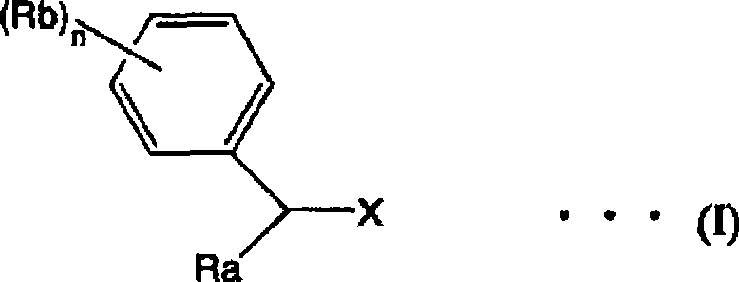
Method for producing 1,2-phenylethane compound using atom transfer radical coupling reaction
一种制造方法、苯基乙烷的技术,应用在1领域,能够解决反应收率低、易发生副反应、除去困难等问题,达到高收率的效果
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0058]
[0059] Add PhCOOEt to 50ml flask 2 -Br 0.39g (1mmol), CuBr 0.14g (1mmol), Cu(0) 0.25g (4mmol), toluene 20ml, degassed. Then add N, N, N', N', N"-pentamethyldiethylenetriamine 0.35g (2mmol), and stir at 80°C for 30 minutes. After the reaction solution is cooled, filter the insoluble matter. Wash the filtrate with water until The coloring disappears in the water layer, with MgSO 4 to dry. After the solvent was distilled off, it was dried under reduced pressure to obtain 0.30 g of white crystals (isolation yield 97%).
Embodiment 2
[0061]
[0062] In a 30ml flask, add 1.15g (5mmol) of 4-(bromomethyl) methyl benzoate, 0.72g (5mmol) of CuBr, 1.27g (20mmol) of Cu(0), 1.56g (10mmol) of bipyridine, 20ml of toluene and After degassing, it was stirred at 100°C for 1 hour. After cooling the reaction solution, 10 ml of chloroform was added, and the insoluble matter was filtered. After washing the filtrate with MgSO 4 to dry. After distilling off the solvent, recrystallization was performed with ethyl acetate / hexane. The obtained crystals were dried under reduced pressure to obtain 0.32 g of pale yellow crystals (isolation yield 43%).
Embodiment 3
[0064]
[0065] 9.6 g (48 mmol) of bis(4-hydroxyphenyl)methane, 11.8 g (117 mmol) of triethylamine, and 200 ml of tetrahydrofuran were placed in a 200 ml flask, and cooled to 0°C. 14.9 g (106 mmol) of benzoyl chloride was added thereto, followed by stirring at room temperature for 1 hour. After removing triethylamine hydrochloride by filtration, the filtrate was concentrated. After dissolving in dichloromethane, it was washed with water three times, and dried over magnesium sulfate. After concentrating the reaction solution, recrystallization was carried out from hexane / ethyl acetate to obtain 16.8 g of pale yellow needle crystals A (isolation yield: 86%).
[0066] 14.4 g (35 mmol) of A synthesized just now, 6.5 g (37 mmol) of N-bromosuccinimide, and 70 ml of benzene were placed in a 200 ml flask, and refluxed for 1 hour. After cooling, the reaction solution was concentrated. After dissolving in dichloromethane, it was washed with water three times, and dried over magnes...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More