Method for detecting quality of medicinal preparation for curing hepatitis
A quality inspection method and technology for pharmaceutical preparations, which are used in pharmaceutical combinations, pharmaceutical formulations, and medical preparations containing active ingredients, etc., can solve the problems of lack of quality control methods and the inability to ensure the effectiveness and safety of clinical medication.
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
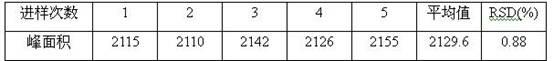
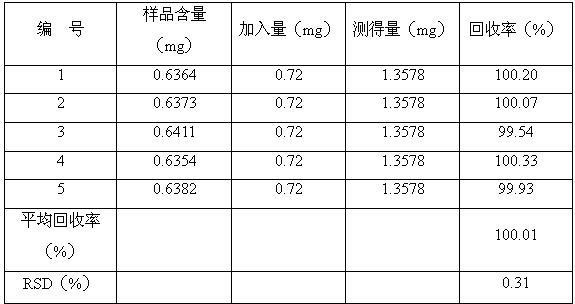
[0117] Implement 1. The quality control method of Jinmagantai Capsules for treating hepatitis is as follows:
[0118] Properties: This product is a hard capsule, the contents are brown to tan granules and powder; fragrant, bitter;
[0119] Identification: (1) Identification of Salvia miltiorrhiza: Take 3g of the contents of Jinmagantai capsule, add 25ml of methanol, heat and reflux for 1 hour, filter, evaporate the filtrate to dryness, add 20ml of water to dissolve the residue, shake and extract 3 times with ethyl acetate, 20ml each time, combined ethyl acetate solution, evaporated to dryness, and dissolved the residue in 1ml of methanol as the test solution. Take another protocatechualdehyde reference substance, add methanol to make a solution containing 1mg per 1ml, as the reference substance solution. According to the thin-layer chromatography (Appendix VIB, Chinese Pharmacopoeia, 2005 edition), draw 0.5-1 μl of the reference substance solution and 2-5 μl of the test solu...
Embodiment 2
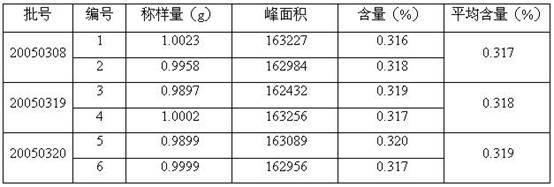
[0132] Implement 2. The quality control method of Jinmagantai tablets for treating hepatitis is as follows:
[0133] Properties: This product is a film-coated tablet, grayish-brown to tan after removing the film-coating; fragrant, bitter;
[0134] Identification: (1) Identification of Salvia miltiorrhiza: Take Jinmagantai tablet, remove the film coating, grind finely, take about 3g, add 25ml of methanol, heat and reflux for 1 hour, filter, evaporate the filtrate to dryness, add 20ml of water to the residue to dissolve, and use ethyl acetate Shake and extract the ester three times, 20ml each time, combine the ethyl acetate solution, evaporate to dryness, add 1ml methanol to the residue to dissolve, and use it as the test solution. Take another protocatechualdehyde reference substance, add methanol to make a solution containing 1mg per 1ml, as the reference substance solution. According to the thin-layer chromatography (Appendix VIB, Chinese Pharmacopoeia, 2005 edition), draw ...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More