Production method of inosinic acid
A technology of hypoxanthine nucleotide and production method, applied in the field of biosynthesis, can solve problems such as difficulty in separating and extracting hypoxanthine nucleotide, and achieve the effects of facilitating separation and purification, reducing investment and reducing equipment requirements
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
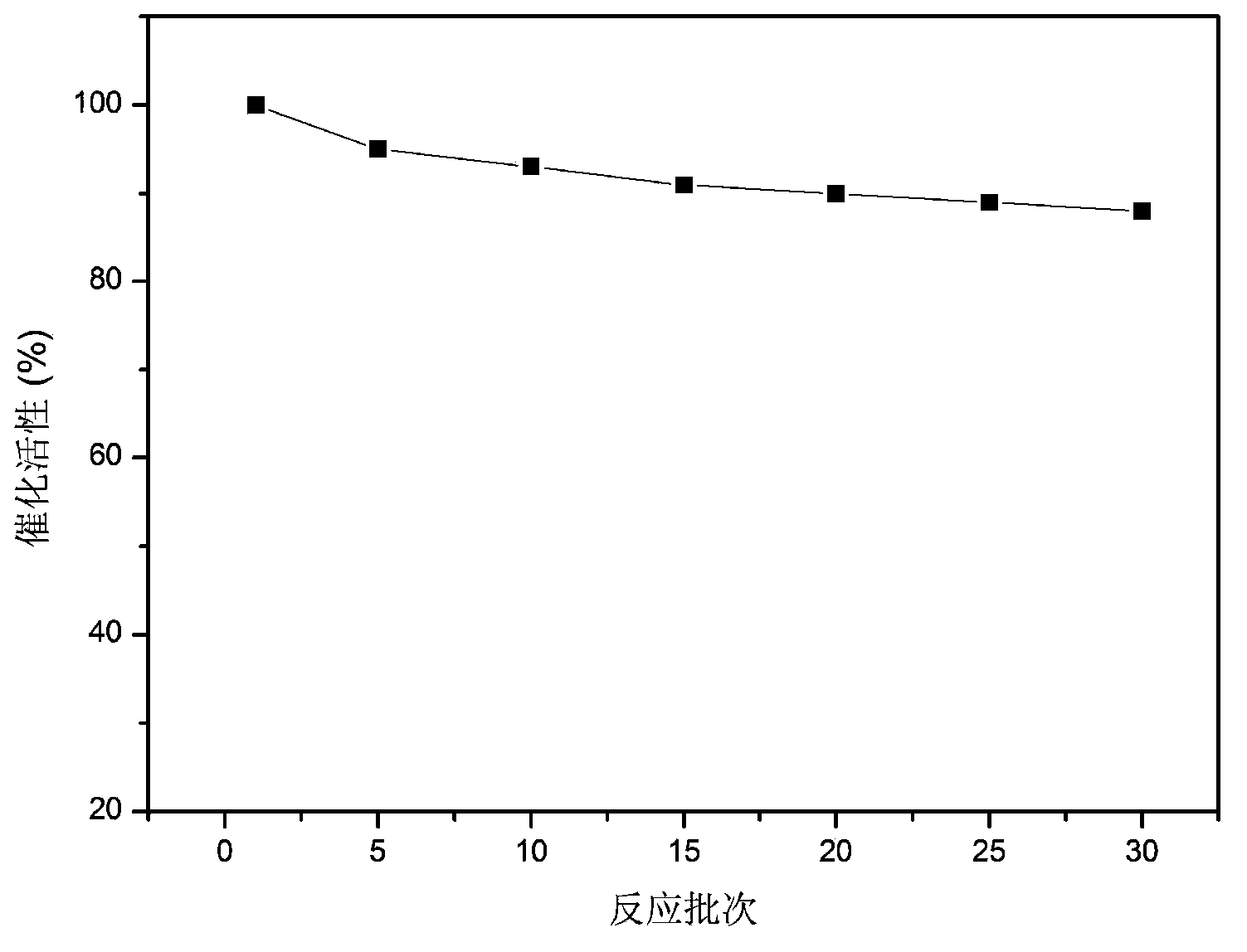
Examples
Embodiment 1
[0060] Pretreatment of resin: Amberlite IRA-900 resin was pretreated according to the following method steps:
[0061] Step 1a, weigh 200g of Amberlite IRA-900 resin in a 1L Erlenmeyer flask, add 600ml of absolute ethanol, soak overnight, wash with deionized water until there is no ethanol residue in the eluent, and then suction filter;
[0062] Step 1b, soak the resin obtained in step 1a with 600ml 1M NaOH solution overnight, wash with deionized water until the eluent is pH neutral, and then suction filter;
[0063] Step 1c, soak the resin obtained in step 1b with 600ml of 1M HCl solution overnight, wash with deionized water until the eluent is pH neutral, and then suction filter;
[0064] Step 1d, soak the resin obtained in step 1c with 600ml of 1M NaOH solution overnight, wash with deionized water until the eluent is pH neutral, and then suction filter;
[0065] Step 1e, soak the resin obtained in step 1d with 600ml of 0.5M NaCl solution overnight, wash with deionized wate...
Embodiment 2
[0069] Resin pretreatment: Amberlite IRA-93 resin was pretreated according to the same method as in Example 1.
[0070] Catalyzed reaction: Weigh 10 g of the above-mentioned pretreated Amberlite IRA-93 resin into a 100 ml Erlenmeyer flask, then add 50 ml of adenylate deaminase solution (10 U / ml, add acetate buffer solution, adjust the pH to 5 ~6), place the flask on a shaker at 20°C and 150rpm for 2 hours, recover the immobilized enzyme, and wash the immobilized enzyme with deionized water until there is no protein and enzyme activity in the eluate. Add the obtained immobilized enzyme (13.17U / g) to 100ml of adenylic acid solution (100g / L, pH5~6) preheated at 40°C, and make the adenylic acid The deamination reaction is carried out, and the pH is controlled to be 5-6 by dropwise adding 1M hydrochloric acid solution. After about 2.5 hours, OD265 no longer drops, liquid phase detection (liquid phase conditions: mobile phase is 0.02M ammonium dihydrogen phosphate solution, contain...
Embodiment 3
[0072] Resin pretreatment: Amberlite IRA-400 resin was pretreated according to the same method as in Example 1.
[0073] Catalytic reaction: Weigh 10 g of the above-mentioned pretreated Amberlite IRA-400 resin into a 100 ml Erlenmeyer flask, then add 50 ml of adenylate deaminase solution (10 U / ml, add acetate buffer solution to adjust the pH to 5 ~6), place the flask on a shaker at 20°C and 150rpm for 2 hours, recover the immobilized enzyme, and wash the immobilized enzyme with deionized water until there is no protein and enzyme activity in the eluate. Add the obtained immobilized enzyme (14.37U / g) into 100ml of adenylic acid solution (100g / L, pH5~6) preheated at 40°C, and make the adenylic acid at 40°C, under the condition of mechanical stirring at 60rpm The deamination reaction is carried out, and the pH is controlled to be 5-6 by dropwise adding 1M hydrochloric acid solution. After about 3.25h, OD265 no longer drops, liquid phase detection (liquid phase conditions: mobile...
PUM
| Property | Measurement | Unit |
|---|---|---|
| concentration | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More