A kind of magnetic material constructed with acylhydrazone Schiff base as a ligand and its preparation method
A technology of magnetic materials and Schiff bases, applied in the direction of magnetism of organic materials/organic magnetic materials, chemical instruments and methods, compounds containing elements of group 3/13 of the periodic table, etc., can solve the problem of rare earth magnetic materials that have not yet been seen low cost of raw materials, good repeatability and high yield
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
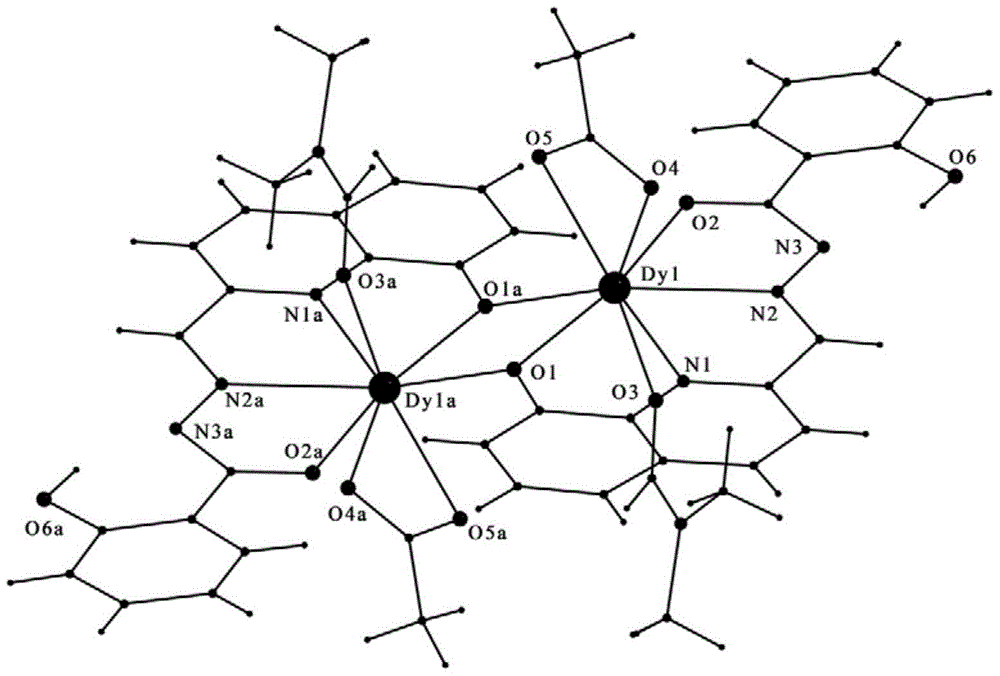
[0033] The magnetic material [Dy 2 L 2 (CH 3 COO) 2 (DMF) 2 ], where L is 2-formyl-8-hydroxyquinoline salicylhydrazone, and DMF is N,N-dimethylformamide.
[0034] The above-mentioned magnetic material [Dy 2 L 2 (CH 3 COO) 2 (DMF) 2 ] The preparation method is:
[0035] Weigh the analytically pure Dy(CH 3 COO) 3 ·6H 2 O and 2-formyl-8-hydroxyquinoline salicylhydrazone (wherein Dy(CH 3 COO) 3 ·6H 2 O is 0.1mmol, that is, 0.0898g), put it in a beaker, then add a mixed solvent consisting of 1.6mL of anhydrous methanol and 0.8mL of N,N-dimethylformamide, dissolve, shake well, and then use analytical pure three Ethylamine was used to adjust the pH of the obtained solution to 8; the obtained mixed solution was transferred into a Pyrex thick-walled glass tube about 25 cm long, and the tube was vacuum-sealed by freezing with liquid nitrogen, and then the fused-sealed Pyrex thick-walled glass tube was placed at 80°C Reaction under heat preservation for 48h; natural cooli...
Embodiment 2
[0047] Repeat Example 1, except that:
[0048] 1) Dy(CH 3 COO) 3 ·6H 2 The amount of O is changed to 0.05mmol, which is 0.0449g;
[0049] 2) Change the mixed solvent to consist of 1.4mL of anhydrous methanol and 0.7mL of N,N-dimethylformamide;
[0050] 3) adjust the pH value of the solution to 8.5;
[0051] 4) The reaction temperature is controlled at 90°C, and the reaction time is controlled at 24h.
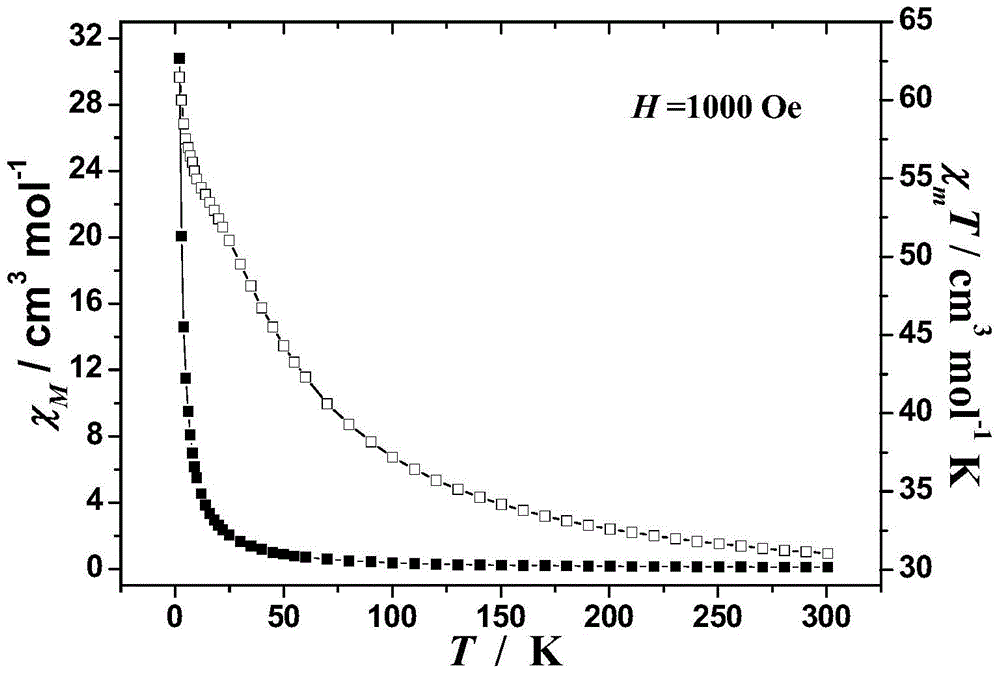
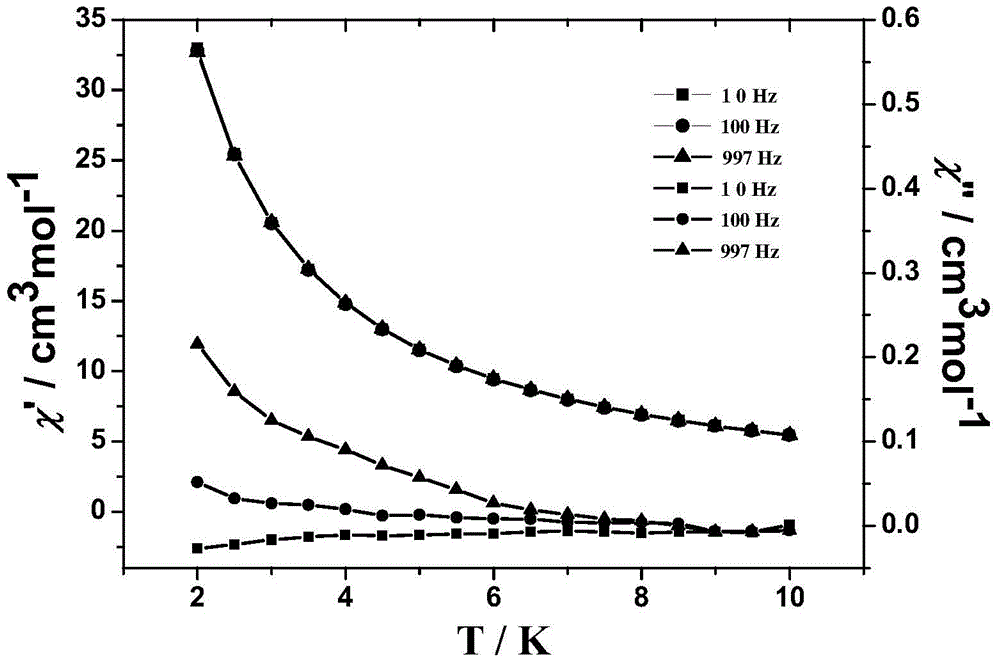
[0052] The obtained product was characterized by single crystal structure and X-ray powder diffraction, and it was determined that the target product was a magnetic material [Dy 2 L 2 (CH 3 COO) 2 (DMF) 2 ], the characterization of the magnetic properties of the product shows that the obtained magnetic material is χ at room temperature M T is 31.2cm 3 Kmol -1 , as the temperature decreases, χ m T increases gradually and reaches a maximum value of 62.4cm at 2K 3 Kmol -1 . and χ M –1 The -T curve obeys the Curie-Weiss law, and the Weiss constant is 44.9K and the ...
Embodiment 3
[0054] Example 1 was repeated except that the pH of the solution was adjusted to 9.0.
[0055] The obtained product was characterized by single crystal structure and X-ray powder diffraction, and it was determined that the target product was a magnetic material [Dy 2 L 2 (CH 3 COO) 2 (DMF) 2 ], the characterization of the magnetic properties of the product shows that the obtained magnetic material is χ at room temperature M T is 31.2cm 3 Kmol -1 , as the temperature decreases, χ m T increases gradually and reaches a maximum value of 62.4cm at 2K 3 Kmol -1 . and χ M –1 The -T curve obeys the Curie-Weiss law, and the Weiss constant is 44.9K and the Curie constant is 58.2cm. 3 Kmol –1 . Positive Weiss constant and χ M The trends of T-T curves all indicate that there is ferromagnetic exchange between dysprosium ions in the molecule, and the overall performance is paramagnetic.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More