Mammalian cell culture processes for protein production
A cell culture and cell technology, applied in the field of new process of culturing mammalian cells, can solve the unreported problems of dextran sulfate or polyvinyl sulfate effect, etc., to achieve increased titer, high cell viability, and extended production period Effect
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 3
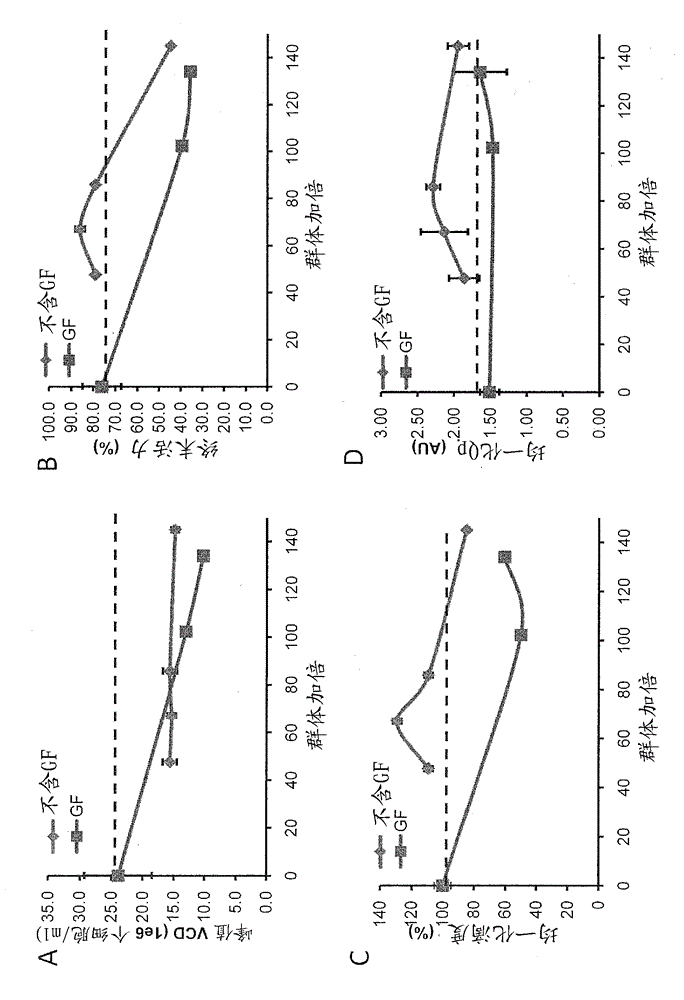
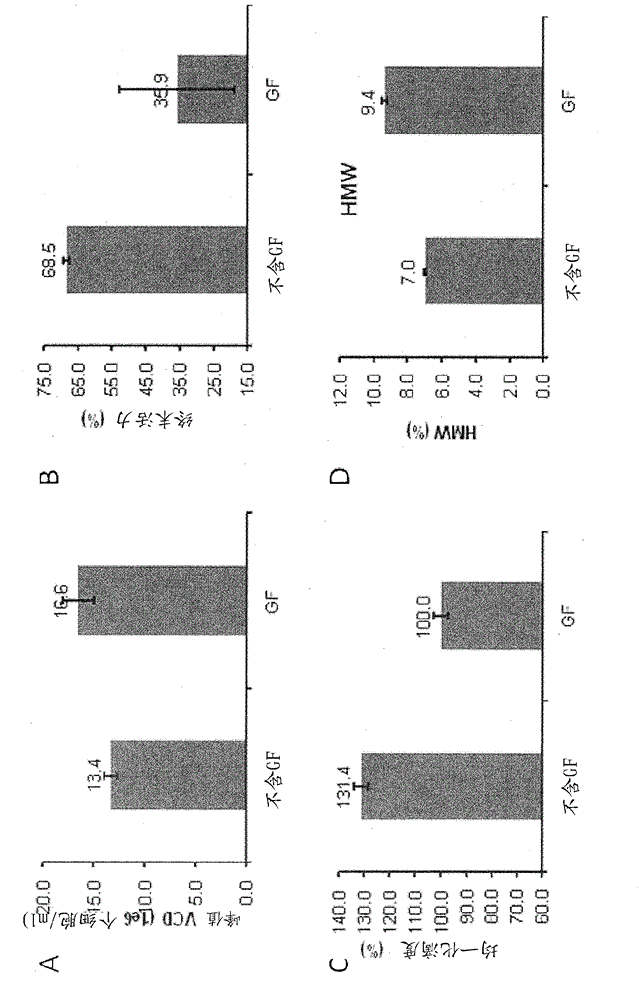
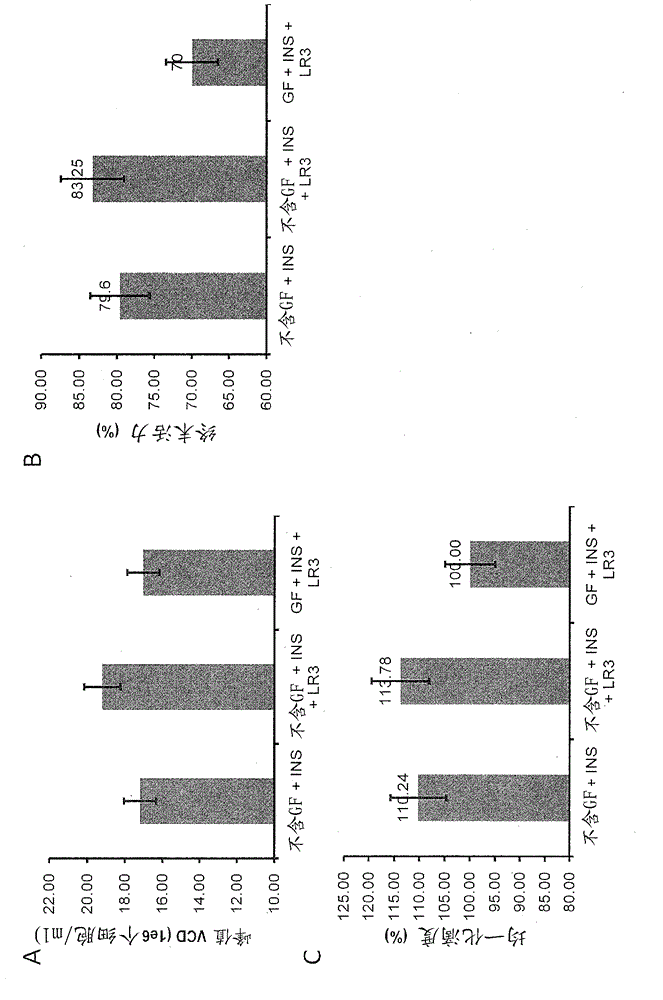
[0040] The fed-batch culture production assay described in Example 3 found that conditions without GF increased the peak viable cell density from 13.0 × 10 6 cells / ml increased to 15.7 × 10 6 cells / ml, and improved cell viability at later stages of culture, from 59.4% to 96.2% in the GF condition on day 8, and from 35.6% to 65.1% on day 14. As a result, the protein titer increased by 80.9%. Furthermore, protein quality was enhanced under GF-free conditions, as macromolecular species decreased from 14.3% to 4.8% under GF conditions (see Figure 4 ).
[0041] also, Figure 5 – 8 shows that the generation stability of cells is improved in the absence of GF in three different clones. After culturing cells under GF conditions, productivity decreased, whereas cells cultured without GF maintained their productivity.
[0042] Overall, our results demonstrate that chemically defined media free of proteins and peptides are able to propagate CHO cells, deliver comparable productivi...
Embodiment 1
[0125] CHO cells are able to grow without GF
[0126] 1. Thaw a new vial of cells from the previous passage and grow in platform medium (containing 1 or 10 mg / L insulin) for 2 passages.
[0127] 2. At passage 3, transfer the cells to basal medium without insulin and inject with 0.6x10 6 The density of cells / ml was kept splitting.
[0128] 3. In the first few passages under insulin-free conditions, cell growth can be significantly slowed and viability can be reduced (~90%). At the same time, ammonium production can be increased due to insufficient glucose uptake and oxidation of amino acids as energy sources. As a result, the pH in the flask can increase. CO 2 Levels may need to be adjusted / increased accordingly in order to control the pH in the range of 7.0-7.3. This is very important for maintaining cells in a healthy state.
[0129] 4. In general, less than 30% of the spent medium maintained to new passages should be maintained. In cases where cells are growing too sl...
Embodiment 2
[0134] Removing insulin did not drastically affect the mTOR pathway
[0135] The purpose of this study was to use antibody arrays to compare the phosphorylation / expression levels of proteins involved in mTOR in cells grown with and without the growth factor insulin, thereby demonstrating that the cells are able to function in Grow without GF.
[0136] Antibody Array Sample Preparation
[0137] Clone A was grown on basal medium without or with insulin. On day 3, sample 5x 10 for each condition 6living cells. Cells were centrifuged down at 500 g for 5 min at 4°C. Cells were washed with 10 ml ice-cold 1xPBS and centrifuged down at 500g for 5 min at 4°C. Keep cells on ice or at 4 °C throughout sample processing. Immediately after pouring the PBS, the cells were frozen at -70°C and stored at -70°C until antibody array analysis.
[0138] Antibody Array Protocol
[0139] protein extraction
[0140] Wash cells with ice-cold 1X PBS. Lysis beads and extraction buffer are added...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com