A kind of preparation method of metadoxine impurity compound
A technology of metadoxine and compounds, which is applied in the field of preparation of metadoxine impurity compounds, can solve problems such as hidden dangers of drug quality, impact on health, and rare preparation methods, and achieve the effect of improving quality standards
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
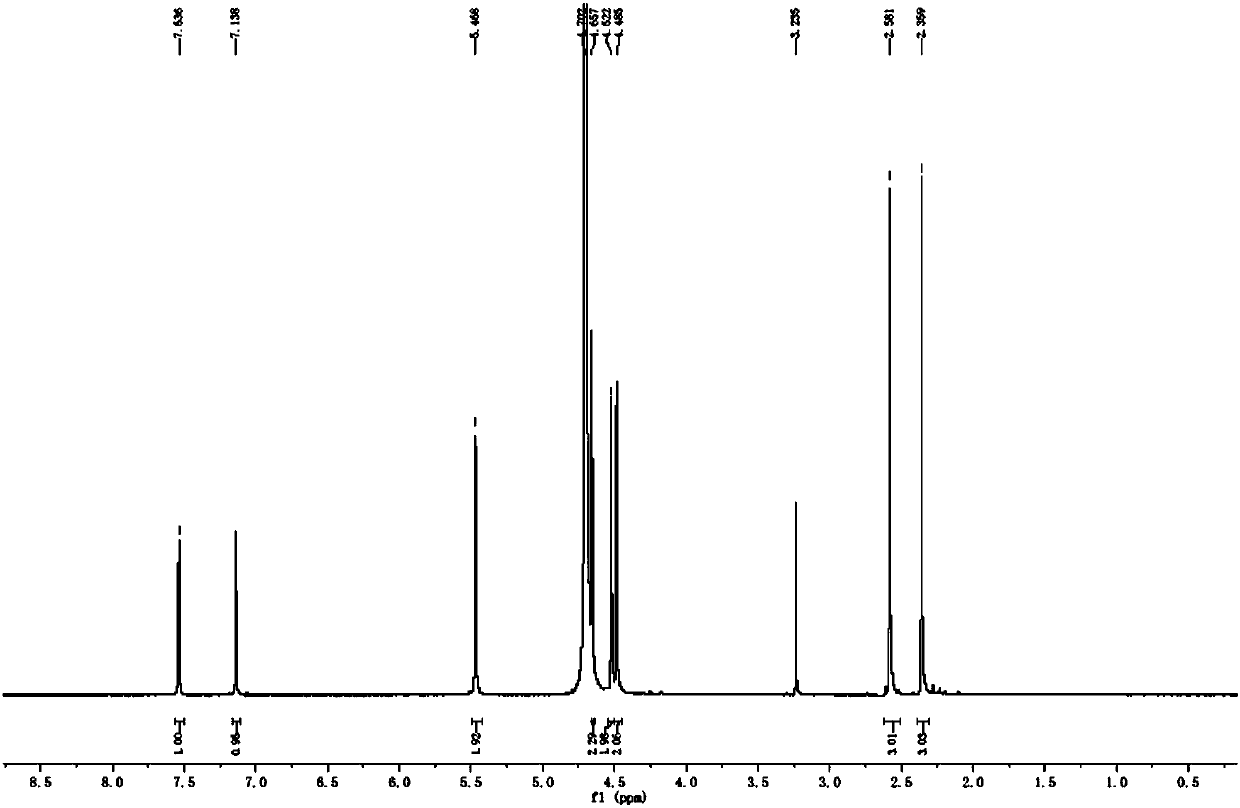
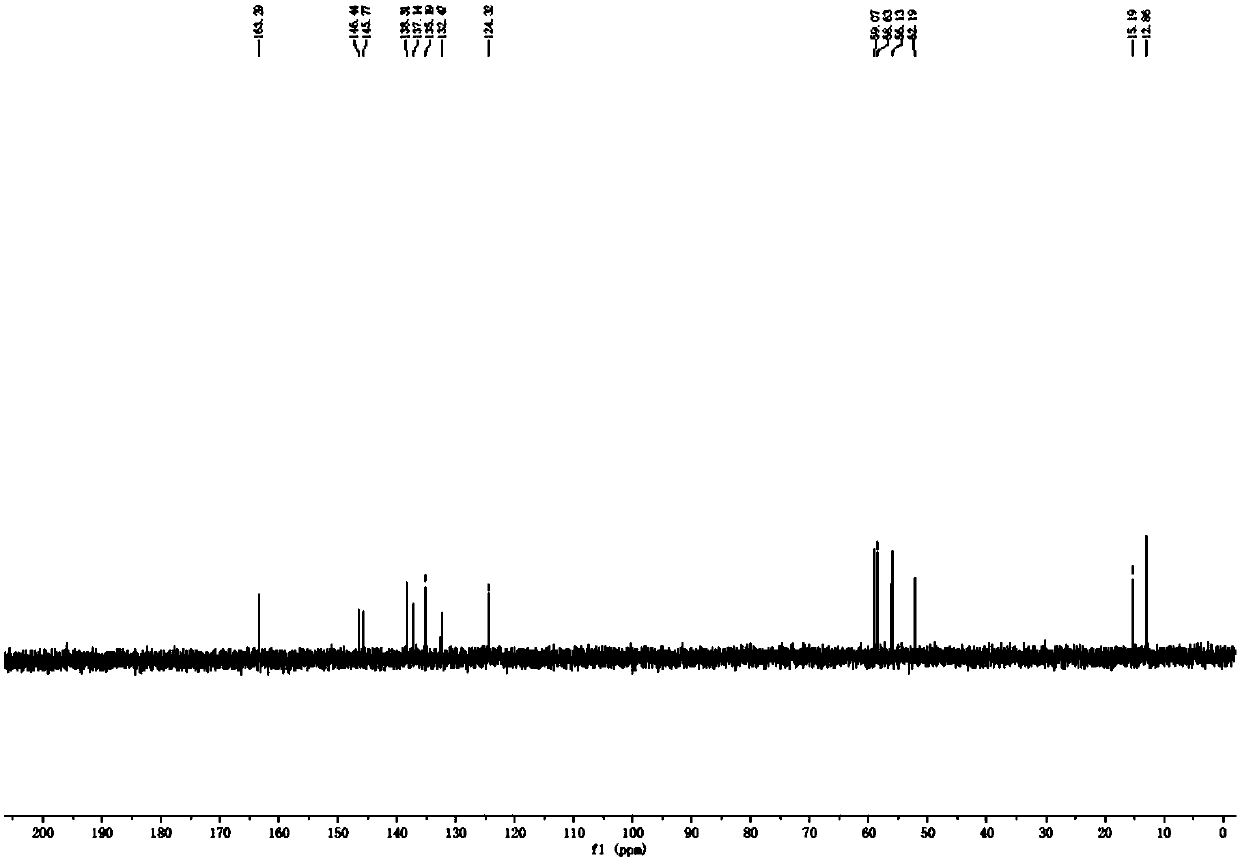
[0028] Under the protection of nitrogen, add 50g of metadoxine and 500ml of water into a 1L pressure reactor; start stirring, and adjust the pH of the reaction system to 6.0 with 20% aqueous sodium hydroxide solution after the solid is dissolved, and control the reaction temperature to 50°C and the reaction pressure 9.5 psi, reaction 3 hours. TLC monitors until the reaction is complete. The reaction solution was concentrated under reduced pressure and purified by column chromatography to obtain a solid, namely metadoxine impurity compound, with a weight of 5.7 g, a yield of 21.9%, and a purity of 98.8%. NMR (in D 2 (O is solvent) and after high-resolution mass spectrometry detection, the structural formula of this metadoxine impurity compound is Its molecular formula is C 16 h 21 N 2 o 5 , whose chemical name is 1-((3-hydroxy-5-(hydroxymethyl)-2-methylpyridin-4-yl)methyl)-4,5-bis(hydroxymethyl)-2-methyl Pyridin-1-ium-3-ol inner salt.
[0029] The following are proton ...
Embodiment 2
[0035] Under nitrogen protection, add 50g of metadoxine, 300ml of methanol and 100ml of water into a 1L pressure reactor; start stirring, and adjust the pH of the reaction system to 6.0 with 20% aqueous sodium hydroxide solution after the solid is dissolved, and control the reaction temperature to 50°C , pressure is 9.5psi, reacts for 3 hours, TLC monitors until the reaction is complete; the reaction solution is concentrated under reduced pressure, and purified by column chromatography to obtain a solid, namely metadoxine impurity compound, with a weight of 9.9g, a yield of 38.1%, a purity of was 98.7%. Other measures are with embodiment 1.
Embodiment 3
[0037] Under nitrogen protection, add 50g metadoxine, 300ml isopropanol and 100ml water in a 1L pressure reactor; start stirring, adjust the pH of the reaction system to 6.0 with 20% aqueous sodium hydroxide solution after the solid is dissolved, and control the reaction temperature to 50°C, reaction pressure 5psi, reacted for 3 hours; TLC monitored until the reaction was complete, the reaction solution was concentrated under reduced pressure, and purified by column chromatography to obtain a solid, namely metadoxine impurity compound, with a weight of 11.2g and a yield of 43.2%. , with a purity of 98.5%. Other measures are with embodiment 1.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com