Methods for producing dicarboxylic acids
A dicarboxylic acid and product technology, applied in the field of dicarboxylic acid production, can solve the problems of reducing activity and catalytic conversion
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0073] Example 1 - Enzyme Engineering
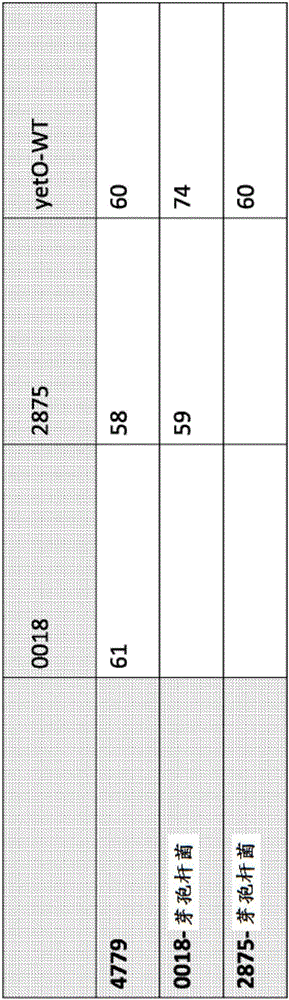
[0074] This example shows the engineering of wild-type (wt) BM3 and wild-type (wt) YetO to generate a set of mutant enzymes with the desired activities of peroxidizing fatty acid subterminals and cleaving C-C bonds. Cloning of BM3 (or CYP102A1) from Bacillus megaterium and a proprietary homolog from a Bacillus strain (YetO from Bacillus 15_F03). There is common media homology between BM3 and YetO (60% identity, 75% similarity). Native gene sequences were used in all plasmids. The YetO gene was cloned from gDNAPCR, while for BM3, the natural DNA sequence was used to make the synthetic gene. Various site-specific mutants from these sequences were then created, as shown in Table 1.
[0075] Table 1: Original BM3 and cloned homologues
[0076]
[0077] This article shows the sequence. For F87 (in BM3) and F89 (in YetO), the phenylalanine site was mutated to alanine in BM3, and to alanine, isoleucine, serine, or valine to produce Mutan...
Embodiment 2
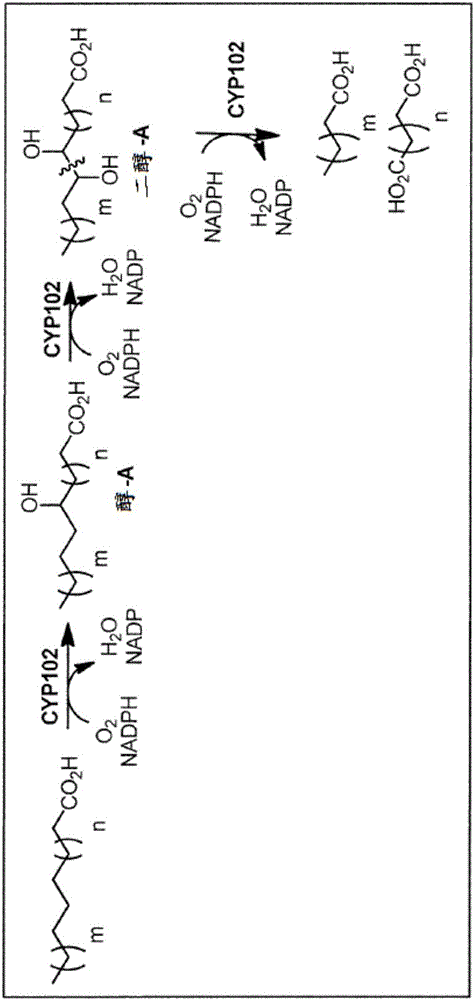
[0083] Example 2 - Oxidation of palmitic acid by BM3-YetO
[0084] Strain BL21DE3 / pSGI-040 (pET24a-BM3-yetO-WT) was grown in 150 mL of M9TV / TB (3 / 1 v / v) + glucose (2 g / L) + FeCl3 (0.05 mM) on. Cells were grown at 30°C for 3-4h until reaching OD600-0.5. At this point, 0.5 mM δ-aminolevulinic acid and 0.25 mM IPTG were added, and the flask was moved to a shaker at room temperature (~23°C), where it was allowed to shake at 120 rpm for an additional 16h.
[0085] Cells were harvested by centrifugation and frozen at -80°C for at least 1 h. Frozen cells were resuspended in 15 mL lysis buffer (100 mM potassium phosphate, 10% glycerol (v / v), 1 mM DTT and 1 mg / mL lysozyme). After thawing the pellet, it was incubated on ice for 15 min before the cells were sonicated twice. Lysed cells were centrifuged at 12,000 rpm to pellet insoluble cell debris. The clarified lysate was used in the enzymatic reaction without any further purification. A P450 of approximately 8-10 uM (or nanomoles...
Embodiment 3
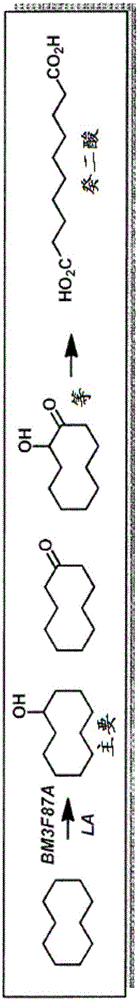
[0088] Example 3 - Oxidation of Cyclohydrocarbons
[0089] This example demonstrates that a mutant CYP102 enzyme (BM3-F87A) oxidizes cyclic hydrocarbons, including cyclooctane and cyclodecane, and also produces cyclodecane C-C bond cleavage. It was found that sebacic acid (sebacic acid) was produced after peroxidation when the enzyme was placed in the presence of cyclodecane.
[0090] By performing the reaction in the presence of deuterated lauric acid, we were able to trace the carbon of the sebacic acid produced to cyclodecane, rather than lauric acid. Therefore using cyclodecane and fully deuterated lauric acid (C 12 D. 23 CO 2 H) to carry out the reaction.
[0091] BL21(DE3) / pSGI-004(pET24a-BM3-F87A) cells were grown and lysed as described above. P450 concentrations of -14-20 [mu]M were obtained under these conditions. Prepare the reaction by mixing 1.5 mL of reaction buffer and 0.5 mL of enzyme lysate. By adding 4 mM deuterated lauric acid (C 12 D. 23 CO 2 H), 1...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com