A kind of synthetic method of n-ethylcarbazole
A technology of ethylcarbazole and synthesis method, applied in the direction of organic chemistry and the like, can solve the problems such as the difficulty of hydrogenation reduction of vinyl, and achieve the effects of high product quality, cost reduction, and reduction of three waste discharges
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0024] In a 50ml glass pressure-resistant bottle, add 2g carbazole, 0.04g proton-removing agent KOH and 10ml N-methylpyrrolidone as a solvent, start stirring and raise the temperature to 80°C for a salt-forming reaction for 30 minutes to generate carbazole potassium salt, and then Pass through acetylene, react for 2.5 hours at 160°C under normal pressure, and obtain N-vinyl carbazole with a yield of 98.6%; in the next step, continue hydrogenation to generate N-ethyl carbazole, according to the catalyst and N-vinyl carbazole The molar concentration ratio of the azole substance is 1:2000, add Pd catalyst (support is MgO), pass 0.3MPaH 2 , hydrogenation reaction was carried out at 70°C for 3 hours to obtain N-ethylcarbazole with a yield of 98.8%.
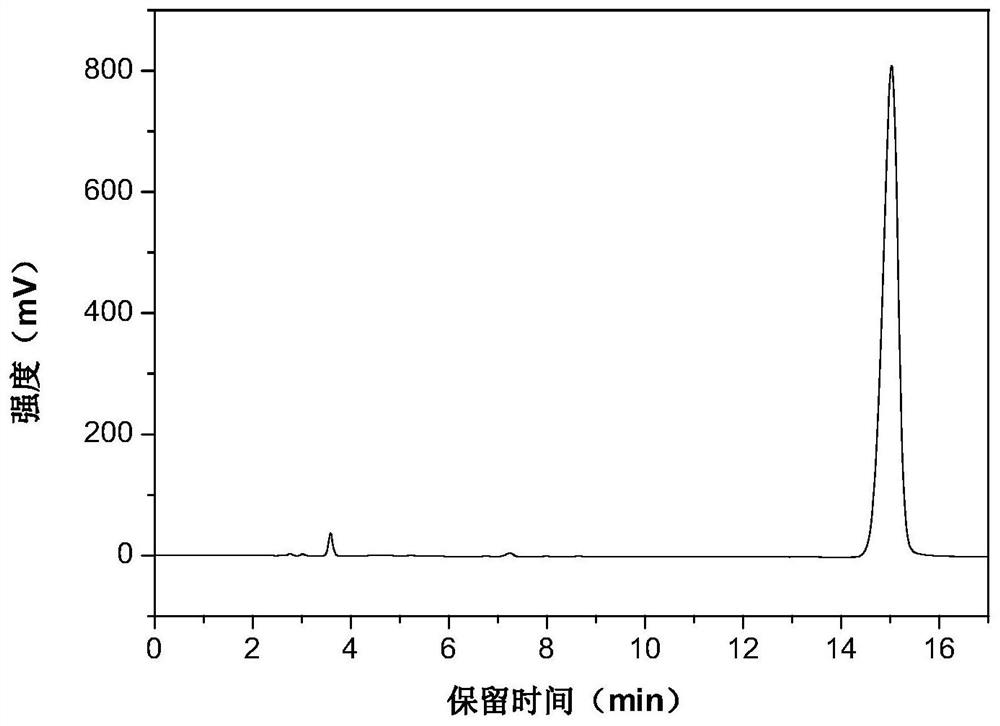
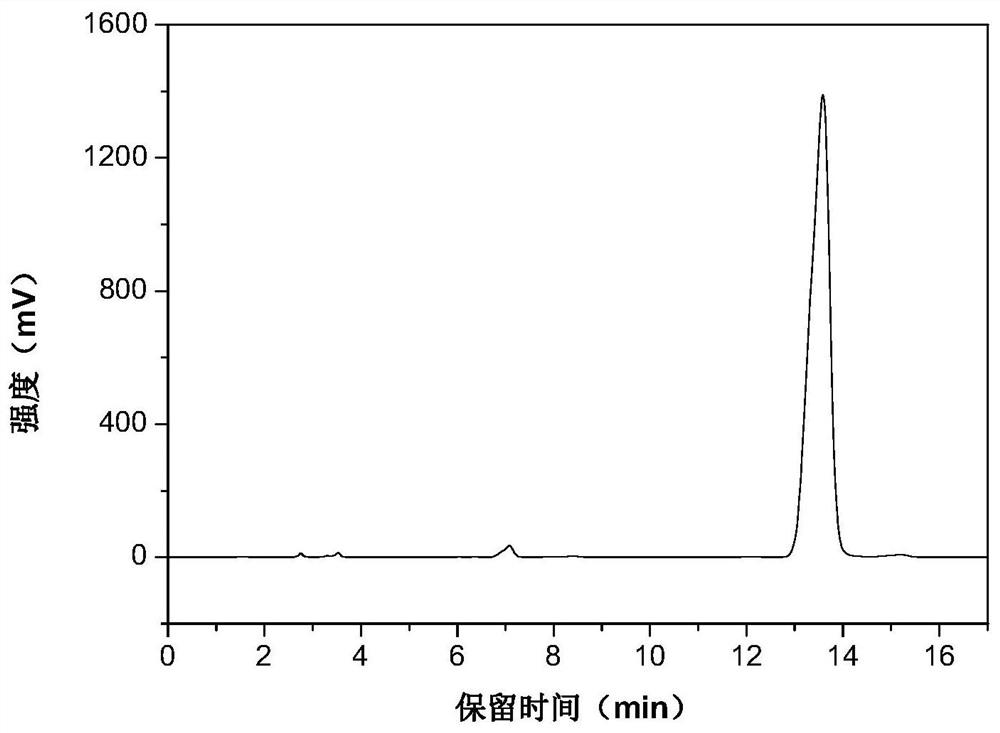
[0025] The high performance liquid phase chromatogram of the intermediate product N-vinylcarbazole of the embodiment of the present invention 1 sees figure 1 , the high performance liquid chromatogram of the product N-ethylcarbazole o...
Embodiment 2
[0027] In a 50ml glass pressure-resistant bottle, add 2g carbazole, 0.08g proton-removing agent KOH and 10ml N-methylpyrrolidone as a solvent, start stirring and heat up to 80°C for a salt-forming reaction for 20 minutes to generate carbazole potassium salt, and then Pass through acetylene, react at 160°C and normal pressure for 5 hours, and obtain N-vinylcarbazole with a yield of 99.0%; in the next step, continue hydrogenation to generate N-ethylcarbazole, according to the catalyst and N-vinylcarbazole The molar concentration ratio of the substance is 1:2000, add Pd catalyst (support is MgO), pass 0.3MPaH 2 , hydrogenation reaction was carried out at 70°C for 3 hours to obtain N-ethylcarbazole with a yield of 98.7%.
Embodiment 3
[0029] In a 50ml glass pressure-resistant bottle, add 2g carbazole, 0.12g proton-removing agent KOH and 10ml N-methylpyrrolidone as a solvent, start stirring and raise the temperature to 80°C for a salt-forming reaction for 20 minutes to generate carbazole potassium salt, and then Pass through acetylene, react for 2.5 hours at 160°C under normal pressure, and obtain N-vinyl carbazole with a yield of 98.9%. The molar concentration ratio of the azole substance is 1:2000, add Pd catalyst (support is MgO), pass 0.3MPaH 2 , hydrogenation reaction was carried out at 70°C for 3 hours to obtain N-ethylcarbazole with a yield of 98.4%.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D
- Intellectual Property
- Life Sciences
- Materials
- Tech Scout
- Unparalleled Data Quality
- Higher Quality Content
- 60% Fewer Hallucinations
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2025 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com