A method for expressing and preparing sucrose phosphorylase
A technology of sucrose phosphorylase and phosphorylase gene, which is applied in the field of genetic engineering, can solve problems such as not suitable for large-scale preparation, low protein expression, and difficulty in separation and purification, and achieve good production technology, simple and fast cultivation, The effect of high application value
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0026] (1) Construction of recombinant expression vector pHT43-SP
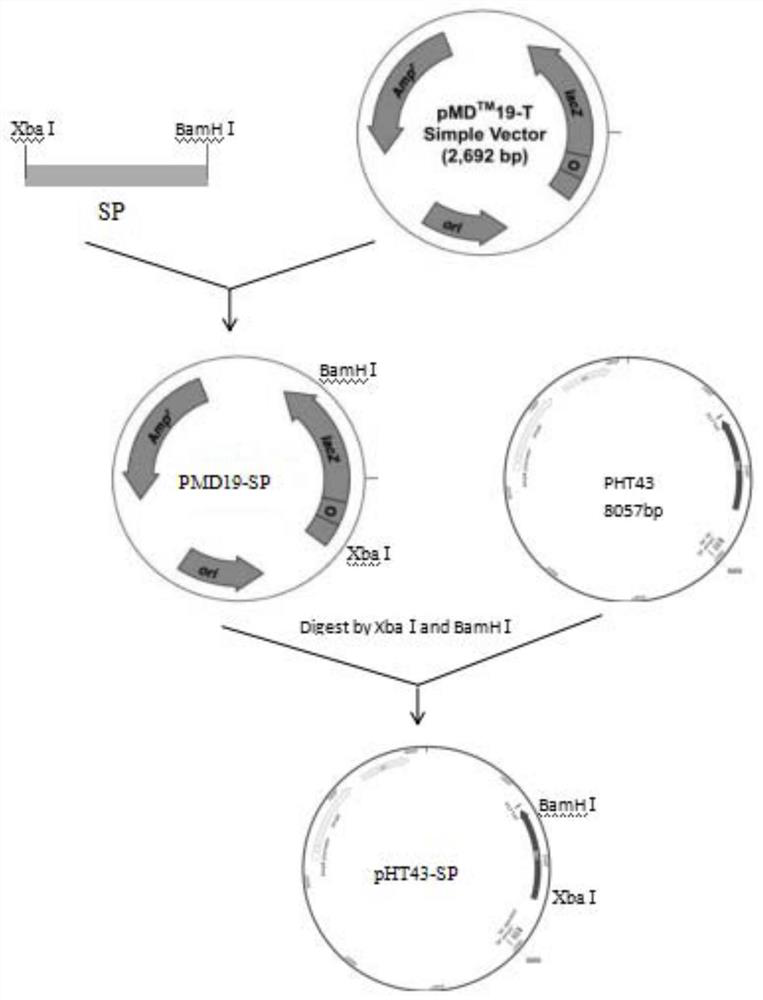
[0027] The sucrose phosphorylase (SP) gene (sequence shown in SEQ ID NO: 1) was derived from Bifidobacterium longum JCM 1217. After PCR amplification and purification, it was connected to the cloning vector pMD19 to construct the recombinant plasmid pMD19-SP.
[0028] The recombinant plasmid pMD19-SP and the expression vector pHT43 were double-digested with XbaI and BamHI respectively, and ligated overnight at 16°C to obtain the recombinant expression vector pHT43-SP.
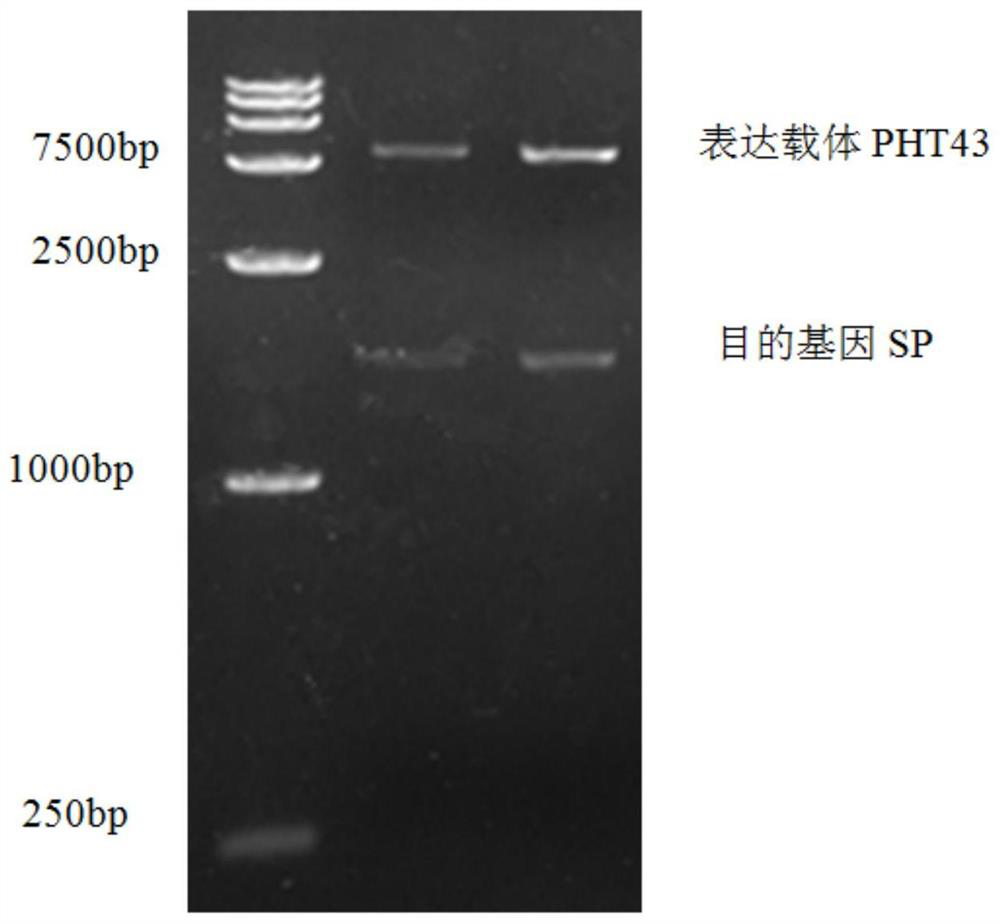
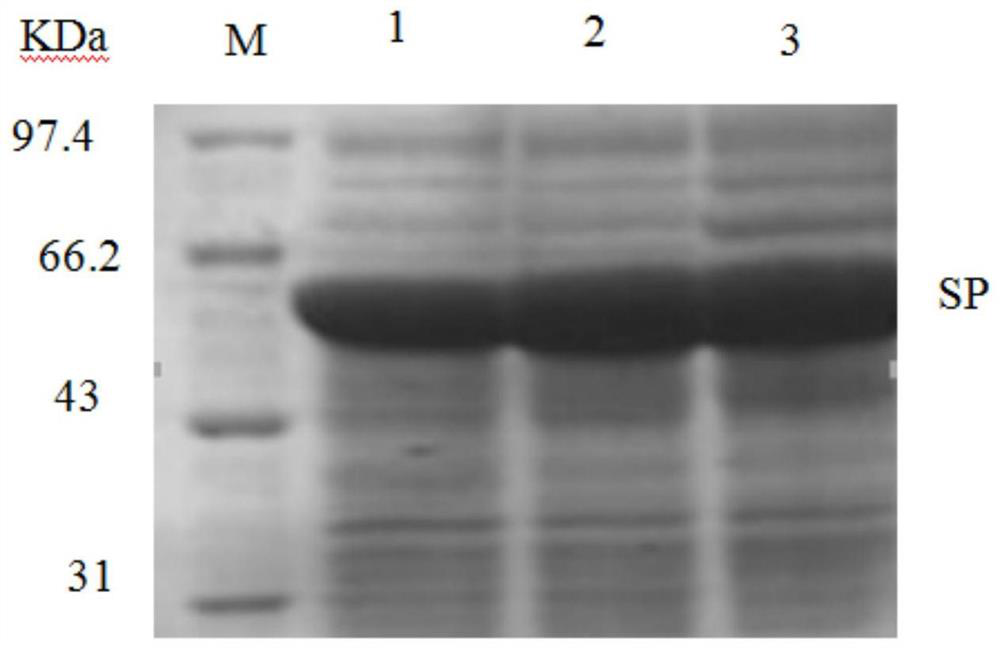
[0029] The recombinant expression vector pHT43-SP was transformed into Bacillus subtilis WB800N, spread on LB plates containing chloramphenicol (5ug / mL) resistance, cultured at 37°C, picked transformants, extracted recombinant plasmids and verified by double enzyme digestion. Such as figure 2 As shown, there are two fragments after enzyme digestion, the sizes are about 8000bp (expression vector pHT43) and 1819bp (sucrose acidase), respectivel...
Embodiment 2
[0046] (1) Construction of recombinant expression vector pHT43-SP
[0047]The recombinant plasmid pMD19-SP prepared in Example 1 and the expression vector pHT43 were double digested with XbaI and BamHI respectively, and ligated overnight at 16°C to obtain the recombinant expression vector pHT43-SP.
[0048] The recombinant expression vector pHT43-SP was transformed into Bacillus subtilis WB800, spread on LB plates containing chloramphenicol (5ug / mL) resistance, cultured at 37°C, picked transformants, extracted recombinant plasmids and verified by double enzyme digestion. Such as figure 2 As shown, there are two fragments after enzyme digestion, the sizes are about 8000bp (expression vector pHT43) and 1819bp (sucrose acidase), respectively, indicating that the connection is successful.
[0049] (2) Recombinant engineering bacteria
[0050] The constructed recombinant expression vector pHT43-SP was transformed by electric shock, and 60ul of Bacillus subtilis WB800 electropora...
Embodiment 3
[0056] (1) Construction of recombinant expression vector pMA5-SP
[0057] The recombinant plasmid pMD19-SP and the shuttle vector pMA5 prepared in Example 1 were double-digested with XbaI and BamHI respectively, and ligated overnight at 16°C to obtain the recombinant expression vector pMA5-SP.
[0058] The recombinant expression vector pMA5-SP was transformed into Bacillus subtilis 168, coated with LB plates containing ampicillin (100ug / mL) resistance, cultured overnight at 37°C, the transformants were picked, the recombinant plasmid was extracted and verified by double enzyme digestion. The build was successful.
[0059] (2) Recombinant engineering bacteria
[0060] The constructed recombinant expression vector pMA5-SP was transformed by electric shock. Bacillus subtilis 168 electroporated competent cells were mixed with the recombinant expression vector pMA5-SP plasmid, and then placed in an electric shock cup for ice bath for 5 minutes before electric shock. Electric shock...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More