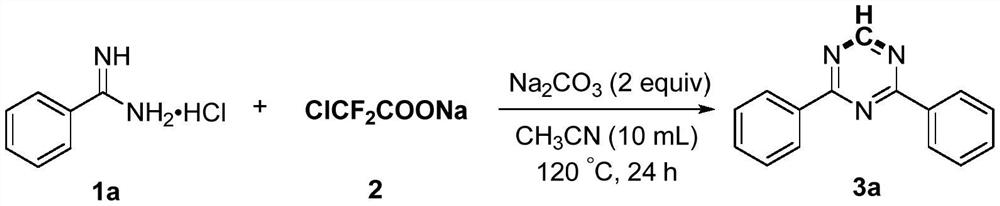A kind of preparation method of multi-substituted 1,3,5-triazine
A multi-substitution and substituent technology, which is applied in the field of preparation of multi-substituted 1,3,5-triazines, can solve problems such as environmentally unfriendly by-products
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0020]
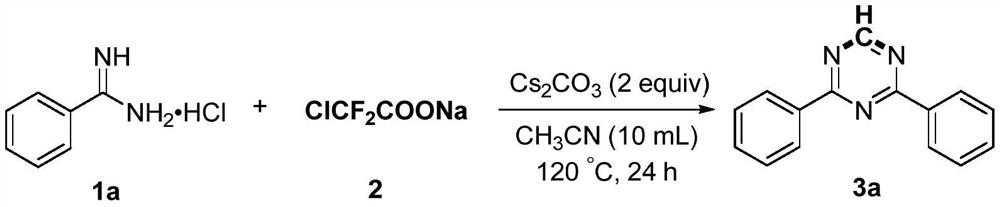
[0021] Add 1a (4mmol), sodium difluorochloroacetate (4mmol), cesium carbonate (8mmol) and acetonitrile (10mL) into the reaction flask, then heat the reaction at 120°C for 24 hours. After the reaction was completed, it was first quenched with water, then extracted with ethyl acetate, and the organic phases were combined and dried with anhydrous sodium sulfate. After concentration, the product 3a was obtained by column chromatography with a mixed solvent of petroleum ether and ethyl acetate. The yield was 96%. Pale yellow solid, Mp: 74-75℃; 1 H NMR (600MHz, CDCl 3 ):δ9.26(s,1H),8.66-8.63(m,4H),7.63-7.59(m,2H),7.57-7.54(m,4H); 13 C NMR (150MHz, CDCl 3 ): δ171.3, 166.7, 135.5, 132.8, 128.9, 128.7.
Embodiment 2
[0023]
[0024] Add 1a (4mmol), sodium difluorochloroacetate (4mmol), sodium carbonate (8mmol) and acetonitrile (10mL) into the reaction flask, then heat the reaction at 120°C for 24 hours. After the reaction was completed, it was first quenched with water, then extracted with ethyl acetate, and the organic phases were combined and dried with anhydrous sodium sulfate. After concentration, the product 3a was obtained by column chromatography with a mixed solvent of petroleum ether and ethyl acetate. The yield was 83%. Pale yellow solid, Mp: 74-75℃; 1 H NMR (600MHz, CDCl 3 ):δ9.26(s,1H),8.66-8.63(m,4H),7.63-7.59(m,2H),7.57-7.54(m,4H); 13 C NMR (150MHz, CDCl 3 ): δ171.3, 166.7, 135.5, 132.8, 128.9, 128.7.
Embodiment 3
[0026]
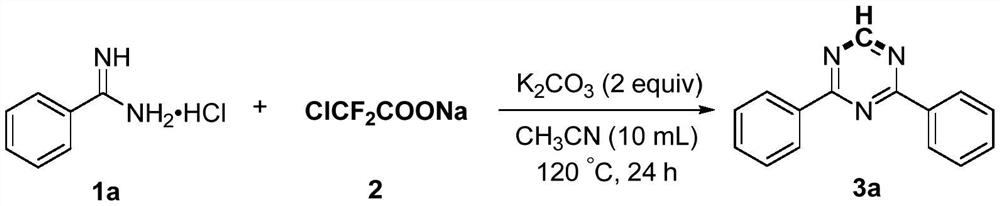
[0027] Add 1a (4mmol), sodium difluorochloroacetate (4mmol), potassium carbonate (8mmol) and acetonitrile (10mL) into the reaction flask, then heat the reaction at 120°C for 24 hours. After the reaction was completed, it was first quenched with water, then extracted with ethyl acetate, and the organic phases were combined and dried with anhydrous sodium sulfate. After concentration, the product 3a was obtained by column chromatography with a mixed solvent of petroleum ether and ethyl acetate. The yield was 78%. Pale yellow solid, Mp: 74-75℃; 1 H NMR (600MHz, CDCl 3 ):δ9.26(s,1H),8.66-8.63(m,4H),7.63-7.59(m,2H),7.57-7.54(m,4H); 13 C NMR (150MHz, CDCl 3 ): δ171.3, 166.7, 135.5, 132.8, 128.9, 128.7.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More