Exogenous mitochondrial vector and compound and preparation method and application of exogenous mitochondrial vector and compound
A mitochondrial carrier, mitochondrial technology, applied in the field of biomedicine
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
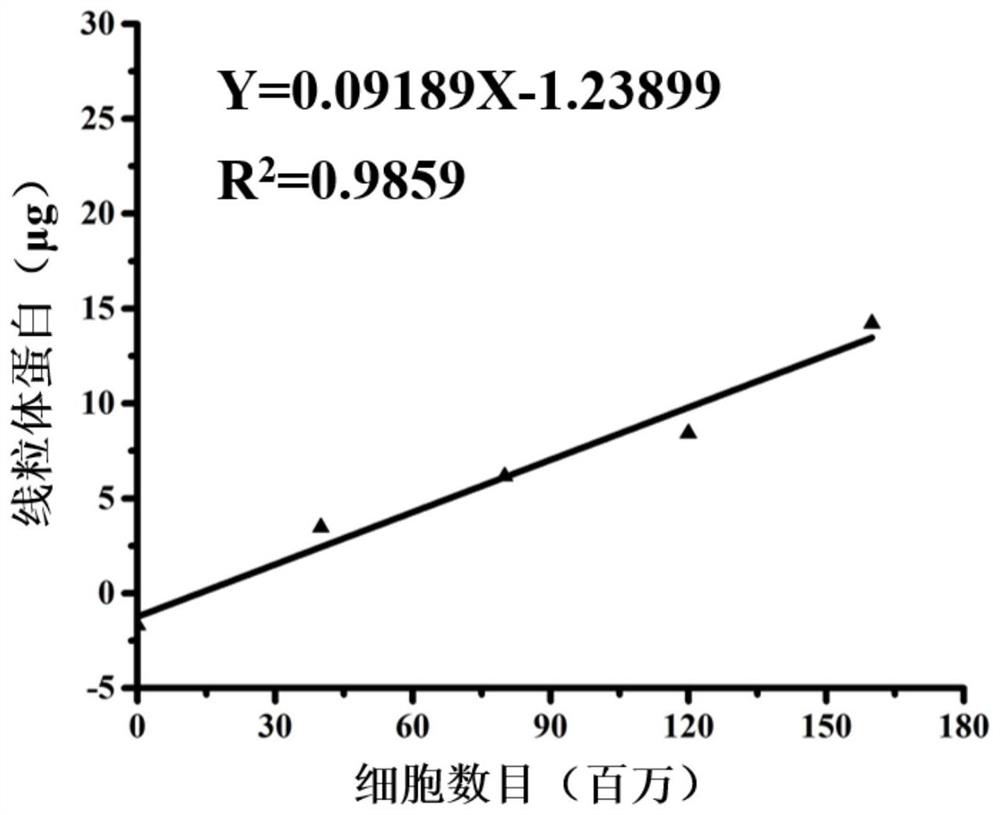
[0034] The mitochondrial donors in this example are selected from various tumor cell lines such as HeLa, MCF-7, HepG-2, L0 2 , LX 2 , H293T and other normal cell lines, and one or more of autologous tissues and organs. Extract exogenous mitochondria Mito using the Mitochondria Extraction Kit according to the instructions. The isolated exogenous mitochondria were lysed, and the mitochondrial protein content was detected using the BCA protein kit, and finally the linear relationship between the mitochondrial protein content and the number of cells was determined.
Embodiment 2
[0036] The preparation method of above-mentioned coating-mito and coating-mito / DNA specifically comprises the following steps:
[0037] 1) Isolated exogenous mitochondria with complete biological functions are extracted from animal tissues or cells cultured in vitro to become mito.
[0038] 2) Prepare a material solution with good biocompatibility, and the final concentration range is 0.1-10 mg / ml.
[0039] 3) Resuspend the isolated exogenous mitochondria in 50-200 μL of a material solution with good biocompatibility, wherein the volume of the material solution with good biocompatibility is determined according to the amount of extracted exogenous mitochondria.
[0040] 4) Incubate the isolated exogenous mitochondria and the material solution with good biocompatibility at room temperature for 30 min.
[0041] 5) Centrifuge at 14,000 rpm for 10 min to obtain a precipitate, and wash the precipitate with phosphate-buffered saline (PBS) several times to obtain the isolated exogen...
Embodiment 3
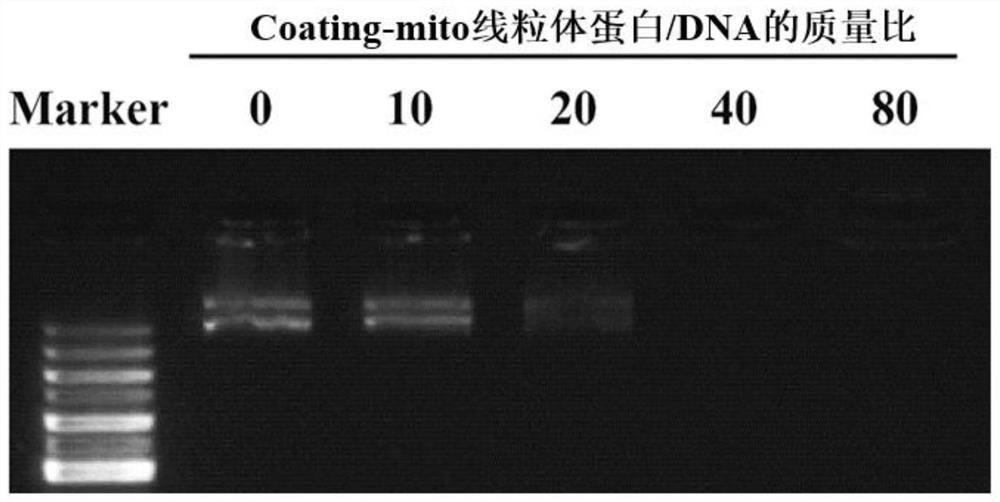
[0044] Calculate the number of mitochondrial protein from the coating-mito obtained in Example 2 according to the linear graph of mitochondrial protein and cell number in Example 1, and dilute it to 2 μg / μL, 4 μg / μL, 8 μg / μL and 16 μg / μL, vortexed with 2 μL (0.2 μg / μL) gene solution for 10 s, and incubated at room temperature for 30 min to obtain the coating-mito-DNA complex. Finally, the binding ability of the material and the gene was detected by gel electrophoresis experiment. Experimental results such as figure 2 As shown, it shows that coating-mito binds to DNA at a mass ratio of 40:1.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More