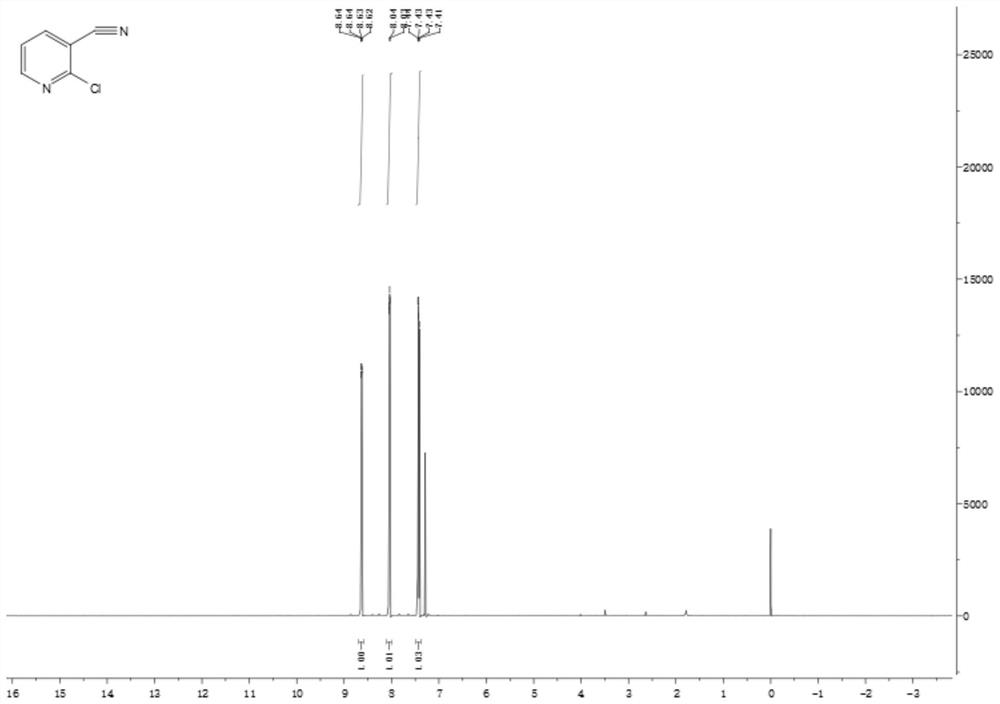
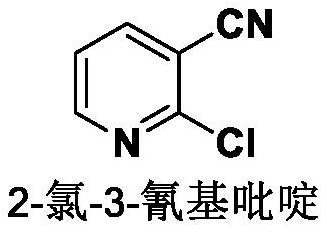
Preparation method of 2-chloro-3-cyanopyridine
A cyanopyridine, phosphorus oxychloride technology, applied in the direction of organic chemistry and the like, can solve the problems of poor selectivity, inability to prepare 2-chloro-3-cyanopyridine with high selectivity, and low yield, etc., and achieve high selectivity High performance, high yield and simple operation
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
preparation example Construction
[0028] The invention provides a preparation method of 2- chloro-3-cyanopyridine, which comprises the following steps:
[0029] Dissolve 3-cyanopyridine N- oxide in phosphorus oxychloride, drip organic alkali at 0-5℃ and control the pH of the system to 9.5-10.5, and then heat up the system to obtain 2- chloro-3-cyanopyridine.
[0030] The above preparation method can be represented by the following reaction formula:
[0031]
[0032] According to the invention, 3-cyanopyridine N- oxide is used as a raw material and phosphorus oxychloride is used as a chlorinating agent, so that 3-cyanopyridine N- oxide is directly dissolved in excessive phosphorus oxychloride for chlorination reaction without using a solvent; and organic alkali is added into the reaction system to neutralize the acid generated in the chlorination reaction, and the organic alkali is dripped at a low temperature to control the reaction speed and avoid the generation of excessive by-products.
[0033]The inventor fo...
Embodiment 1
[0060] The preparation method of 2- chloro-3-cyanopyridine in this example is as follows:
[0061] 1) preparation of 3-cyanopyridine N- oxide
[0062] Add 500 g (4.80 mol) of 3-cyanopyridine and 1600g of concentrated sulfuric acid into a 3,000 ml four-necked flask, raise the temperature to 90℃ to reflux, slowly drop 400mL of 30% hydrogen peroxide into the reaction system in the reflux state, and continue the reflux reaction for 2 hours after dropping, so as to detect that the raw material 3-cyanopyridine has been completely converted;
[0063] The reaction solution was cooled to room temperature and filtered to obtain a solid product, which was dried and weighed at 50℃ to obtain 548g of 3-cyanopyridine N- oxide with a yield of 95%.
[0064] 2) Preparation of 2-chloro-3-cyanopyridine
[0065] Add 500 g (4.16 mol) of 3-cyanopyridine N- oxide and 2000g(13.04mol) of phosphorus oxychloride into a 3000mL four-necked flask, stir, and drop 3 mol equivalents of cyclohexylamine at 200g per ...
Embodiment 2
[0071] The preparation method of 2- chloro-3-cyanopyridine in this example is as follows:
[0072] 1) preparation of 3-cyanopyridine N- oxide
[0073] Add 500 g (4.80 mol) of 3-cyanopyridine and 1600g of concentrated sulfuric acid into a 3,000 ml four-necked flask, raise the temperature to 90℃ to reflux, slowly drop 400mL of 30% hydrogen peroxide into the reaction system in the reflux state, and continue the reflux reaction for 2 hours after dropping, so as to detect that the raw material 3-cyanopyridine has been completely converted;
[0074] The reaction solution was cooled to room temperature and filtered to obtain a solid product, which was dried and weighed at 50℃ to obtain 548g of 3-cyanopyridine N- oxide with a yield of 95%.
[0075] 2) Preparation of 2-chloro-3-cyanopyridine
[0076] Add 500g(4.16mol) of 3-cyanopyridine N- oxide and 2000g(13.04mol) of phosphorus oxychloride into a 3000mL four-necked flask, stir, and drop 3 mol equivalents of pyridine at 200g per hour in an...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More