Nanometer antibody targeting rotavirus protein and application thereof
A nanobody and rotavirus technology, applied in the field of antibody technology and biomedicine, can solve the problems of high immunogenicity, long production cycle and weak penetration ability.
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0022] Example 1. Panning of Nanobodies Targeting Rotavirus Proteins by Phage Displaying Nanobody Random Library
[0023] The phage display library was subjected to affinity panning in the manner of binding, washing, elution and amplification, and the VP8 protein of rotavirus was used as the antigen for panning anti-rotavirus nanobodies. In the experiment, non-specific adsorption was reduced by alternately unblocking and 5% milk blocking, and the coating concentration of viral protein was 10 μg / mL. The specific steps of panning are as follows: add 100 μL of viral protein antigen at a concentration of 10 μg / mL (1 μg / well) to the wells of the eight-continuous-well enzyme-labeled strip, and coat at 37°C for 2 hours. Discard the supernatant, wash the enzyme-labeled strip 5 times with 0.05% PBST, add 200 μL of 5% milk to each well when blocking, and block at 37°C for 2 hours. Discard the blocking solution, wash the enzyme-labeled strips 5 times with 0.05% PBST, and add 100 μL of p...
Embodiment 2
[0024] Embodiment 2. Identification of positive clones
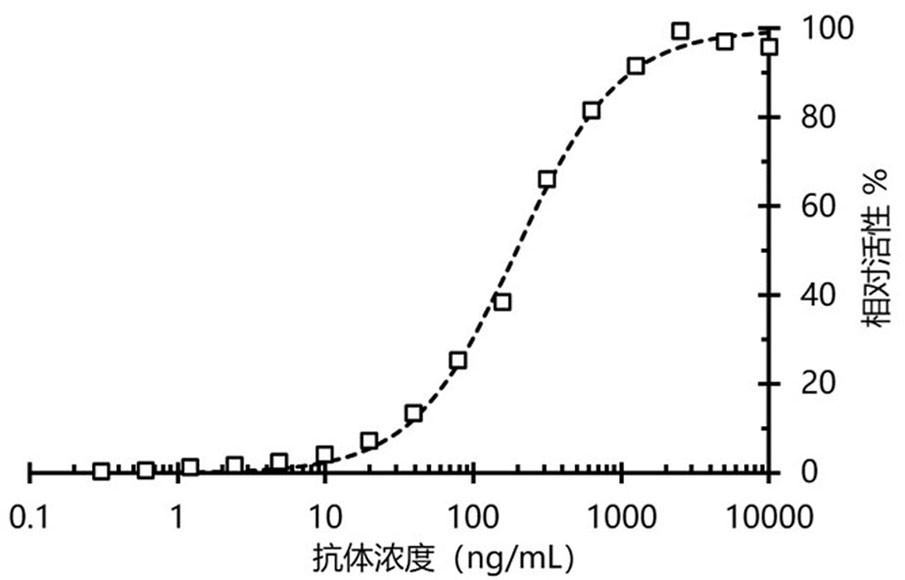
[0025] A single colony was randomly picked from the titered plate for phage rescue and purification, and then positive clones were identified by ELISA. The specific steps are as follows: add 100 μL of viral protein antigen at a concentration of 10 μg / mL (1 μg / well) to the wells of the eight-continuous-well enzyme labeling strip, and coat at 37°C for 2 hours. Discard the supernatant, wash the enzyme-labeled strips 5 times with PBST, add 200 μL of 5% milk to each well, and make another strip as a negative control, block at 37°C for 2 hours. The enzyme-labeled strips were washed 5 times with PBST, 100 μL of amplified monoclonal phage suspension was added to each well, and incubated at room temperature with shaking on a shaker for 1 hour. The plate was washed 5 times with PBST, 100 μL of mouse-derived anti-HA primary antibody was added to each well, and incubated for 1 hour at room temperature with shaking on a shaker. The...
Embodiment 3
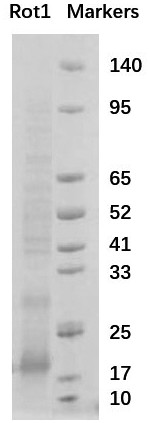
[0026] Example 3. Fusion of Nanobody Rot1 with Histidine Tag and Human Influenza Virus Hemagglutinin Tag
[0027] In the phage display library, the nanobody is fused to the capsid protein P3 of the M13 phage and displayed on the surface of the phage particle. We cut the base sequence of the obtained Nanobody Rot1 into gene fragments with restriction enzymes NdeI and XhoI, and inserted them into the pET23a plasmid, and introduced a histidine tag and human influenza virus hemagglutinin at the C-terminus of the Nanobody. The tag is convenient for the separation, purification and identification of recombinant proteins using nickel columns. The C-terminal amino acid sequence of the Nanobody Rot1 fusion protein is: GQAGQHHHHHHHGAYPYDVPDYALEHHHHHH*.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More