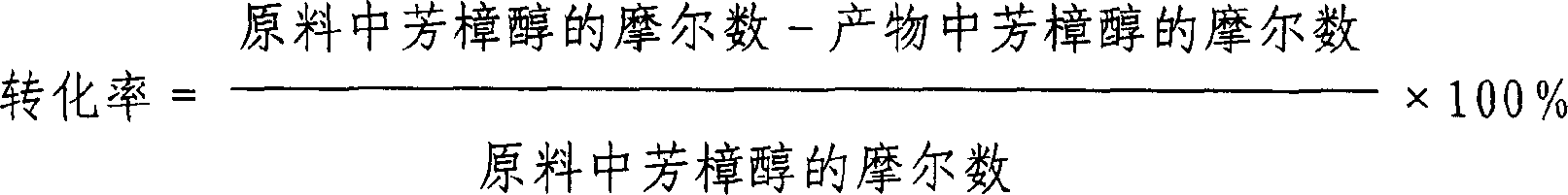Preparation method of linalyl acetate
A technology of linalyl acetate and acetic anhydride, applied in the field of preparation of linalyl acetate, can solve problems such as low linalyl acetate yield, difficult separation and recovery of products, and increased production cost of linalyl acetate
Inactive Publication Date: 2006-02-15
SINOPEC SHANGHAI PETROCHEMICAL CO LTD
View PDF0 Cites 0 Cited by
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Because linalool is an allyl tertiary alcohol, it is unstable in the presence of protonic acid, and isomerization, cyclization and elimination reactions easily occur, generating a large amount of by-products, resulting in a very low yield of linalyl acetate, generally only Can reach 40%
Relatively speaking, the yield of linalyl acetate is higher by using a basic catalyst such as dimethylaminopyridine, but this basic catalyst is dissolved in the reaction solution, and it is difficult to separate and recover from the product, which not only increases the yield of linalyl acetate. Production costs, but also affect the quality of the product
Method used
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
View moreImage
Smart Image Click on the blue labels to locate them in the text.
Smart ImageViewing Examples
Examples
Experimental program
Comparison scheme
Effect test
Embodiment 1~9
[0014] In the 250ml there-necked flask placed on the heating oil bath, drop into linalool and acetic anhydride according to the envisaged molar ratio, and then add the required amount of catalyst p-toluenesulfonic acid. The temperature was raised to the reaction temperature under stirring and maintained for 10 to 150 minutes. After cooling, wash with dilute lye and water to remove the catalyst in the reaction product, and then obtain the product linalyl acetate through vacuum distillation.
[0015] The specific reaction conditions of each embodiment are shown in Table 1, and the reaction results are shown in Table 2.
[0016] temperature reflex
[0017] Conversion rate(%)
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More PUM
 Login to View More
Login to View More Abstract
The invention discloses a process for preparing linalyl acetate from linalool and acetic anhydride as raw material through esterification reaction at the presence of catalyst, the raw material mol ratio of linalool / acetic anhydride is 1 / 1-4, the reaction temperature is 5-45 deg. C, the reaction pressure is atmospheric pressure, the reaction time is 10-150 min, the catalyst is p-toluene sulfonic acid, the consumption of catalyst is 0.05-0.25 wt% of the total amount of the reactants. Compared with the prior art, the advantage of the invention is high catalyst activity.
Description
Technical field [0001] The invention relates to a method for preparing linalyl acetate through esterification reaction using linalool and acetic anhydride as raw materials. Background technique [0002] Linalyl acetate is a valuable spice that is widely used in the spice and food flavor industries. Linalool can be reacted with a suitable acylating agent such as acetyl chloride, ketene, acetic acid or acetic anhydride to obtain linalyl acetate. Since the raw material for preparing linalool comes from the carbon 5 fraction of the by-product of petroleum cracking to produce ethylene, and ethanol Acid anhydride has strong acylating ability and is relatively cheap. Therefore, the preparation of linalyl acetate from the reaction of linalool and acetic anhydride is the most economically advantageous artificial synthesis method of linalyl acetate. [0003] In the prior art, the esterification reaction of linalool and acetic anhydride uses acidic or alkaline catalysts, such as H 3 ...
Claims
the structure of the environmentally friendly knitted fabric provided by the present invention; figure 2 Flow chart of the yarn wrapping machine for environmentally friendly knitted fabrics and storage devices; image 3 Is the parameter map of the yarn covering machine
Login to View More Application Information
Patent Timeline
 Login to View More
Login to View More Patent Type & Authority Patents(China)
IPC IPC(8): C07C69/145C07C67/08
Inventor 朱志庆吕自红郭世卓刘亚涛顾超然胡霞美
Owner SINOPEC SHANGHAI PETROCHEMICAL CO LTD