[0006]Described herein are compositions and methods for the treatment of pain and
inflammation using corticosteroids. The compositions and methods provided herein use one or more corticosteroids in a
microparticle formulation. The
corticosteroid microparticle formulations provided herein are effective at treating pain and / or
inflammation with minimal long-term side effects of
corticosteroid administration, including for example, prolonged suppression of the HPA axis. The corticosteroid microparticle formulations are suitable for administration, for example, local administration by injection into
a site at or near the site of a patient's pain and / or
inflammation. The corticosteroid microparticle formulations provided herein are effective in slowing, arresting, reversing or otherwise inhibiting structural damage to tissues associated with progressive
disease with minimal long-term side effects of corticosteroid administration, including for example, prolonged suppression of the HPA axis. The corticosteroid microparticle formulations are suitable for administration, for example, local administration by injection into
a site at or near the site of structural
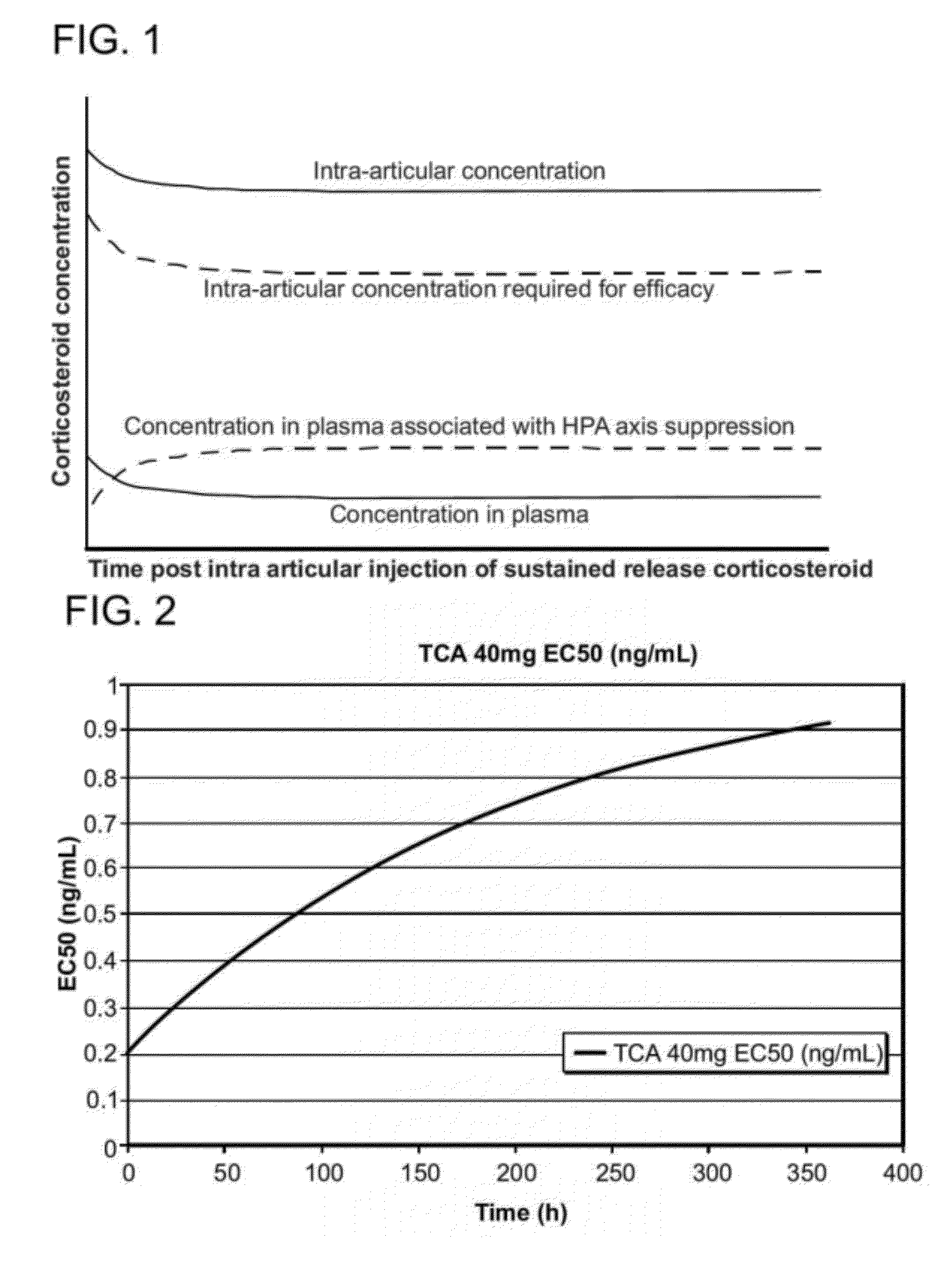
tissue damage. As used herein, “prolonged” suppression of the HPA axis refers to levels of cortisol suppression greater than 35% by day 14 post-administration, for example post-injection. The corticosteroid microparticle formulations provided herein deliver the corticosteroid in a
dose and in a controlled or sustained release manner such that the levels of cortisol suppression are at or below 35% by day 14 post-administration, for example post-injection. In some embodiments, the corticosteroid microparticle formulations provided herein deliver the corticosteroid in a
dose and in a controlled or sustained release manner such that the levels of cortisol suppression are negligible and / or undetectable by 14 post-administration, for example post-injection. In some embodiments, the corticosteroid microparticle formulations provided herein deliver the corticosteroid in a
dose and in a controlled or sustained release manner such that the levels of cortisol suppression are negligible at any time post-injection. Thus, the corticosteroid microparticle formulations in these embodiments are effective in the absence of any significant HPA axis suppression. Administration of the corticosteroid microparticle formulations provided herein can result in an initial “burst” of HPA axis suppression, for example, within the first few days, within the first two days and / or within the first 24 hours post-injection, but by day 14 post-injection, suppression of the HPA axis is less than 35%.
[0007]In certain embodiments, a sustained release form of corticosteroids is administered locally to treat pain and inflammation. Local administration of a corticosteroid microparticle formulation can occur, for example, by injection into the intra-articular space, peri-articular space, soft tissues, lesions,
epidural space, perineural space, or the foramenal space at or near the site of a patient's pain. In certain embodiments, the formulation additionally contains an
immediate release component. In certain preferred embodiments of the invention, a sustained release form of corticosteroids is administered (e.g., by
single injection or as sequential injections) into an intra-articular space for the treatment of pain, for example, due to osteoarthritis, rheumatoid
arthritis,
gouty arthritis,
bursitis,
tenosynovitis, epicondylitis,
synovitis or other
joint disorder. In certain preferred embodiments of the invention, a sustained release form of corticosteroids is administered (e.g., by
single injection or as sequential injections) into soft tissues or lesions for the treatment of inflammatory disorders, for example, the inflammatory and pruritic manifestations of corticosteroid-responsive dermatoses such as
psoriasis. In certain preferred embodiments of the invention, a sustained release form of corticosteroids is administered (e.g., by
single injection or as sequential injections) into an
epidural space, a perineural space, a foramenal space or other spinal space for the treatment of corticosteroid-responsive degenerative musculoskeletal disorders such as Neurogenic
Claudication. In certain preferred embodiments of the invention, a sustained release form of corticosteroids is administered (e.g., by single injection or as sequential injections) into an intra-articular space or into soft tissues to slow, arrest, reverse or otherwise inhibit structural damage to tissues associated with progressive disease such as, for example, the damage to
cartilage associated with progression of osteoarthritis.
[0008]In certain embodiments of the invention, a combination of an immediate release form and a sustained release form of corticosteroids is administered (e.g., by single injection or as sequential injections) into an intra-articular space for the treatment of pain, for example, due to osteoarthritis, rheumatoid
arthritis or other
joint disorder(s). In certain embodiments of the invention, a combination of an immediate release form and a sustained release form of corticosteroids is administered (e.g., by single injection or as sequential injections) into an intra-articular space or into soft tissues to slow, arrest, reverse or otherwise inhibit structural damage to tissues associated with progressive disease such as, for example, the damage to
cartilage associated with progression of osteoarthritis. The formulations and methods of embodiments of the invention can achieve immediate relief of the acute symptoms (e.g., pain and inflammation) of these diseases or conditions and additionally provide a sustained or long term therapy (e.g., slowing, arresting, reversing or otherwise inhibiting structural damage to tissues associated with progressive disease), while avoiding long term systemic side effects associated with corticosteroid administration, including HPA suppression.
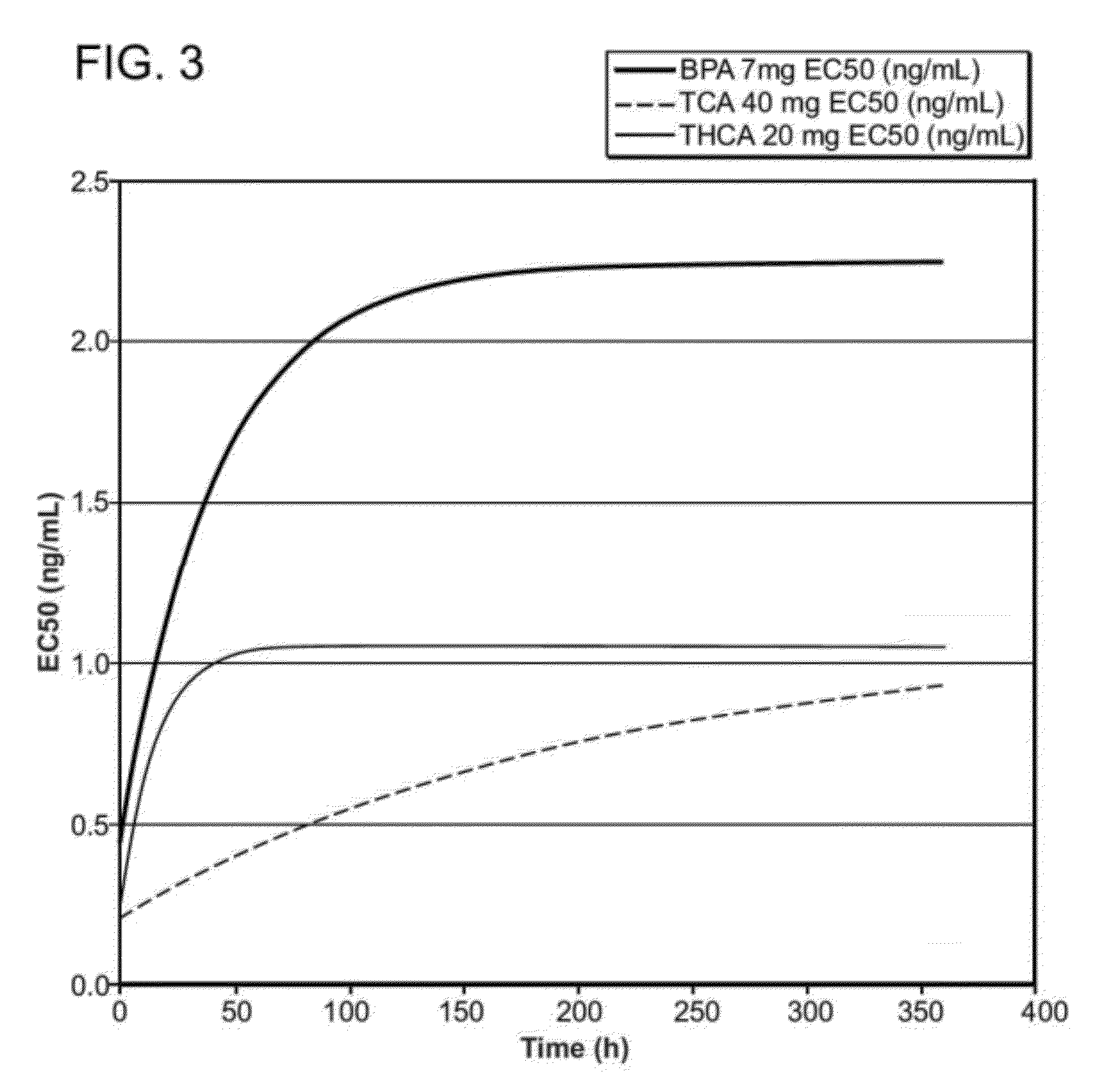
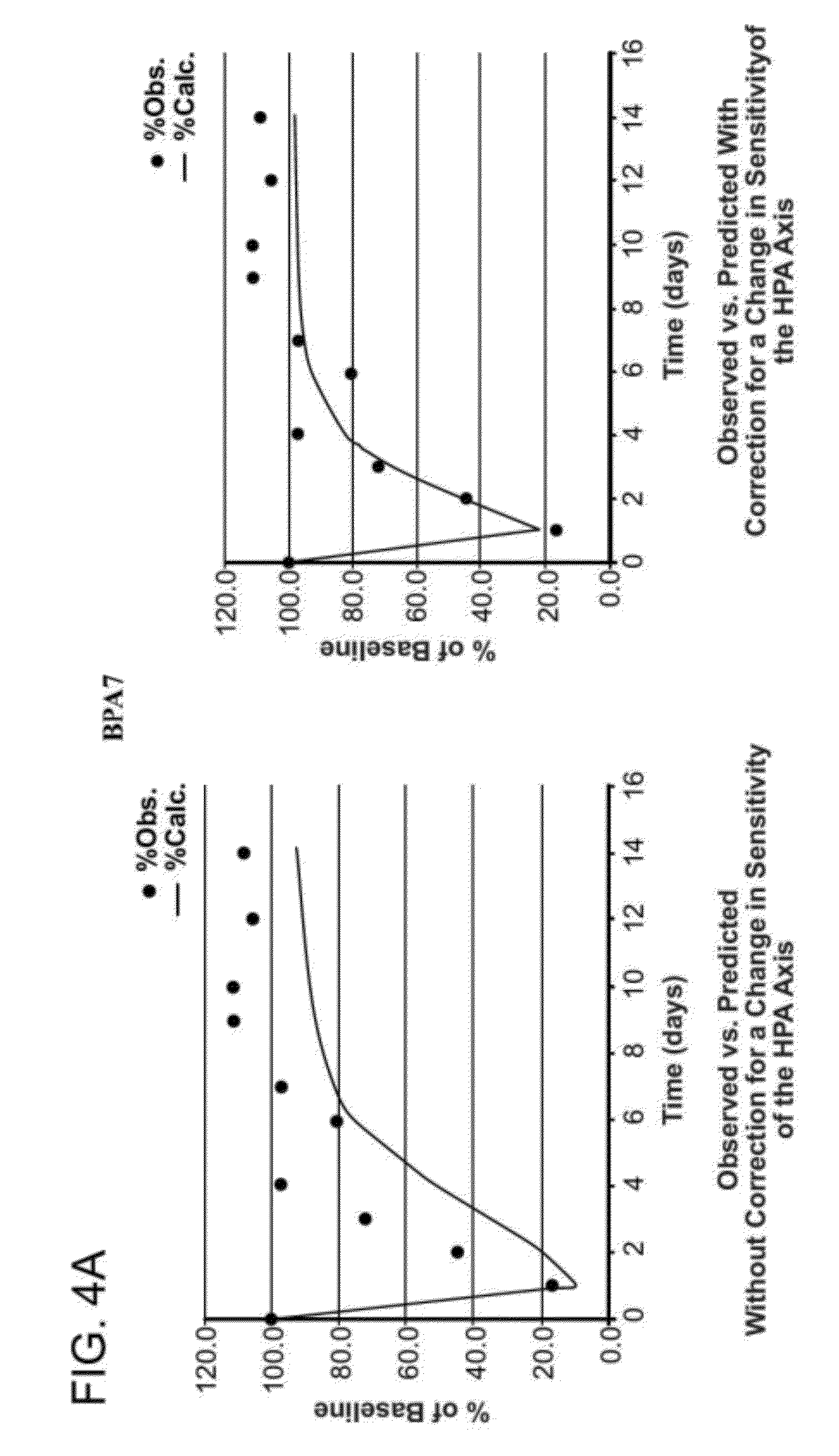
[0010]The corticosteroid microparticle formulation retains sustained
efficacy even after the corticosteroid is no longer resident at the site of administration, for example, in the intra-articular space, and / or after the corticosteroid is no longer detected in the
systemic circulation. The corticosteroid microparticle formulation retains sustained efficacy even after the corticosteroid microparticle formulation is no longer resident at the site of administration, for example, in the intra-articular space, and / or the corticosteroid microparticle formulation is no longer detected in the
systemic circulation. The corticosteroid microparticle formulation retains sustained efficacy even after the corticosteroid microparticle formulation ceases to release therapeutically effective amounts of corticosteroid. For example, in some embodiments, the corticosteroid released by the microparticle formulation retains efficacy for at least one week, at least two weeks, at least three weeks, at least four weeks, at least five weeks, at least six weeks, at least seven weeks, at least eight weeks, at least nine weeks, at least twelve weeks, or more than twelve-weeks post-administration. In some embodiments, the corticosteroid released by the microparticle formulation retains efficacy for a time period that is at least twice as long, at least three times as long, or more than three times as long as the residency period for the corticosteroid and / or the corticosteroid microparticle formulation. In some embodiments, the sustained,
steady state release of corticosteroid will not adversely suppress the HPA axis.
[0021]These Class B corticosteroid microparticle formulations, preparations, and populations thereof, when administered to a patient, exhibit reduced undesirable side effects
in patient, for example, undesirable effects on a patient's
cartilage or other structural tissue, as compared to the administration, for example administration into the intra-articular space of a joint, of an equivalent amount of the Class B corticosteroid absent any microparticle or other type of incorporation, admixture, or encapsulation.
[0032]These TCA microparticle formulations, preparations, and populations thereof, when administered to a patient, exhibit reduced undesirable side effects
in patient, for example, undesirable effects on a patient's cartilage or other structural tissue, as compared to the administration, for example administration into the intra-articular space of a joint, of an equivalent amount of TCA absent any microparticle or other type of incorporation, admixture, or encapsulation.
 Login to View More
Login to View More