Equine disease model for herpesvirus neurologic disease and uses thereof
a neurologic disease and herpesvirus technology, applied in the field of herpesvirus neurologic disease equine disease model, to achieve the effects of high experimental production rate, easy determination or readily titration, and immune protection level
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Benefits of technology
Problems solved by technology
Method used
Image
Examples
examples 1-4
Materials and Methods
Experimental Animals:
[0062]The experimental sample of animals used in these studies consisted of 36 adult horse mares ranging in age from 4 to 28 years. Twenty-five of the experimental horses were Thoroughbred broodmares donated to the M. H. Gluck Equine Research Center (Department of Veterinary Science, University of Kentucky, Lexington, Ky.) by local breeding farms, while the remaining 11 mixed-breed mares were provided from the experimental horse herd maintained by the University of Kentucky's equine research farm. None of the horses had been vaccinated for EHV-1 during the year preceding their use in these studies. For experimental infection with EHV-1, the horses were kept as 3 randomly assigned groups of 12 animals / group in half-acre, grassy lots at the University's equine isolation facilities. Daily health inspections, feeding and watering, preventive health activities (e.g., fly and parasite control, hoof care, vaccinations, etc.), and handling and restr...
example 1
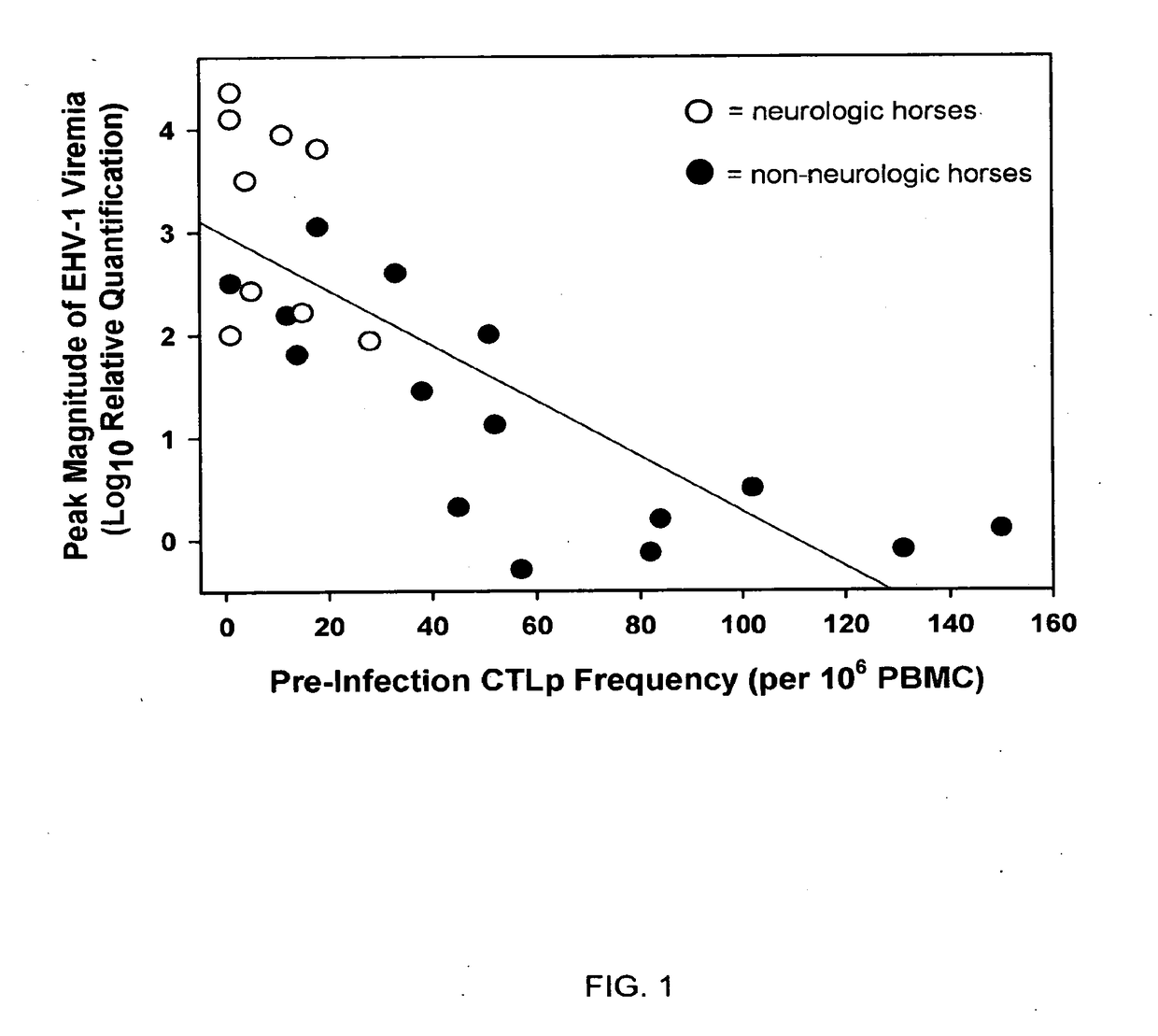
Relationship Among CTLp Frequency, EHV-1 Viremia and Development of Neurologic Disease
[0073]To summarize the study design, 36 horse mares were used for all of the studies described herein. The 24 elderly horses (>20 years) were randomly assigned to either of two experimental groups of 12 horses each (groups A and B). The remaining 12, younger horses (7 PFU (plaque forming units) of a neuropathogenic strain of EHV-1 (T953). The 12 group B horses were inoculated with 107 PFU of a non-neuropathogenic strain of EHV-1 (T262). Statistical comparisons of response variables (e.g., development of neurologic signs, magnitude of viremia, etc.) were made between experimental horse groups that differed in one or more predictor variables (e.g., challenge strain of EHV-1, age-category, breed, pre-exposure levels of CTLp or SNA titers, etc.).
[0074]Nine of the 24 horses inoculated with the neuropathogenic strain of EHV-1 (Ky T953) developed clinical signs of central nervous system disease. Grade 2 n...
example 2
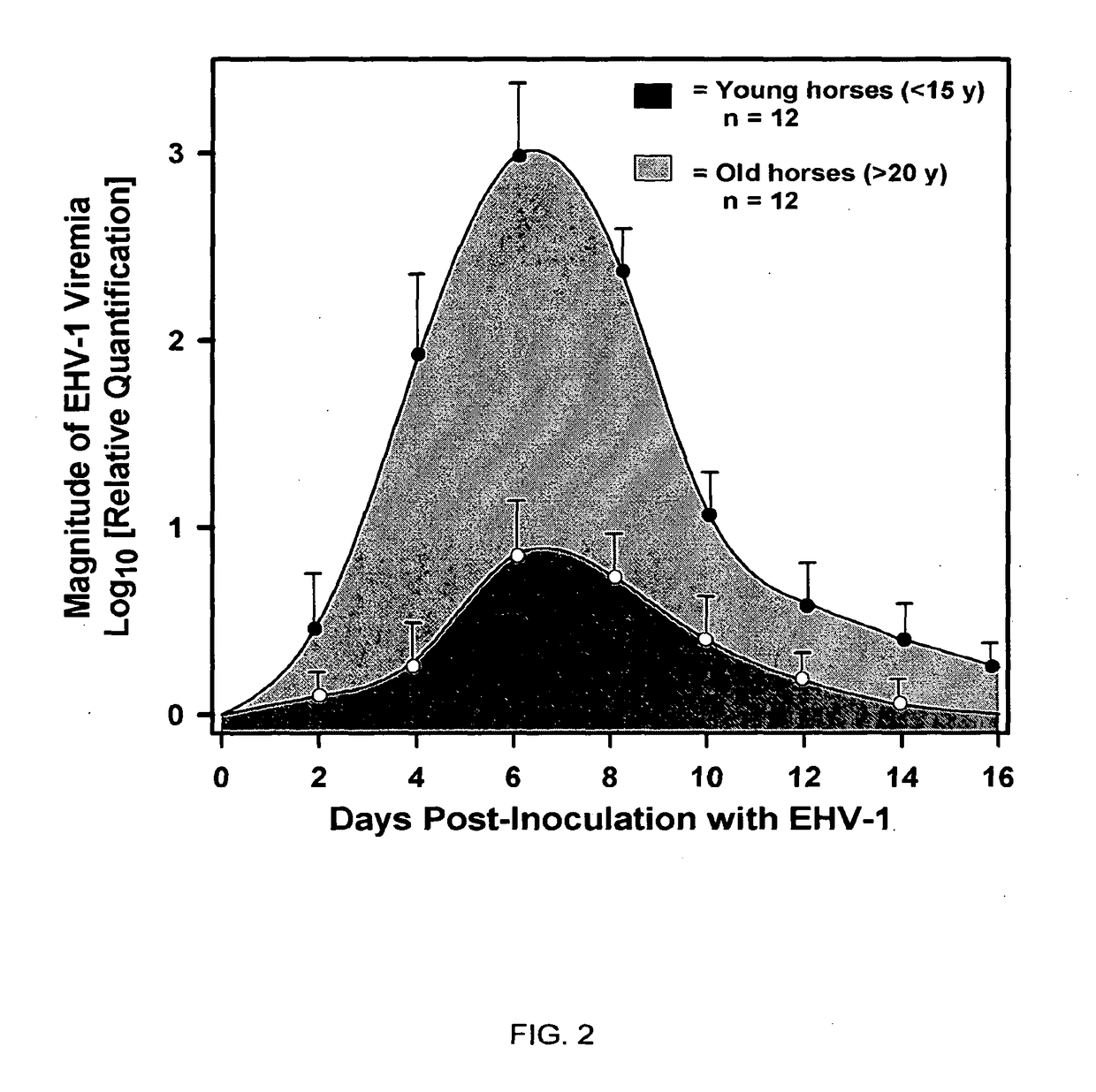
Relationship Between Age of EHV-1 Exposed Horse and Development of Neurological Disease
[0077]The association between “mare age-category” and post-inoculation development of “clinical signs of EHV-1 neurological disease” was examined. The null hypothesis was that, following inoculation of mares of different age categories with neuropathogenic EHV-1, the incidence of neurological disease would be the same in young (20 years) age categories of horses.
[0078]To test the hypothesis, 12 mares belonging to each of the two age groups (20 years) were inoculated intranasally with 107 PFU (plaque forming units) of a neuropathogenic strain of EHV-1 (T953) and observed twice daily for 3 weeks for neurologic signs. Only one mare from the <15 y age category exhibited post-inoculation neurologic signs consisting of hindlimb incoordination and dribbling of urine (grade 2) that necessitated the placement of an indwelling urinary catheter. The mare made a full recovery after 2 weeks. In contrast, 8 of ...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More