Compositions and methods for inhibiting adhesions
A composition and group technology, applied in botanical equipment and methods, pharmaceutical formulations, animal repellants, etc., can solve problems such as adhesion, insufficient flexibility, and incompatibility of laparoscopic procedures
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
example 1
[0256] Example 1: Preparation and Characterization of Hyaluronic Acid Derivatives and Crosslinked Hydrogels
[0257] Materials and Methods
[0258] Hyaluronic acid: Hyaluronic acid (HA, nominally 1.36 MD: high MW and 490 kD: medium MW) was purchased from Genzyme Corporation (Cambridge, MA). HA (nominal 50 kD: low MW) was purchased from Lifecore Biomedical, Inc. (Chaska, MN). All other reagents were purchased from Sigma-Aldrich (St. Louis, MO, USA).
[0259] Preparation of crosslinkable hyaluronic acid: In situ crosslinkable HA derivatives were synthesized following previously reported methods (JiaX, Colombo G, Padera R, Lang Langer R, Kohane DS. Prolongation of sciatic nerve blockade by in situ cross-linked hyaluronic acid. (Biomaterials) 2004; 25(19):4797-4804, which is incorporated herein by reference). Briefly, by reacting HA (medium MW unless otherwise stated) with a 30-fold molar excess of adipate dihydrazide in the presence of 1-ethyl-3-carbodioxide at pH 6.8 and r...
example 2
[0272] Example 2: Biocompatibility of HAX hydrogels in vitro
[0273] Materials and Methods
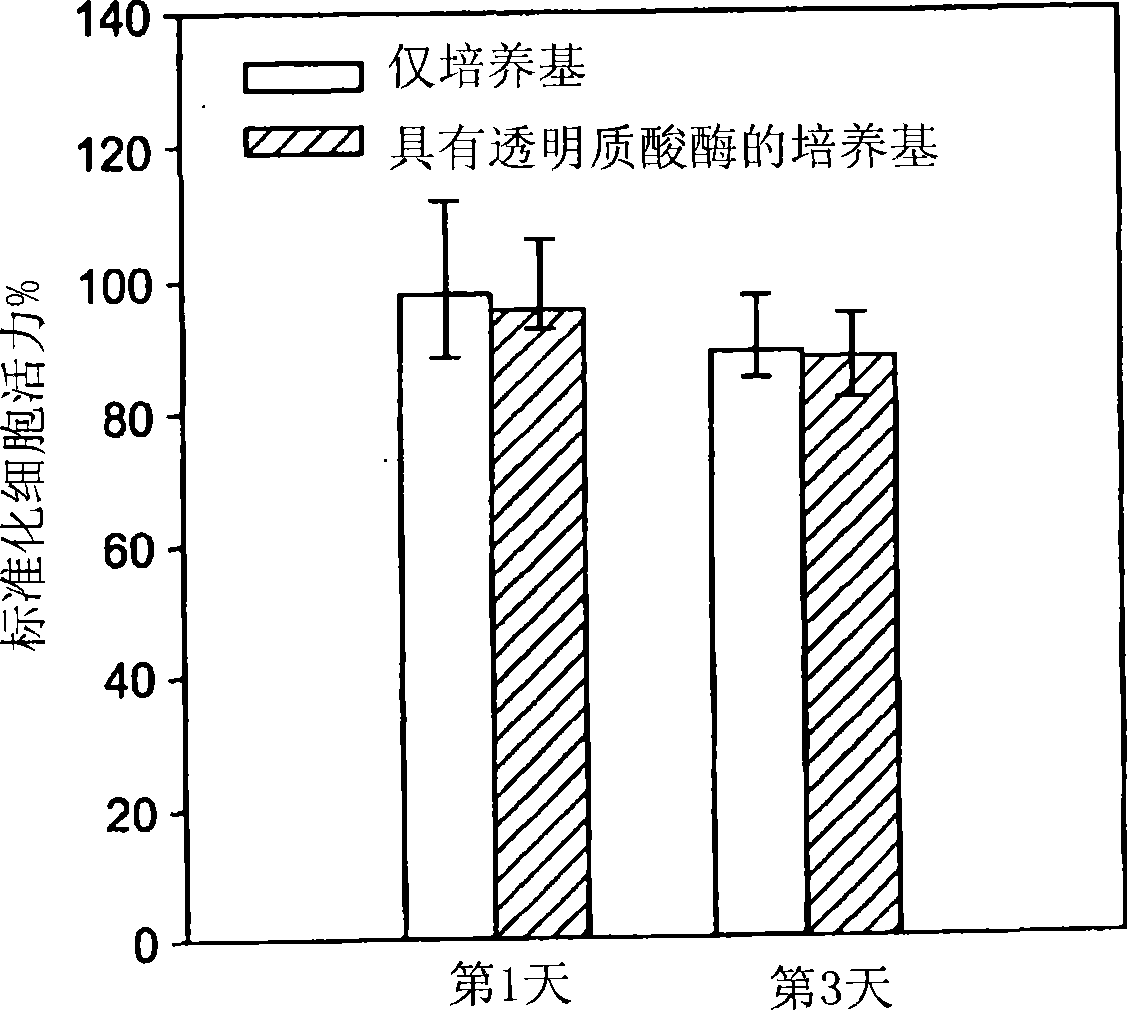
[0274] In vitro cell viability analysis: Human mesothelial cells (ATCC, CRL-9444) were cultured in a medium containing Earle's salt, L-glutamine and 2.2g / L sodium bicarbonate supplemented with 3.3nM In medium 199 of epidermal growth factor, 400 nM hydrocortisone, 870 nM insulin, 20 mM HEPES and 10% fetal bovine serum. Cells from passage 5 to passage 25 were used in the following studies. Mesothelial cells were seeded at a density of 50,000 cells per well in 1 ml of medium in 24-well culture plates. After overnight incubation, the medium was replaced with fresh medium or fresh medium with 10 U / ml hyaluronidase, and 100 μl of cylindrical (diameter: 5 mm, height: 5.1 mm) HAX gel (20 mg / ml) was aseptically prepared. ml) and added to each well. Cell viability was assessed using the MTT assay kit (Promega CellTiter 96 Non-Radioactive Cell Proliferation Assay) after incubation in the p...
example 3
[0277] Example 3: Prevention of peritoneal adhesions by in situ cross-linking of HAX gels
[0278] Materials and Methods
[0279] In vivo administration of HAX gel was performed according to protocols approved by the Massachusetts Institute of Technology Committee on Animal Care, following NIH guidance on the care and use of laboratory animals (NIH Bulletin #85-23, revised 1985 ) care for animals. Female albino rabbits (Oryctolagus cuniculus; New Zealand White, Covance, Hazleton, PA) (3±0.5 kg) were used as model animals . Anesthesia was induced with Ketamine (35 mg / kg, intramuscular) and Xylazine (5 mg / kg, intramuscular); with 1-3% isoflurane in oxygen administered via an endotracheal tube Alkanes (isoflurane) to achieve maintenance. Use aseptic technique throughout the procedure. Animals were given lactated Ringer's solution throughout the procedure and vital signs were continuously monitored. A 10 cm long midline incision was made along the linea alba on the abdomin...
PUM
| Property | Measurement | Unit |
|---|---|---|
| Concentration | aaaaa | aaaaa |
| Concentration | aaaaa | aaaaa |
| Concentration | aaaaa | aaaaa |
Abstract
Description
Claims
Application Information
 Login to View More
Login to View More