Human tnfα monoclonal antibody, its pegized nanoparticle and application thereof
A monoclonal antibody and nanoparticle technology, applied in the field of biotechnology and medicine
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0024] Example 1 Obtaining of B cells secreting anti-human TNFα antibody
[0025]Take 5 milliliters of peripheral blood from patients with active rheumatoid arthritis, separate leukocytes with lymphocyte separation medium, culture them, and identify positive clones according to the results of ELISA.
[0026] 1. Select blood sample
[0027] In order to obtain human B cells secreting anti-human TNFα, human TNFα (purchased from Shenzhen Jingmei Company) was used to routinely coat a 96-well plate, and each well was coated with 250ng of the protein overnight. Then, it was blocked with 5% skimmed milk powder at room temperature for 2 hours, and the milk powder was prepared with pH7.2 PBS. After washing, 100 microliters of sera from different patients were added and incubated at room temperature for 1 hour. Then add peroxidase-labeled goat anti-human IgG and let stand at room temperature for 1 hour. After washing at least 5 times, add TMD or other chromogenic reagents and treat at...
Embodiment 2
[0045] Example 2 Obtaining of anti-human TNFα antibody gene
[0046] Take 5000-10000 positive clone cells obtained in Example 1, use Trizol (GIBCO) to isolate total RNA, and use MMLV reverse transcriptase (Promega company product) to obtain cDNA. The above operations were carried out in accordance with the manufacturer's instructions. PCR was carried out with the following primers and conditions, and the amplification enzyme used was ClonTech's Pfu to ensure that possible mutations were reduced during amplification.
[0047] Light chain upstream primer: GACATCGAGCTGACCCAGTC (SEQ ID NO: 7)
[0048] Light chain downstream primer: CTAACACTCTCCCCTGTTGAAGC (SEQ ID NO: 8)
[0049] Heavy chain upstream primer: GAGGTGCAGCTGGTGGAGTC (SEQ ID NO: 9)
[0050] Heavy chain downstream primer: CTAGCATGTGTGAGTTTTGTCACAAG (SEQ ID NO: 10)
[0051] PCR conditions:
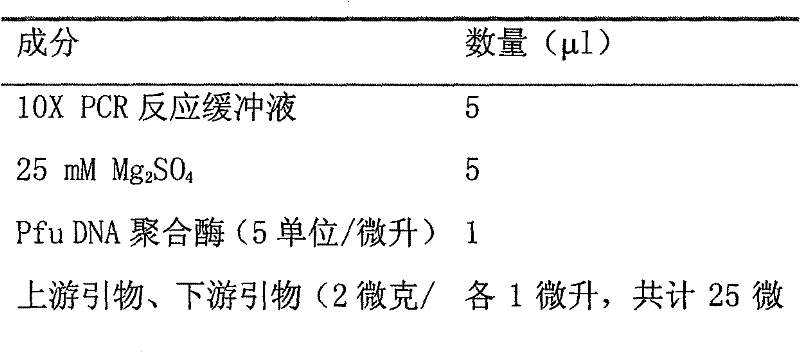
[0052] a) The composition of the reaction system:
[0053]
[0054]
[0055] b) Reaction conditions:
[0056] Pre-denat...
Embodiment 3
[0070] Example 3 Preparation of anti-human TNFα antibody
[0071] 1. Obtaining the coding sequence of human IgG1 heavy chain Fc fragment
[0072] In order to carry out the gene expression of the full-length antibody, we synthesized the heavy chain Fc sequence of human IgG1 in order to constitute the complete anti-human TNFα antibody gene. According to the literature, the DNA coding sequence of the Fc fragment of the human IgG1 heavy chain is as follows:
[0073] DNA sequence of human IgG1 Fc fragment (5'→3') (SEQ ID NO: 5)
[0074] ACATGCCCACCGTGCCCCAGCACCTGAACTCCTGGGGGGACCGTCAGTCTTCCTCTTCCCCCCAAAACCCAAGGACACC
[0075] CTCATGATCTCCCGGACCCCTGAGGTCACATGCGTGGTGGTGGACGTGAGCCACGAAGACCCTGAGGTCAAGTTCAAC
[0076] TGGTACGTGGACGGCGTGGAGGTGCATAATGCCAAGACAAAGCCGCGGGAGGAGCAGTACAACAGCACGTACCGTGTG
[0077] GTCAGCGTCCTCACCGTCCTGCACCAGGACTGGCTGAATGGCAAGGAGTACAAGTGCAAGGTCTCCAAACAAAGCCCTC
[0078] CCAGCCCCCATCGAGAAAACCATCTCCAAAGCCAAAGGGCAGCCCCGAGAACCACAGGTGTACACCCTGCCCCCATCC
[0079]CGGGAT...
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More