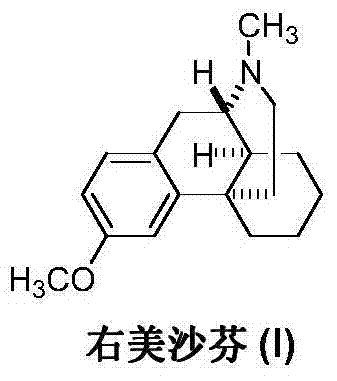
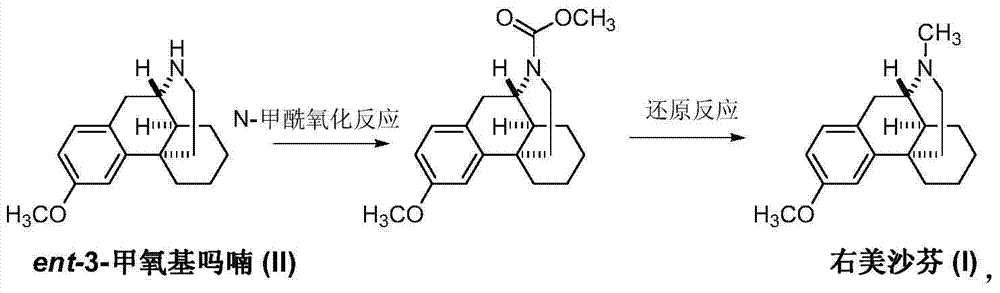
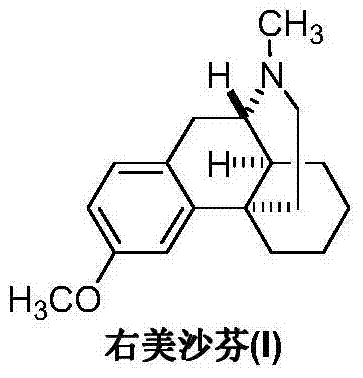
The preparation method of dextromethorphan
A technology of dextromethorphan and white solid, which is applied in the field of preparation of dextromethorphan, can solve the problems of high price of trimethylphenyl ammonium hydroxide, increased production cost of dextromethorphan, and difficulty in obtaining it, so as to promote economic and technological development Effect
- Summary
- Abstract
- Description
- Claims
- Application Information
AI Technical Summary
Problems solved by technology
Method used
Image
Examples
Embodiment 1
[0021] Example 1: Intermediate (II) (25.7 g, 0.1 mol), 40% formaldehyde (15.0 g, 0.2 mol) and 200 mL of methanol were sequentially added into a 500 mL three-necked flask, and reacted with slow stirring for 12 hours. Transfer the reaction solution into a hydrogenation reactor, add 3 g of Raney nickel, keep room temperature and a pressure of 2-4 kg, react for 4 hours, and TLC detects that the reaction is complete. The catalyst was recovered by filtration, the mother liquor was concentrated under reduced pressure, and the residue was recrystallized from methanol to obtain 22.5 g of off-white solid dextromethorphan (I), with a yield of 83.0%.
Embodiment 2
[0022] Embodiment two: add intermediate (II) (25.7g, 0.1mol), paraformaldehyde (6.0g, 0.2mol), solid potassium carbonate (13.8g, 0.1mol), methanol 150mL and Add 50 mL of water, raise the temperature to 65-75°C, and start stirring to react for 6 hours. The reaction solution was transferred to a hydrogenation reactor, 3 g of palladium carbon was added, the temperature was raised to 40-45° C., and the pressure was maintained at 2-4 kg, and the reaction was carried out for 6 hours, and the reaction was detected by TLC. The catalyst was recovered by filtration, the mother liquor was concentrated under reduced pressure, and the residue was recrystallized from methanol to obtain 23.8 g of off-white solid dextromethorphan (I), with a yield of 87.8%.
Embodiment 3
[0023] Example 3: Add intermediate (II) (25.7 g, 0.1 mol), sodium methoxide (10.8 g, 0.2 mol), and 150 mL of methyl formate to a 500 mL three-necked flask in sequence, raise the temperature to 50-55 ° C, and stir the reaction 6 After 1 hour, TLC detected that the reaction was over. The solvent was removed under reduced pressure, and the residue was dissolved in ethanol. Sodium borohydride (4.6 g, 0.12 mol) was added in batches, and the reaction was carried out at room temperature for 12 hours, and the reaction was detected by TLC. The mother liquor was concentrated, and the residue was dissolved in ethyl acetate, washed with dilute hydrochloric acid and water to neutrality, and dried over anhydrous sodium sulfate. Concentrate under reduced pressure, and recrystallize the residue from methanol to obtain 21.6 g of off-white solid dextromethorphan (I), with a yield of 79.6%.
PUM
 Login to View More
Login to View More Abstract
Description
Claims
Application Information
 Login to View More
Login to View More - R&D Engineer
- R&D Manager
- IP Professional
- Industry Leading Data Capabilities
- Powerful AI technology
- Patent DNA Extraction
Browse by: Latest US Patents, China's latest patents, Technical Efficacy Thesaurus, Application Domain, Technology Topic, Popular Technical Reports.
© 2024 PatSnap. All rights reserved.Legal|Privacy policy|Modern Slavery Act Transparency Statement|Sitemap|About US| Contact US: help@patsnap.com